Abstract
In the present study, we introduce a novel, self-organized task-switching paradigm that can be used to study more directly the determinants of switching. Instead of instructing participants to randomly switch between tasks, as in the classic voluntary task-switching paradigm (Arrington & Logan, 2004), we instructed participants to optimize their task performance in a voluntary task-switching environment in which the stimulus associated with the previously selected task appeared in each trial after a delay. Importantly, the stimulus onset asynchrony (SOA) increased further with each additional repetition of this task, whereas the stimulus needed for a task switch was always immediately available. We conducted two experiments with different SOA increments (i.e., Exp. 1a = 50 ms, Exp. 1b = 33 ms) to see whether this procedure would induce switching behavior, and we explored how people trade off switch costs against the increasing availability of the stimulus needed for a task repetition. We observed that participants adapted their behavior to the different task environments (i.e., SOA increments) and that participants switched tasks when the SOA in task switches approximately matched the switch costs. Moreover, correlational analyses indicated relations between individual switch costs and individual switch rates across participants. Together, these results demonstrate that participants were sensitive to the increased availability of switch stimuli in deciding whether to switch or to repeat, which in turn demonstrates flexible adaptive task selection behavior. We suggest that performance limitations in task switching interact with the task environment to influence switching behavior.
Similar content being viewed by others
Our cognitive abilities are fundamentally limited when dealing with multiple cognitive tasks (for reviews, see, e.g., Koch, Poljac, Müller, & Kiesel, in press; Pashler, 2000; Salvucci & Taatgen, 2010). Accordingly, performance costs have been extensively documented not only when people have to perform two tasks simultaneously (i.e., dual-tasking/concurrent multitasking; e.g., Pashler, 1984; Welford, 1952) but also when people alternate between different discrete tasks (i.e., task-switching/sequential multitasking; e.g., Jersild, 1927; Rogers & Monsell, 1995). However, multiple task requirements have become ubiquitous in many different contexts (e.g., González & Mark, 2004; Hembrooke & Gay, 2003; Strayer & Drews, 2007), and thus we can hardly avoid multitasking to some extent. For example, rapidly changing circumstances in an individual’s environment (e.g., changes in task difficulty, task availability, or rewards for task completion) might favor working on tasks other than the currently performed one and thus promote sequential multitasking (e.g., Cohen, McClure, & Yu, 2007; Wisniewski, Reverberi, Tusche, & Haynes, 2015). Given that adequate behavior is usually not externally signaled by the environment, people need to flexibly schedule their activities and they have to decide, for example, when to switch between tasks (e.g., Burgess, Veitch, de Lacy Costello, & Shallice, 2000; Kushleyeva, Salvucci, & Lee, 2005; Payne, Duggan, & Neth, 2007). Thus, successful adaptive multitasking behavior requires our cognitive system to keep track of the environment but also to consider the performance costs that are involved in switching tasks (i.e., switch cost; Rogers & Monsell, 1995). The latter seems especially relevant in light of the fact that multitasking costs can differ between tasks (e.g., Allport, Styles, & Hsieh, 1994; Monsell, Yeung, & Azuma, 2000), can differ between people (e.g., Kray & Lindenberger, 2000; Lawo, Philipp, Schuch, & Koch, 2012; Poljac et al., 2010; Redick et al., 2016; Umemoto & Holroyd, 2016), and can also depend on intra-individual factors such as wakefulness (Bratzke, Rolke, Steinborn, & Ulrich, 2009; Plessow, Kiesel, Petzold, & Kirschbaum, 2011) and stress (Plessow, Kiesel, & Kirschbaum, 2012).
In the present study, we introduce a novel voluntary task-switching paradigm that was designed to shed light on the questions of whether and how people adapt to switch costs. Specifically, participants can freely self-organize their behavior in a task-switching environment that sometimes favors a task switch, in terms of expected objective task performance. This is realized by delaying the appearance of the stimulus for the current task in the following trial and by systematically increasing this delay with the number of task repetitions, until a task switch resets it. In the present experiments, we wanted to know whether this dynamic manipulation of repeat-versus-switch stimulus availability would induce task switches in participants’ choice behavior without the instruction to randomly choose tasks, as is typically done in voluntary task-switching experiments (e.g., Arrington & Logan, 2004). If so, we could also explore how these availability benefits are traded off against (temporal) switch costs.
Task switching
Clearly, switch costs (i.e., responding more slowly and less accurately in a task switch than in a task repetition trial) are a basic and robust phenomenon across different task-switching procedures. Usually the stimuli associated with two separate tasks are presented in each trial and participants are instructed to perform the two tasks in a predictable order (e.g., Rogers & Monsell, 1995) or to follow external task cues (e.g., Koch & Allport, 2006; Meiran, 1996) that indicate which task to perform in a given trial (for reviews, see Kiesel et al., 2010; Monsell, 2003; Vandierendonck, Liefooghe, & Verbruggen, 2010). Of central interest for the present study is that some accounts of the underlying mechanisms producing these switch costs provide a good theoretical foundation to assume that stimulus availability might influence task choice behavior: Although within some accounts switch costs are mainly attributed to active top-down processes that establish a new task set (e.g., Rogers & Monsell, 1995; Rubinstein, Meyer, & Evans, 2001), many researchers agree that the carryover of activation from the preceding task contributes at least partially to worsening task performance in switch trials (e.g., Allport et al., 1994; Altmann & Gray, 2008; Hoffmann, Kiesel, & Sebald, 2003; Meiran, Chorev, & Sapir, 2000, Schuch & Koch, 2003; Sohn & Anderson, 2001; Yeung & Monsell, 2003).
In general, passive activation carryover models of switch costs assume that interfering effects from remaining activation of the task set applied in trial n–1 delay responses when applying a new task set in a switch trial n (e.g., Allport et al., 1994; Gilbert & Shallice, 2002). Strong evidence for the contribution of remaining activation on task switching comes from the findings of so-called “asymmetrical switch costs” (e.g., Allport et al., 1994; Yeung & Monsell, 2003)—that is switch costs are higher when switching from a less familiar, weaker task (e.g., color naming in a Stroop task) to a well-practiced, stronger task (e.g., word reading in a Stroop task) than in the opposite direction. Accounts with activation carryover components to explain switch costs offer a quite natural explanation for this finding by assuming that the weaker task must be activated to a larger degree in order to overcome the tendency to respond to the strong task and that this increased task activation carries over to the next trial, resulting in increased interference when a switch to the strong task is required (e.g., Gilbert & Shallice, 2002; for alternative explanations of asymmetrical switch costs, see Bryck & Mayr, 2008; Schneider & Anderson, 2010).
Importantly, some passive accounts of switch costs strongly imply that a stimulus in the current trial can prime task-set retrieval (e.g., Allport & Wylie, 2000; Waszak, Hommel, & Allport, 2003) and involuntarily activate a task depending on the decay of task-set activation from the preceding trial (e.g., Gilbert & Shallice, 2002; Koch & Allport, 2006; Yeung & Monsell, 2003). Furthermore, the response congruency effects found in many task-switching studies (e.g., Kiesel, Wendt, & Peters, 2007; Koch & Allport, 2006; Meiran & Kessler, 2008; Schneider, 2015a, 2015b, 2017; Yeung, 2010) basically suggest that both relevant and irrelevant task sets are active in a given trial (see Koch, 2001, for a similar suggestion of simultaneous task activation in an incidental task sequence-learning switching paradigm), so that a corresponding task-irrelevant stimulus is automatically processed to a certain degree and interferes with controlled-task processing of the relevant stimulus, based on the current task set. Recently, Schneider (2015a, 2015b, 2017) even provided empirical evidence that these response congruency effects cannot be explained merely by irrelevant (and interfering) response retrievals based on stimulus–response links. Instead, the corresponding irrelevant stimulus is translated into a response via the corresponding task set, which implies the existence of dual-task processing—an account that seems compatible with models of dual-task limitations that allow for parallel central processing of multiple tasks (e.g., Mittelstädt & Miller, 2017; Navon & Miller, 2002; Tombu & Jolicœur, 2003). Thus, it seems quite reasonable to assume that increasing the availability of a stimulus per se might also promote the activation of a task set related to this stimulus. Crucially, as we describe in more detail below, several findings from the voluntary task-switching (VTS) paradigm provide evidence that tasks are often selected on the basis of the most active task set (e.g., Arrington & Logan, 2005).
Voluntary task switching
In the VTS paradigm, each task is usually mapped to one hand and participants select which task they want to perform on a given trial (e.g., Arrington & Logan, 2004; Poljac & Yeung, 2012; Vandamme, Szmalec, Liefooghe, & Vandierendonck, 2010). For example, participants may be instructed to categorize a number as odd or even by pressing the index or middle finger of the left hand and to categorize a letter as vowel or consonant by pressing the index or middle finger of the right hand. In each trial, a letter and a number are presented simultaneously and thus participants can choose to perform either the number task or the letter task using their corresponding response hand. Importantly, however, their task choices are restricted to some degree by global instructions. Specifically, participants are typically instructed to perform both tasks equally often and in a random sequence. These instructions are used to provide sufficient numbers of switches and repeats, because without these instructions participants only switch tasks rarely (Arrington & Reiman, 2015; Arrington, Reiman, & Weaver, 2014; Kessler, Shencar, & Meiran, 2009), which indicates that participants tend to prefer task repetitions when they have full control over task transitions.
Notably, the preference for task repetitions is even present in VTS studies with randomness instructions: Although participants generally comply well with the instructions to perform each task equally often, the order of tasks is not random. Instead, participants tend to repeat tasks more often than expected by chance (e.g., Arrington & Logan, 2005; Dignath, Kiesel, & Eder, 2015; Masson & Carruthers, 2014; Reuss, Kiesel, Kunde, & Hommel, 2011; Yeung, 2010). This so-called repetition bias is especially remarkable because it stands in contrast to the finding that when generating random binary sequences, there is a tendency to alternate more often than to repeat (Nickerson, 2002; Rapoport & Budescu, 1997).
Thus, the finding of a repetition bias suggests that task-switching limitations can be reflected in task choice as well as task performance (i.e., robust switch costs that are typically found; e.g., Arrington & Logan, 2005; Mayr & Bell, 2006). A straightforward causal interpretation of these two findings (i.e., repetition bias and switch costs) is that people prefer task repetitions because task switches are experienced as more time-consuming and/or more effortful (e.g., Arrington & Logan, 2005; Mittelstädt, Dignath, Schmidt-Ott, & Kiesel, 2018; Vandierendonck, Demanet, Liefooghe, & Verbruggen, 2012), which is compatible with the underlying claim that people adapt to switch costs when selecting tasks.
However, the repetition bias provides only an indirect hint about people’s voluntary switching behavior, and it remains unclear whether the avoidance of switching reflects a constant repetition bias or instead is specifically determined by the size of the switch costs. In this regard, it is important to highlight how the corresponding task selection mechanisms—that seem to consider the costs of switching tasks—might operate in VTS studies. Arrington and Logan (2005) have proposed that tasks are selected on the basis of the most active task set whenever participants fail to comply with the instruction to select a task on the basis of a mental representation of a random sequence (see also Arrington, 2008; Arrington & Logan, 2005). The assumption that the degree of activation of a task set guides task selection is consistent with the idea of a mechanism that modulates switching depending on the size of the switch costs. Specifically, it seems fair to argue that task switch costs are larger when the previously performed task set is more active than when this task set is less active. Thus, the idea that tasks are selected on basis of the most active task set fits nicely with passive accounts of switch costs in which the most recently applied task set is the most active one (e.g., Allport et al., 1994). Note, however, that this account of task selection behavior does not exclude the involvement of active top-down processes. Indeed, it seems quite conceivable that participants might rely on passive bottom-up processes partly to avoid effortful active top-down processes that are necessary to finally execute a task switch—for example, inhibiting the activation of previously used task sets (e.g., Koch, Gade, Schuch, & Philipp, 2010; Lien & Ruthruff, 2008; Mayr & Keele, 2000) and/or activating new task sets (e.g., Rogers & Monsell, 1995).
Importantly, factors that seem to influence task-set activation also modulate switching behavior. In this respect, two findings provide further hints that switching limitations are reflected in choice behavior by showing that changes in switch costs produce corresponding modulations in switching behavior. First, switch costs and repetition rates decrease when the response–stimulus interval (RSI) between trials increases (e.g., Arrington & Logan, 2005; Liefooghe, Demanet, & Vandierendonck, 2009). Presumably, the previous task set’s activation decays over time resulting in less influence on task choice and task performance in the current trial. Second, the findings of asymmetrical switch costs are also reflected in switching behavior: Participants tend to perform the weaker task more often than the stronger task (Yeung, 2010), presumably because their task choice is guided by the remaining activation of the previously performed task set.
Critically, different types of VTS studies have demonstrated that stimuli can automatically (i.e., in so far as their influence counters the instructed goal to select tasks randomly) influence the selection of a task—most likely because these stimulus-driven effects on task choice somehow increase the activation of the corresponding task set. First, participants are more likely to repeat a task when the stimulus repeats (e.g., Demanet, Verbruggen, Liefooghe, & Vandierendonck, 2010; Mayr & Bell, 2006), and this might be due to the retrieval of the specific association between a stimulus and response that was established in the previous trial (Demanet et al. 2010). Second, stimulus–response congruency effects can also bias participants to select the task that is response congruent to a stimulus in a given trial (Chen & Hsieh, 2013). Third, and probably most relevant for the present purposes, Arrington (2008; see also Arrington & Weaver, 2015; Butler, Arrington, & Weywadt, 2011) systematically investigated the influence of stimulus availability on task selection in VTS by presenting two stimuli with variable stimulus onset asynchrony (SOA). Their results showed that participants were more likely to perform the task associated with the first stimulus and that this likelihood increased with increases in the SOA between the two stimuli. This indicates that small temporal differences between stimuli have an impact on task selection in the VTS paradigm. Taken together, stimulus availability, stimulus repetition, and stimulus–response congruency effects on task choice in VTS studies provide evidence for influences of the environment on voluntary control, because these effects demonstrate that stimuli can influence task selection processes (e.g., Haggard, 2008; Hommel, 2000; Teuchies et al., 2016).
The present approach
Overall, the VTS paradigm has become an important development of standard task-switching procedures for studying self-organized multitasking, because it allows investigating switching performance when people have control over their decisions and it reveals insights into the determinants of task selection. Specifically, the repetition bias and some studies using the VTS paradigm have already indirectly supported the idea that people may be influenced by the difficulty of switching tasks when they select tasks (e.g., repetition bias and its reduction with increased RSI; Arrington & Logan, 2005), presumably because they guide their task selections on the basis of the most active task set (e.g., Arrington, 2008).
However, these findings are only indirect and imprecise measures due to the requirement in the VTS paradigm to select tasks randomly. This instruction requires participants to fulfill a further mental requirement in addition to switching tasks, and this additional requirement might lead to extra cognitive costs. Furthermore, the repetition bias is basically a violation of the randomness instruction, and it seems fair to argue that participants might follow these instructions better when they try to ignore their switch costs as much as possible. Thus, to study the determinants of switching behavior more directly, it seems useful to provide participants with a task environment without any randomness instruction and that directly requires participants to take switch costs into account—if possible—when deciding whether to switch.
In the present study, we implemented these requirements in a new adaptive self-organized task-switching paradigm by explicitly pitting switch stimulus availability against switch costs. Thus, we investigated whether the idea that stimulus characteristics can potentially influence the degree of task activation and influence task selection processes can be used to induce task switches. More precisely, we presented two stimuli associated with separate tasks in each trial, but we delayed the onset of the stimulus for the task most recently performed. Thus, if participants choose to repeat a task, they had to wait longer for the task-relevant stimulus on the next trial, and the stimulus onset asynchrony (SOA) increased further with each additional repetition of that task. The stimulus needed for a task switch was always presented without any delay, so the time between switch and repetition stimuli—and thus the associated benefit for a switch—increased with the number of task repetitions. Whenever a participant switched tasks, the SOA was reset to the first SOA step size. Thus, the trials in any experimental block could be considered as a sequence of individual runs of task repetitions, each of which ended with a switch at a certain SOA (or the block ended).
As was pointed out earlier, it seems reasonable to assume that the two task sets associated with each stimulus are to a certain degree active in each trial throughout the experiment and that task selection behavior is strongly guided by the task set with the highest activation. We assume that tasks are selected as soon as a certain threshold of task-set activations is reached (i.e., task activation-selection threshold) before (final) task processing takes place. Critically, (1) the degree of task-set activations in a given trial n depends on which task has been performed on trial n–1 in such a way that the previously relevant task set is usually the one with the highest activation at the beginning of trial n (i.e., before stimulus onset in trial n) and (2) the degree of task-set activations can be further increased or primed by stimuli during trial n (i.e., after stimulus onset). Following up on the race metaphor proposed by Arrington (2008) to account for the effects of (random) stimulus SOA on task selection behavior, the task activation-selection threshold should be reached earlier for the task associated with the repetition stimulus when stimuli are simultaneously presented (i.e., in each trial SOA = 0 ms). With the adaptive switch stimulus availability manipulation used here, however, the switch stimulus is presented increasingly earlier with further task repetitions, thereby gradually increasing switch-task activation and correspondingly decreasing the chance of a task repetition. In other words, we assume that increasing switch stimulus availability (or decreasing repetition stimulus availability) should increase the probability of the switch task set to win the race (i.e., reaching the task activation-selection threshold first) against the repetition task set. It should be noted that our idea is only based on passive bottom-up processes influencing the activation level of a task set (i.e., task recency in advance of a trial and stimulus-driven effects during a trial). However, active top-down processes may also influence task-set activations in advance of a trial (e.g., inhibiting task-set activation) or modulate the impact of stimulus-driven factors during a trial (e.g., biasing visual attention to one of the two stimuli). We will return to this issue in our General Discussion.
Overall, then, the basic procedure of our experiments resembles the VTS paradigm in that participants can decide which of two tasks to perform in each trial. In contrast to the VTS paradigm, however, we did not use any global instructions to constrain task transitions or choices. Instead, we instructed participants to respond as quickly and accurately as possible from the beginning of each trial. The major goal of the present experiments was to see if under these instructions our procedure is suitable to induce sufficient switches (while of course still observing sufficient repetitions). Naturally, we expected that task-switching costs in RTs would be found in the present paradigm as they have in many other VTS studies (e.g., Arrington & Logan, 2004; Mayr & Bell, 2006).
If the present procedure is suitable for monitoring both switching behavior and switch costs, it would be helpful to examine switching behavior more closely. Specifically, this procedure would allow us to explore how much extra switch stimulus availability (i.e., size of SOA by which repetition stimulus is delayed) would be necessary to elicit task switches in individual runs (i.e., distribution of switches at different SOAs). By using this additional (temporal) measure of task selection behavior (i.e., switch SOA), we can also explore how participants trade off the costs of switching to a new task versus the increasing availability of the stimulus related to the new task (i.e., comparison of temporal switch costs vs. switch SOA).
To further explore how switch costs and task choice are related to each other, we also correlated these two measures across participants. Given that some within-subjects comparisons described above indicate that participants are able to somehow adapt their task-switching behavior to switch costs, one might intuitively also assume that individuals with higher switch costs would have a stronger tendency to repeat tasks. Somewhat surprisingly, however, the repetition bias is typically independent of the switch costs (e.g., Arrington & Yates, 2009; Yeung, 2010) or is only weakly related to these costs (Mayr & Bell, 2006) when correlating these measures across participants. We speculated that the randomness instructions in VTS studies might induce additional task selection strategies that obscure any potential correlations between the measures of switch costs and repetition rates. Thus, we also explored whether this correlation is present when explicitly instructing participants only to optimize their performance. Although the direction of causality cannot be established with this correlation, it would at least demonstrate that task selection and task performance are related when participants are instructed to select tasks for optimal performance.
Experiments 1a and 1b
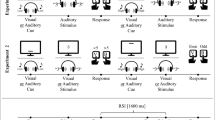
The basic tasks used in these experiments were the number and letter tasks used by Rogers and Monsell (1995). Each task was mapped to the index and middle fingers of one hand. Participants first trained on these tasks in an alternating-runs procedure (see Rogers & Monsell, 1995). Then the main experiments followed, in which participants could choose which task to perform in each trial (see Fig. 1). In these free-choice blocks, the number and the letter stimulus were both presented in each trial, but the SOA between the two stimuli depended on the prior task choices. The stimulus needed for a task switch was always presented first, and the stimulus needed for a task repetition was delayed by an SOA that increased linearly with each repetition of that task. For example, if a participant had performed the letter task on trial n–1 after working on the number task on trial n–2 (i.e., trial n–1 was a switch trial) in Experiment 1a, the letter stimulus was presented with an SOA of 50 ms on trial n. For each consecutive task repetition, the SOA increased linearly (i.e., 100 ms on trial n+1, 150 ms on trial n+2 . . .). Thus, in this setting, task choice behavior could be studied by examining the SOAs in trials on which participants decided to switch (i.e., “switch SOA”). In the example in Fig. 1, a number response on trial n+1 corresponded to a task switch trial, and the switch SOA was 100 ms in Experiment 1a, on the basis of two previous letter responses and the SOA increments used in this experiment.
Typical trial sequences in the free-choice blocks of Experiments 1a and 1b. Stimuli were always presented within the fixation rectangle, but only the stimulus of the previously unselected task was presented immediately after the fixation time of 250 ms. The stimulus of the previously selected task was presented with a stimulus onset asynchrony (SOA) that depended on (a) the previous task choice history (i.e., how often this task had been selected before) and (b) the experiment-specific SOA increase (i.e., 50 ms in Exp. 1a and 33 ms in Exp. 1b). The intertrial interval (ITI) was 500 ms following correct responses
Experiment 1b was similar to Experiment 1a, except that the SOA increase was set to 33 ms per repetition, to see whether a smaller SOA increase would also induce switches and to allow for a more fine-grained measurement of switch SOAs. In the following sections we will describe the methods of these two experiments together and then separately present the results of each experiment. Within the results section of Experiment 1b, we will also provide a comparison of the main measures between the experiments, to show how participants adapted to the slightly different task environments and to check the robustness of the results of Experiment 1a.
Method
Participants
In Experiment 1a, 31 native German speakers (23 female, eight male) were individually tested at the University of Freiburg, Germany. They ranged in age from 19 to 39 years (M = 24.81), and 30 were right-handed. In Experiment 1b, a fresh sample of 32 participants (23 female, nine male) from the same pool was tested. They ranged in age from 19 to 40 years (M = 24.19), and 31 were right-handed. Two additional participants in Experiment 1a and one additional participant in Experiment 1b were tested, but one participant in Experiment 1a was excluded due to accuracy below 60%, and the other two participants did not understand the instructions (assessed by self-report during and after the experiment) and were eliminated prior to any data analysis.
Apparatus and stimuli
Stimulus presentation and the recording of responses were controlled by E-Prime software running on a Fujitsu Eprimo P920 computer with 24-in. monitor. All stimuli were presented in white 25-pt Courier New font on a black background; the stimuli were approximately 7 mm in height and 5 mm in width. The viewing distance was not constrained but was approximately 60 cm. In the free-choice blocks, a white unfilled “fixation rectangle” (13 mm × 13 mm) was always presented centrally. In the training blocks, a 2×2 grid (55 mm in length) was also permanently presented at the center of the screen. In these blocks the fixation rectangle was positioned within the center of one of the four squares of the grid (see the Procedure section). The target stimuli were the numbers 2–9 for the number task (i.e., even/odd) and the uppercase letters A, E, G, I, K, M, R, and U for the letter task (i.e., consonant/vowel). The two stimuli were presented side by side. The specific identities and positions of the two stimuli were selected randomly, with the constraints that each letter and number appeared equally often on each side and that no stimulus was presented twice consecutively. Responses were registered by using two left-sided and two right-sided external response buttons that were separated by a distance of 10 cm. Responses for a task were made with the index and middle finger of the same hand, and the specific mappings were counterbalanced across participants.
Procedure
Each participant was tested in three alternating-runs training blocks of 60 trials per block (180 trials in total), followed by eight free-choice test blocks of 100 trials per block (800 trials in total).
In the training blocks, the stimuli were always presented simultaneously in one square of the grid and the squares switched clockwise between trials (see Rogers & Monsell, 1995). In the free-choice blocks (see Fig. 1), the fixation rectangle appeared on the screen for 250 ms at the beginning of each trial. The stimuli (i.e., one number and one letter) were then presented inside the fixation rectangle. The stimuli were only presented simultaneously in the first trial of a block, whereas in the remaining trials only the stimulus of the previously unselected task was presented immediately after fixation. The other stimulus was presented with an SOA that depended on the length of the current run of responses to this task. In Experiment 1a, the SOA was first 50 ms and increased linearly by 50 ms each time that a task was selected again. In Experiment 1b, the SOA increments were approximately 33 ms.Footnote 1 The stimuli (or stimulus) remained on the screen until a response was made. Following correct responses, the intertrial interval was 500 ms; then the fixation rectangle for the next trial appeared. Following an error, an additional error message was displayed for 500 ms, followed by an instructional screen indicating the stimulus–response mappings for the two tasks for 3,500 ms.
For the training blocks, half of the participants were instructed to perform the letter task when the stimuli appeared in either of the top two squares and the number task when the stimuli appeared in either of the bottom two squares. This assignment was reversed for the other half of the participants.
For the free-choice blocks, participants were instructed that they could freely choose which task to perform, but that they should try to respond as quickly and accurately as possible. Participants were also told that either the letter or number would appear first in a trial, but they were instructed that the total trial time measurement started with onset of the first stimulus. Thus, the total trial time was relevant for participants. Specifically, participants received the following instructions:
You have to perform 100 tasks (i.e., trials) in one block. One of the tasks (i.e., number or letter) appears earlier than the other task in one trial. Reaction time measurement starts with the onset of the first task, and responses can be given after this onset. You can freely decide which task you want to perform in one trial, and it is up to you how often you perform each task in one block. However, try to be as fast as possible without committing errors.
Breaks between blocks were self-paced, and participants received performance feedback (i.e., their mean total trial time and number of errors) after each block.
Results and discussion
Experiment 1a
The training blocks and the first trial of each block were excluded from the analyses. We categorized the task performed on each trial on the basis of the hand used to respond. Then trials were classified as repetition or switch trials on the basis of the task performed on trials n and n–1. The reported reaction times (RTs) always indicate the time from the onset of the stimulus related to the task that the participant performed until the key press. Note again, however, that participants were instructed to minimize the total time in each trial. In switch trials, the total trial time was equal to the RT, whereas in repetition trials the total trial time was the sum of the RT and the trial-specific SOA.
For all analyses, we excluded trials following errors (4.97%) and repetition trials in which participants responded prior to the onset of the repetition stimulus (0.02%) The remaining trials were used for percentage error (PE) analyses. For the task choice and RT analyses, we additionally removed error trials (5.00%), and finally excluded trials with RTs less than 200 ms (0.11%) or greater than 3,000 ms (0.52%) as outliers.
Task choice
We first analyzed how switching behavior changed over the course of the experiment. As can be seen in Fig. 2a, the mean switch rates for the first two blocks were noticeably lower (.28 and .29, respectively) than for the last six blocks (.36–.39). This suggests that participants needed some time to learn the structure of the task environment, but they showed rather stable switching behavior once they had done so. For the following analyses, we excluded the first two free-choice blocks.Footnote 2
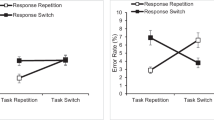
The left panels display the results of Experiment 1a, and the right panels display the corresponding results of Experiment 1b. Panels a and b show the mean switch rates, Pr(switch), across the eight free-choice blocks. Panels c and d show the overall frequency distribution of switch trials across the different SOA levels (Exp. 1a: 50, 150, 200 . . . ; Exp. 1b: 33, 67, 100, . . .). Panels e and f display the cumulative distribution functions of switch SOAs—that is, the probability of switch trials less than or equal to a specific SOA
The overall switch rate of the remaining six blocks was .38 (see Table 1), meaning that the corresponding repetition rate was .62. This switch rate differed from chance (.50), t(30) = 3.60, p = .001. This avoidance of task switches indicates that participants did not just randomly select a task in each trial of the present paradigm. We also checked whether there was any general preference for either the letter or the number task. Participants performed the two tasks equally often, with a mean proportion of .49 (SE = .02) for performing the letter task, and this rate did not differ from chance, p = .684.
Next, we investigated how much stimulus availability was needed to elicit a task switch. For this purpose, we first calculated the frequency distribution of SOAs in switch trials over all participants. As is evident in Fig. 2c, the largest number of switches were already occurring at the first SOA level (i.e., SOA = 50 ms). We then calculated the relative frequency distribution of switch SOAs separately for each participant. Assume, for example, that a participant had 100 switch trials in total, with 30 switches at the first and 50 switches at the second SOA level. This participant would obtain switch proportions of .30 at SOA = 50 ms and .50 at SOA = 100, respectively. Following this, we computed the corresponding individual cumulative distribution function (i.e., this would be .80 at SOA = 100 ms for the participant in our example). Figure 2e displays the cumulative distribution function averaged over all participants. As can be seen in this figure, the mean proportion of switches with an SOA of 50 ms was already .37, and within an SOA of 100 ms or less the proportion of switches exceeded .50. Taken together, these results indicate that the majority of switches were made with very short SOAs.
On the basis of the strongly skewed distribution of switch SOAs, we decided to calculate the median switch SOA for each participant, as a summary measure for task choice behavior as a function of SOA. Note that the median switch SOA for each participant varies in discrete steps according to the corresponding SOA step size used in this experiment (i.e., 50, 100, 150 ms . . .). This means, for example, that a participant with cumulative probabilities of .40 at SOA = 50 ms and .80 at SOA = 100 ms would obtain a median switch SOA of 100 ms. The resulting average median switch SOA was 130 ms (see Table 1).
Task performance
For the RT analyses, we calculated switch costs by using the median switch and repetition RTs, because the use of the median makes the measure of switch costs more comparable to the median switch SOAs. Table 1 shows the averaged median switch RT, the median repetition RT, and the corresponding switch costs (i.e., switch RT – repetition RT). As can be seen in Table 1, the median RTs were on average 118 ms larger on switch than on repetition trials, and these switch costs were reliable, t(30) = 7.81, p < .001. Overall, the PE was low (4.7%), and the PEs did not differ between switch (4.8%) and repetition (4.7%) trials, p = .861.
Relation between task choice and task performance
We then checked how switch SOAs were related to switch costs. As can be seen in Table 1, the median switch SOAs were quite similar to the median switch costs, and a paired t test indicated no reliable difference, p = .566. Thus, this comparison suggests that, on average, participants switched tasks when the switch stimulus availability delay approximately matched the switch costs.
Next, to examine individual differences, we plotted the individual median switch costs against the individual switch rates to explore how switch costs and switching behavior were related across participants. Figure 3a shows the corresponding scatterplot. There was a substantial negative relation between these two measures, r(31) = – .45, p = .010, indicating that switch rates were lower with higher switch costs. The correlation of switch costs and switch SOA across participants was only small and not reliable, r(31) = .13, p = .469. However, as we already mentioned above, the median switch SOA varied in discrete steps, and as is evident in Fig. 3c, many participants had identical median switch SOAs (i.e., 50 or 100 ms). Thus, this discreteness seems to reduce the sensitivity of this variable, which would be needed to detect the potential correlation with the continuous measure of switch costs.Footnote 3
The left panels display the results of Experiment 1a, and the right panels display the corresponding results of Experiment 1b. Panels a and b show scatterplots of individual switch costs against individual switch rates. Panels c and d show scatterplots of individual switch costs against the individual median SOAs of switch trials. Dashed lines represent the corresponding regression lines
Finally, in post-hoc analyses we also considered the data from the training blocks, to get some hints about the causal direction of the correlations between voluntary switch costs and switch rates across participants.Footnote 4 Specifically, we computed the correlations across participants between switch costs in the training blocksFootnote 5 (Mmd = 441 ms) and during voluntary task-switching blocks (see Fig. 4a), and also between switch costs in the training blocks and switch rates in the voluntary task-switching blocks (see Fig. 4b). We found a substantial positive correlation between the individual median switch costs during the training and voluntary task-switching blocks, r(31) = .44, p = .012, indicating that individuals with higher switch costs in the training blocks also had higher switch costs when they had control of their task transitions. Thus, this analysis suggests some individual stability of switching limitations across the two switching procedures (i.e., instructed vs. voluntary). More interestingly, individual training switch costs were negatively and reliably correlated with individual switch rates in the voluntary task-switching blocks, r(31) = – .37, p = .038 [r(31) = .37, p = .041, for the correlation between training switch costs and voluntary median switch SOAs]. Thus, the finding that individuals with higher switch costs during training had lower switching rates in the voluntary blocks suggests that the individual switch costs were a causal factor influencing switching rates.
The left panels display the results of Experiment 1a, and the right panels display the corresponding results of Experiment 1b. Panels a and b show scatterplots of individual switch costs in the training (alternating-run) task-switching blocks against individual switch costs in the voluntary task-switching blocks. Panels c and d show scatterplots of individual switch costs in the training task-switching blocks against individual switch rates in the voluntary task-switching blocks. Dashed lines represent the corresponding regression lines
Experiment 1b
We followed the same data preparation procedure as in Experiment 1a. First, repetition responses prior to repetition stimulus onset were excluded (0.01%). Then, the 5.10% posterror trials were excluded from all analyses. For the RT and choice analyses, the 5.11% error trials were additionally excluded, and we also excluded trials with RTs less than 200 ms (0.04%) and greater than 3,000 ms (0.33%) from these analyses.
Task choice
As can be seen in Fig. 2b, participants’ switch rate was lower for the first block (i.e., .24) than for the other blocks (i.e., .28–.31). This suggests that participants again adapted their switching behavior to the SOA manipulation, and apparently did so slightly faster than in Experiment 1a. For the remaining analyses, the first block was excluded.
The overall switch rate was .30 (i.e., repetition rate of .70), which differed from the .50 switch rate that would be predicted by random task choices, t(31) = 7.04, p < .001. Interestingly, as can be seen in Table 1, participants descriptively switched tasks less often in this experiment than in Experiment 1a (.38), which had a higher SOA step size. However, a two-sample t test indicated that this difference was not quite significant, p = .058. In contrast to Experiment 1a, participants performed the letter task on a higher proportion of trials than the number task (i.e., .54, SE = .02), and this mean probability differed from chance (.50), t(31) = 2.18, p = .037.
Figure 2d shows the frequency distribution of switch SOAs, and again a high number of switches were made at the first SOA level. The difference as compared to the next two SOA levels, however, was not as large as in Experiment 1a. Furthermore, as can be seen in Fig. 2e and f, the mean proportion of switches for the first SOA level (.26) was lower than the corresponding proportion for the first SOA level in Experiment 1a (.37), and this difference was significant, t(61) = 2.86, p = .006. Figure 2f also shows that—similar to Experiment 1a—the majority proportion of switches occurred within the first few SOA levels. Interestingly, the .50 proportion of switches was again exceeded at SOA = 100 ms, as in Experiment 1a. Note that this corresponds to the third SOA level in this experiment, whereas this SOA size had occurred at the second SOA level in Experiment 1a. This suggests that participants differentially adapted their behavior to the different dynamic task environments.
Further hints of this adaptive behavior are provided by comparing the median switch SOAs between the experiments: As can be seen in Table 1, the median switch SOA in Experiment 1b amounted to 112 ms, and was thus only slightly and not significantly reduced, p = .402, in Experiment 1b relative to Experiment 1a.
Task performance
As can be seen in Table 1, RTs were again larger on switch than on repetition trials, and these switch costs of 134 ms were reliable, t(31) = 7.60, p < .001. Overall, the PE was again low (5.1%), and PEs did not differ between switch (5.6%) and repetition (4.6%) trials, p = .394.
We also conducted an analysis of variance (ANOVA) on median RTs, with the between-subjects factor experiment (i.e., Exp. 1a vs. Exp. 1b) and the within-subjects factor transition (i.e., repeat vs. switch), to see whether task performance differed significantly between the two experiments. This ANOVA only yielded a significant main effect of transition (p < .001), and no significant effects of either experiment (p = .649) or the interaction (p = .496). Thus, this analysis does not imply any differences between switch costs in these two slightly different task environments.
Relation of task choice and task performance
As in Experiment 1a, the median switch costs were approximately the same size as the median switch SOAs (see Table 1), and a paired t test yielded no significant differences between these measures, p = .244.
Finally, we explored the relation of switching behavior and switch costs on an individual level. Figure 3b shows a scatterplot of the individual median switch costs against the individual switch rates. We found a negative correlation similar to the one in Experiment 1a, but this correlation was not significant, r(32) = – .30, p = .090. Note, however, that the correlation was driven mainly by the three participants with switch costs higher than 350 ms [i.e., excluding them resulted in r(29) = .02, p = .924]. Similarly, the correlation between switch costs and median switch SOA (Fig. 3d) was significant, r(32) = .35, p = .049, but not when the three outlier participants were excluded, r(29) = – .05, p = .814.
We then again computed correlations with individual median switch costs in the training blocks (Mmd = 452 ms). Similar to Experiment 1a, there was a significant positive correlation between this measure and voluntary switch costs (see Fig. 4b), r(32) = .52, p = .002. Figure 4d displays a scatterplot of the individual training switch costs against the individual switch rates observed in the voluntary blocks. As in Experiment 1a, we observed a negative—although not reliable—correlation between these two measures, r(32) = – .29, p = .112 [r(32) = .41, p = .020, for the correlation between training switch costs and voluntary median switch SOA].
General discussion
In the present study we introduced a new adaptive task-switching paradigm that could be useful to study the determinants of switching behavior more directly—in particular to investigate whether and how individuals adapt to switching limitations in self-organized task-switching. In this paradigm, participants could select which task to perform on each trial, but in contrast to the classic VTS paradigm we used no instructions to select these tasks in a random sequence (e.g., Arrington & Logan, 2004). Instead, the availability of tasks was dynamically adjusted on the basis of an individual’s immediately preceding task selections. More precisely, whereas a potential switch stimulus was always immediately available, the stimulus belonging to the previously selected task was delayed by an amount (i.e., SOA) that increased with each successive repetition of that task. We reasoned that increasing switch stimulus availability should correspondingly increase activation of the switch task set, and we wanted to know whether this would increase the likelihood of task switches.
The findings of two experiments demonstrate that our switch stimulus availability manipulation successfully induced switching behavior. First, participants increased their switching behavior after facing the constraints of the task environment (e.g., higher switch rates after the first blocks). Second, participants showed reasonable switching behavior with overall switch rates of .38 (Exp. 1a) and .30 (Exp. 1b) whereas some previous VTS studies observed little switching behavior without additional global randomness instructions (for a discussion of these global instructions, see Arrington et al., 2014). For example, 50 of 66 participants were removed due to insufficient switches in a study by Arrington and Reiman (2015), and switch rates were less than .14 across all experimental conditions in a free-choice task-switching study by Kessler et al. (2009). Third, in particular the between-experiments comparisons (i.e., a higher overall switch rate and higher switch rate at the first SOA level in Exp. 1a than in Exp. 1b) suggest that participants adapted their switching behavior to the slightly different dynamic task environments (i.e., SOA increases of 50 vs. 33 ms), whereas switch costs remained stable. Together, these findings indicate that participants’ switching behavior was sensitive to the increased delay of repetition-stimulus availability—presumably because switch stimulus availabilities increase activations of potential switch tasks, making it more likely that these tasks are selected.
As was outlined in the introduction, several VTS studies have also found influences of stimulus-driven effects on task choice (e.g., Arrington, 2008; Arrington & Weaver, 2015; Demanet et al., 2010; Mayr & Bell, 2006; Teuchies et al., 2016). For example, our results are particularly in line with Arrington’s (2008) finding that stimulus availability influenced task selection. Here, we extended this finding by showing that steadily increasing switch stimulus availability can induce a task switch. Recent task-switching studies have also successfully induced switching behavior without a randomness instruction by intermixing free- and forced-choice trials (Fröber & Dreisbach, 2017) or by rewarding task switches with points (Braun & Arrington, 2018). Thus, the present study provides further evidence of flexible adaptive switching behavior by showing that people are also able to adapt their task selection behavior to the current task environment, including task availabilities. Considering again the between-experiment comparisons (i.e., differences in switch rates but stable switch costs), the strong avoidance of switching within the first voluntary task-switching block(s), and that afterward a bias to repeat tasks was still present (i.e., tasks were not just randomly selected as one would expect if performance differences between these transitions play no role), it seems very likely that the adaptation to the task environment was also further modulated by switching limitations.
Importantly, the self-organized task-switching paradigm has advantages for exploring the finding of adaptive task selection behavior in more detail while also focusing on task performance, because both switching behavior and switching limitations are measured on a common scale—namely, time. More precisely, our experimental procedure provided us with the opportunity to explore how the temporal costs of task switching are traded off against the temporal benefits of increased switch stimulus availability. Results showed that in the two experiments the median size of SOAs in switch trials was similar to switch costs. Note again that switch costs were constant across experiments and only the external temporal dynamics of stimulus availability changed across experiments. Thus, this suggests that participants tended to switch tasks when the availability benefits matched switch costs—a strategy that helped them to complete the overall block of trials more rapidly.Footnote 6
In retrospect, there is considerable evidence for the idea that the adaptation of behavior to the environment might be driven by cost–benefit considerations in which our cognitive system trades off its own limitations in an optimal manner against environmental constraints (Anderson, 1990; Carlson & Stevenson, 2002; Chater & Oaksford, 1999; Howes, Lewis, & Vera, 2009; Gray, Sims, Fu, & Schoelles, 2006). For example, participants are able to adopt optimal information acquisition strategies in arithmetic tasks when they adapt their involved cognitive processes to small temporal delays in information availability (Carlson & Stevenson, 2002; Stevenson & Carlson, 2003). Furthermore, a study that investigated the neural correlates of task selection in a dynamically changing task environment somewhat similar to the one used in the present experiments has provided hints of a neural connection between task selection and task performance (Wisniewski et al., 2015).Footnote 7 Specifically, in this study participants saw a single stimulus but could choose among three S–R mappings (i.e., “task rules”) on a trial-by-trial basis. Stimulus discriminability was reduced each time the same S–R mapping was repeated, thus giving participants an incentive to switch task rules. The results suggested that the same brain regions (i.e., dorsal medial prefrontal cortex and dorsal anterior cingulate cortex) were involved in both choosing a task rule and monitoring stimulus discriminability. Thus, considering all of this evidence in light of participants’ sensitivity to the temporal delays in the present study, the finding that the participants in our sequential multitasking setting selected trade-offs to minimize the time required to complete a trial supports the idea that self-organized sequential multitasking also involves balancing internal costs (i.e., switch costs) and external benefits (i.e., stimulus availability) in order to optimize task performance. In other words, our results suggest that task selection in self-organized sequential multitasking is partly driven by (expected) task performance. At this point, it should also be emphasized, however, that we had no a priori assumption about the switch cost–SOA trade-offs. Although this finding was observed across two slightly different task environments (i.e., different SOA increments), the observed matches of these two measures in the two experiments might be merely coincidences of some specific design features present in both experiments. Thus, the question of optimal adaptive task selection behavior (in which the trial-specific size of switch costs is equally traded against the trial-specific task availabilities) should be directly tackled in future studies. In addition, the basic idea of dynamic task availabilities should be applied to modified task environments (e.g., different tasks) other than the ones used in the present experiments (i.e., different SOA increments).
So far, we have interpreted the results on a rather functional level—that is, participants adapt to the task environment in such a way that switching limitations interact with stimulus availability to influence switching behavior. Although the question of optimality remains open, one of the issues that emerges in particular from this discussion is how the underlying mechanisms operate to configure task-selection behavior in the current task environment. As was elaborated in the introduction, our approach was primarily motivated by the idea of passive bottom-up factors influencing switching behavior (i.e., priming activation of the switch task set by increasing switch stimulus availability to counteract the carryover activation boost from the repetition task set)—thereby neglecting the involvement of active top-down factors. However, the jump of switch rates after the first block(s) provides a strong suggestion that active top-down processes are also involved in adjusting switching behavior. Unfortunately, albeit also in line with findings from many VTS studies in which task-selection behavior reflects interaction of top-down and bottom-up processes (e.g., Arrington, 2008; Demanet et al., 2010), we currently have no sophisticated account of how these processes interacted in the present experiments to influence task selection and task performance.
In this context, intriguing questions regarding awareness of the processes involved in trading off switch costs and stimulus availability emerge—first and foremost, whether there is some metacognitive awareness of switch costs. Intuitively, the avoidance of task switching (e.g., repetition bias in VTS studies with randomness instruction) indirectly suggests that people must possess some awareness of the costs associated with switching tasks, and this idea seems also to be supported by recent studies in which people’s introspection was sensitive to very small variations in task performance (e.g., Questienne, Atas, Burle, & Gevers, 2018; Questienne, van Dijck, & Gevers, 2018). The idea that metacognitive evaluation of switch costs is a prerequisite factor to “successfully” trading off switch costs against stimulus availability also fits with our post-hoc interpretation of optimal switching behavior. Interestingly, however, people do not seem to be aware of their multitasking costs in dual-task settings (e.g., Bratzke & Bryce, 2016; Bryce & Bratzke, 2014). Nevertheless, participants adapt their dual-task strategies to maximize their overall performance in response to changes in task difficulty (Janssen & Brumby, 2015; Janssen, Brumby, Dowell, Chater, & Howes, 2011; Leonhard, Ruiz Fernández, Ulrich, & Miller, 2011), and they even appear to shift from a more serial to a more parallel processing mode when the likelihood of short SOAs between the two task stimuli increases, which can increase the overall efficiency of task performance (Miller, Ulrich, & Rolke, 2009). Thus, it would be an interesting issue not only to directly examine the introspection of switch costs but also to see whether (and how) this potential metacognitive ability modulates adaptation to the current task environment (e.g., do people with more accurate introspective knowledge select more optimal switch SOA–cost trade-offs than do people with less knowledge?).
A somewhat surprising finding from the present experiments—in particular, in light of the overall matching of switch SOA and switch costs—was that switches were distributed over a wide range of SOAs, with many switches occurring already at the first SOA level. Specifically, from an intuitive point of view, we would expect to observe a more narrow range of switch SOAs with a frequency peak at the size of the switch costs (i.e., at the second or third SOA level). In retrospect, it seems conceivable that switch costs might—similar to the switch SOA—differ substantially between trials, and that very small costs were involved in many trials. Thus, stimulus availability might have had less opportunity to influence task choice in a consistent and systematic manner because of random variability in the size of switch costs. This variability might result from random variations in the task-set activations that, as we described in the introduction, appear to influence both switch costs and task choice behavior. Another possible explanation for the larger number of switches at short SOAs might be that some or all stimuli are directly translated into a response without retrieving the corresponding task set. Thus, switch task processing might start immediately after switch stimulus onset and proceed in parallel with repetition task processing after onset of the repetition stimulus. As we mentioned in the introduction, resource-sharing accounts of dual-task interference even allow for parallel response selection processing (e.g., Mittelstädt & Miller, 2017; Navon & Miller, 2002; Tombu & Jolicœur, 2003). Thus, tasks might also be selected as soon as a certain processing threshold is reached, before final task processing takes place. Without the retrieval of task sets, it seems quite conceivable that even a short switch stimulus head start (i.e., switch SOA) might be sufficient for the switch task to win the race.
However, it also seems very likely that other aspects of the task environment in our experiments influence task choice behavior in addition to stimulus availability. One component is worth mentioning in particular: Stimulus position and thus task location changed randomly on a trial-by-trial basis. Arrington and Weaver (2015) found that participants are more likely to repeat a task when the task location repeats than when it changes and attributed this effect to so-called outsourcing strategies that have already been proposed in a cued task-switching paradigm (Mayr & Bryck, 2007). Indeed, our data show the same pattern, because switch rates were lower when the task location repeated (Exp. 1a = .36, Exp. 1b = .25) than when it changed (Exp. 1a = .40, Exp. 1b = .34), although the difference (i.e., Exp. 1a = .04, Exp. 1b = .09) was reliable only in Experiment 1b (p = .031) and not in Experiment 1a (p = .084).Footnote 8 A possible post-hoc interpretation for the effect of stimulus location on task choice behavior might be that spatial attention is connected more strongly to the location of the previously selected stimulus. Thus, this attentional location bias might additionally boost the effects of stimulus availability and thus task activation.
Furthermore, participants might also guide their behavior partially by task sequences, to avoid actively engaging in—probably effortful—task selection processes on each trial. This would be in line with growing evidence that the cognitive system is biased to avoid effortful processes (e.g., Dunn, Lutes, & Risko, 2016; Kool & Botvinick, 2014; Kool, McGuire, Rosen, & Botvinick, 2010). The idea that task sequences guide task selection behavior is also incorporated in the chain-retrieval model, introduced by Vandierendonck, Demanet, Liefooghe, and Verbruggen (2012) to explain task selection behavior in VTS studies. According to this model, task selection in VTS is guided by task sequences retrieved from long term memory. Importantly, however, this model also suggests that environmental factors can override the task choice retrieved from a chunked task sequence—in line with the findings of many VTS studies (e.g., Arrington, 2008; Mayr & Bell, 2006), as well as the present study, that the precise stimulus conditions can influence task choices. Interestingly, switching tasks in sequences involves hierarchical control processes to initiate these sequences, which modulate switch costs and involve “sequence-switching costs” (Schneider & Logan, 2006a). Thus, the overall adaptation of switch costs to stimulus availability in the present study might also have involved high-level processes—that is, participants selected task sequences that were sensitive to the costs of task switches as well as sequence switches.
Finally, the investigation of the relation of individual switch costs and individual switching behavior provides further hints that task performance (in terms of switch costs) and task selection may reflect similar aspects of cognitive control. Specifically, we observed a correlation of switch costs and switch rates across participants in Experiment 1a and this correlation also seemed to be—although slightly weaker and not reliable—present in Experiment 1b. Although these correlations might suggest that participants’ switching behavior is influenced by their switch costs, it is also possible that the size of switch costs is influenced by switching behavior. Note that many task-switching studies with predetermined task order have found that switch costs decrease as task-switch frequency increases (e.g., Mayr, 2006; Mayr, Kuhns, & Rieter, 2013; Monsell & Mizon, 2006; Schneider, 2016; Schneider & Logan, 2006b). Thus, it is not surprising to observe a correlation between switch costs and switch rates across participants—at least at first glance. As we mentioned in our introduction, however, previous VTS studies with the instruction to select tasks randomly have not observed this correlation (Arrington & Yates, 2009; Yeung, 2010). We think that the present approach of inducing task switches by instructing participants to select tasks in order to optimize performance, while adaptively manipulating the availability of task stimuli (i.e., without any global randomness instruction), provides an interesting avenue to jointly study task selection and task performance within one paradigm. Beyond this rather methodical point of view, post hoc correlational analyses using the training block data (i.e., in which task transitions were predetermined by using the alternating-run procedure) as a baseline estimate of individual switch costs provides some interesting hints about the causal nature of the observed voluntary switch costs-rate correlations. Specifically, we also observed negative correlations between training block switch costs and switch rates in the subsequent VTS blocks in both Experiment 1a and 1b. This finding supports the view that switch costs in the voluntary switching blocks are at least partially responsible for modulating switching behavior.
It should be emphasized, however, that an investigation of individual differences will require larger samples than the ones used in the present study (i.e., 32 and 31 participants in Exps. 1a and 1b, respectively)Footnote 9 and that the correlation in the voluntary switching blocks in Experiment 1b was driven mainly by three participants with high switch costs. Moreover, estimation of individual switch costs with the training blocks data might be subject to extra noise because participants need time to familiarize themselves with the experimental setting and the single tasks. It is also an open question whether the “baseline” measure of switch costs with the alternating-run training procedure used here is suitable to represent switching limitations that are supposedly also present in the voluntary task-switching blocks: Although the substantial positive correlations between training and voluntary switch costs indeed suggest that these limitations rely on similar processes, it should be noted that even switch costs in procedures differing only in how task order is determined (i.e., explicit cuing vs. alternating runs) should not be treated as identical measures (Altmann, 2007), and many studies found that voluntary switch costs are often smaller than switch costs in externally controlled task switching (e.g., Arrington & Logan, 2005; Chen & Hsieh, 2015; Demanet & Liefooghe, 2014; Orr & Weissman, 2011). Thus, all correlational data (i.e., correlations within the voluntary switching blocks and between training and voluntary switching blocks) must be interpreted with caution, and future studies with larger samples are clearly needed if researchers wish to investigate individual differences with the present paradigm. For this purpose, we recommend using switch rates instead of median switch SOAs to capture switching behavior. When median switch SOA varies only in coarse steps, as in the present experiments, it is less suited than the continuous variable of switch rates to this type of analysis.
Conclusion
In the present study, we presented a novel multitasking paradigm in which switching behavior is induced by the increased availability of switch tasks instead of additional randomness instructions as in the classic VTS paradigm (Arrington & Logan, 2004). Specifically, the availability of task stimuli was dynamically adjusted on the basis of individual choices in such a way that the stimulus for the previously selected task was presented with an SOA that increased with additional task repetitions. The results of two experiments with different SOA increments suggest that participants were sensitive to the increased availability of switch stimuli, and that this sensitivity reflects adaptive task selection behavior. Furthermore, the findings that participants tended to switch tasks when the size of the SOA corresponded approximately to their switch costs and that individual switch rates were related to individual switch costs provide further hints that people might consider their switch costs when adapting their multitasking behavior to the environment.
Notes
Due to the screen refresh rate, the SOA increments were always loops of four times 33 ms and then one time 17 ms (i.e., 33, 67, 100, 133, 150, 183, 217 . . .).
In both Experiments 1a and 1b, qualitatively very similar results were also obtained in analyses with other block exclusion procedures (i.e., excluding no blocks, excluding only the first block, and excluding the first two blocks) and in analyses with other data preparation procedures on a trial level (i.e., including posterror trials, including error trials, and including RT outliers).
For this reason, we also explored the correlations between individual mean switch costs (i.e., voluntary and training) and mean switch SOAs in the two experiments. In Experiment 1a, the corresponding correlation between voluntary switch costs (M = 142 ms) and switch SOAs (M = 159 ms) was r(31) = .41, p = .021. The correlation between training switch costs (M = 442 ms) and switch SOA was r(31) = .42, p = .020. In Experiment 1b, the correlation between voluntary switch costs (M = 154 ms) and switch SOAs (M = 148 ms) was r(32) = .33, p = .064. The correlation between training switch costs (M = 432 ms) and switch SOA was r(32) = .30, p = .101.
We thank two reviewers for suggesting this analysis.
Note that this switch cost–availability trade-off seems to be rather consistent with a local strategy to minimize current RTs. It would be also possible to use a proactive switching strategy to complete the overall block of trials faster. More precisely, participants could also switch tasks when the availability benefits were still smaller than the switch costs, to reset the SOA so that they would be affected by lower SOAs on the following repetition trials. This strategy would be more consistent with a global strategy of minimizing mean rather than current RTs. Although the present experiments were not designed to test any optimization accounts, it should be noted that individuals’ strategy selections are often driven mainly by local optimization, at the expense of suboptimal global task performance (Anderson, 1990; Fu & Gray, 2006).
The present study was developed independently of Wisniewski et al.’s (2015) article, and their study was not designed to investigate how switch costs are related to switching behavior. Moreover, several aspects of their experimental procedure and findings make it difficult to derive any post-hoc conclusions about this research question. First, only one stimulus was presented in each trial. The different “tasks” were different S–R mappings of the possible stimulus categories onto response keys. Second, participants indicated which S–R mapping they would use in advance of stimulus onset. This allowed them plenty of time to prepare for task switches, which is known to reduce the cost of switching. Third, the behavioral results indicated no switch costs in task performance—an obvious prerequisite for exploring the question of how participants adapt their switching behavior to their switch costs.
We also visually inspected differences in switch rates between task location switches and task location repetitions separately for each participant: This difference was larger than .50 for one participant in Experiment 1a (i.e., .52 for the difference between switch rates on location switch [.58] and location repetition trials [.06]) and for two participants in Experiment 1b (i.e., .98 [switch rates of .99 on location switch and .01 on location repetition trials] and .96 [switch rates of .98 on location switch and .02 on location repetition trials], respectively) suggesting that these participants consistently used task locations to guide their task selection behavior. Note that excluding these participants revealed similar descriptive patterns in switch rates for task location repetitions (Exp. 1a = .37, Exp. 1b = .26) and task location switches (Exp. 1a = .39, Exp. 1b = .30), and the difference (Exp. 1a = .02, Exp. 1b = .04) was again significant only in Experiment 1b (p = .003) but not in Experiment 1a (p = .119).
The correlation of individual median voluntary switch costs and individual switch rates across the two experiments was r(63) = – .38, p = .002. The correlation of individual median training switch costs and individual switch rates across the two experiments was r(63) = – .32, p = .011.
References
Allport, A., Styles, E. A., & Hsieh, S. (1994). Shifting intentional set: Exploring the dynamic control of tasks. In C. Umiltà & M. Moscovitch (Eds.), Attention and performance XV: Conscious and nonconscious information processing (pp. 421–452). Cambridge, MA: MIT Press.
Allport, A., & Wylie, G. (2000). Task-switching, stimulus–response bindings, and negative priming. In S. Monsell & J. S. Driver (Eds.), Control of cognitive processes: Attention and performance XVIII (pp. 35–70). Cambridge, MA: MIT Press.
Altmann, E. M. (2007). Comparing switch costs: Alternating runs and explicit cuing. Journal of Experimental Psychology: Learning, Memory, and Cognition, 33, 475–483.
Altmann, E. M., & Gray, W. D. (2008). An integrated model of cognitive control in task switching. Psychological Review, 115, 602–639. doi:https://doi.org/10.1037/0033-295X.115.3.602
Anderson, J. R. (1990). The adaptive character of thought. Hillsdale, NJ: Erlbaum.
Arrington, C. M. (2008). The effect of stimulus availability on task choice in voluntary task switching. Memory & Cognition, 36, 991–997.
Arrington, C. M., & Logan, G. D. (2004). The cost of a voluntary task switch. Psychological Science, 15, 610–615.
Arrington, C. M., & Logan, G. D. (2005). Voluntary task switching: Chasing the elusive homunculus. Journal of Experimental Psychology: Learning, Memory, and Cognition, 31, 683–702. doi:https://doi.org/10.1037/0278-7393.31.4.683
Arrington, C. M., & Reiman, K. M. (2015). Task frequency influences stimulus-driven effects on task selection during voluntary task switching. Psychonomic Bulletin & Review, 22, 1089–1095.
Arrington, C. M., Reiman, K. M., & Weaver, S. M. (2014). Voluntary task switching. In J. Grange & G. Houghton (Eds.), Task switching (pp. 117–136). Oxford, UK: Oxford University Press.
Arrington, C. M., & Weaver, S. M. (2015). Rethinking volitional control over task choice in multitask environments: Use of a stimulus set selection strategy in voluntary task switching. Quarterly Journal of Experimental Psychology, 68, 664–679.
Arrington, C. M., & Yates, M. M. (2009). The role of attentional networks in voluntary task switching. Psychonomic Bulletin & Review, 16, 660–665. doi:https://doi.org/10.3758/PBR.16.4.660
Bratzke, D., & Bryce, D. (2016). Temporal discrimination of one’s own reaction times in dual-task performance: Context effects and methodological constraints. Attention, Perception, & Psychophysics, 78, 1806–1816.
Bratzke, D., Rolke, B., Steinborn, M. B., & Ulrich, R. (2009). The effect of 40 h constant wakefulness on task-switching efficiency. Journal of Sleep Research, 18, 167–172. doi:https://doi.org/10.1111/j.1365-2869.2008.00729.x
Braun, D. A., & Arrington, C. M. (2018). Assessing the role of reward in task selection using a reward-based voluntary task switching paradigm. Psychological Research, 82, 54–64. doi:10.1007/s00426-017-0919-x
Bryce, D., & Bratzke, D. (2014). Introspective reports of reaction times in dual-tasks reflect experienced difficulty rather than timing of cognitive processes. Consciousness and Cognition, 27, 254–267.
Bryck, R. L., & Mayr, U. (2008). Task selection cost asymmetry without task switching. Psychonomic Bulletin & Review, 15, 128–134. doi:https://doi.org/10.3758/PBR.15.1.128
Burgess, P. W., Veitch, E., de Lacy Costello, A., & Shallice, T. (2000). The cognitive and neuroanatomical correlates of multitasking. Neuropsychologia, 38, 848–863.
Butler, K. M., Arrington, C. M., & Weywadt, C. (2011). Working memory capacity modulates task performance but has little influence on task choice. Memory & Cognition, 39, 708–724.
Carlson, R. A., & Stevenson, L. M. (2002). Temporal tuning in the acquisition of cognitive skill. Psychonomic Bulletin & Review, 9, 759–765.
Chater, N., & Oaksford, M. (1999). Ten years of the rational analysis of cognition. Trends in Cognitive Sciences, 3, 57–65.
Chen, P., & Hsieh, S. (2013). When the voluntary mind meets the irresistible event: Stimulus–response correspondence effects on task selection during voluntary task switching. Psychonomic Bulletin & Review, 20, 1195–1205.
Chen, P., & Hsieh, S. (2015). At will or not at will: Electrophysiological correlates of preparation for voluntary and instructed task-switching paradigms. Psychonomic Bulletin & Review, 22, 1389–1402.
Cohen, J. D., McClure, S. M., & Yu, A. J. (2007). Should I stay or should I go? How the human brain manages the trade-off between exploitation and exploration. Philosophical Transactions of the Royal Society B, 362, 933–942. doi:https://doi.org/10.1098/rstb.2007.2098
Demanet, J., & Liefooghe, B. (2014). Component processes in voluntary task switching. Quarterly Journal of Experimental Psychology, 67, 843–860.
Demanet, J., Verbruggen, F., Liefooghe, B., & Vandierendonck, A. (2010). Voluntary task switching under load: Contribution of top-down and bottom-up factors in goal-directed behavior. Psychonomic Bulletin & Review, 17, 387–393. doi:https://doi.org/10.3758/PBR.17.3.387
Dignath, D., Kiesel, A., & Eder, A. B. (2015). Flexible conflict management: Conflict avoidance and conflict adjustment in reactive cognitive control. Journal of Experimental Psychology: Learning, Memory, and Cognition, 41, 975–988.
Dunn, T. L., Lutes, D. J., & Risko, E. F. (2016). Metacognitive evaluation in the avoidance of demand. Journal of Experimental Psychology: Human Perception and Performance, 42, 1372–1387.
Fröber, K., & Dreisbach, G. (2017). Keep flexible—Keep switching! The influence of forced task switching on voluntary task switching. Cognition, 162, 48–53.
Fu, W. T., & Gray, W. D. (2006). Suboptimal tradeoffs in information seeking. Cognitive Psychology, 52, 195–242.
Gilbert, S. J., & Shallice, T. (2002). Task switching: A PDP model. Cognitive Psychology, 44, 297–337.
González, V. M., & Mark, G. (2004). “Constant, constant, multi-tasking craziness”: Managing multiple working spheres. In E. Dykstra-Erickson & M. Tscheligi (Eds.), Proceedings of the SIGCHI Conference on Human Factors in Computing Systems (pp. 113–120). New York, NY: ACM Press. doi:https://doi.org/10.1145/985692.985707
Gray, W. D., Sims, C. R., Fu, W. T., & Schoelles, M. J. (2006). The soft constraints hypothesis: A rational analysis approach to resource allocation for interactive behavior. Psychological Review, 113, 461–482.
Haggard, P. (2008). Human volition: Towards a neuroscience of will. Nature Reviews Neuroscience, 9, 934–946.
Hembrooke, H., & Gay, G. (2003). The laptop and the lecture: The effects of multitasking in learning environments. Journal of Computing in Higher Education, 15, 46–64.
Hoffmann, J., Kiesel, A., & Sebald, A. (2003). Task switches under Go/NoGo conditions and the decomposition of switch costs. European Journal of Cognitive Psychology, 15, 101–128.
Hommel, B. (2000). The prepared reflex: Automaticity and control in stimulus–response translation. In S. Monsell & J. Driver (Eds.), Control of cognitive processes: Attention and performance XVIII (pp. 247–273). Cambridge, MA: MIT Press.
Howes, A., Lewis, R. L., & Vera, A. (2009). Rational adaptation under task and processing constraints: Implications for testing theories of cognition and action. Psychological Review, 116, 717–751.
Janssen, C. P., & Brumby, D. P. (2015). Strategic adaptation to task characteristics, incentives, and individual differences in dual-tasking. PLoS ONE, 10, e0130009. https://doi.org/10.1371/journal.pone.0130009
Janssen, C. P., Brumby, D. P., Dowell, J., Chater, N., & Howes, A. (2011). Identifying optimum performance trade-offs using a cognitively bounded rational analysis model of discretionary task interleaving. Topics in Cognitive Science, 3, 123–139.
Jersild, A. T. (1927). Mental set and shift. Archives of Psychology, 14(Whole No. 89), 1–81.
Kessler, Y., Shencar, Y., & Meiran, N. (2009). Choosing to switch: Spontaneous task switching despite associated behavioral costs. Acta Psychologica, 131, 120–128.
Kiesel, A., Steinhauser, M., Wendt, M., Falkenstein, M., Jost, K., Philipp, A. M., & Koch, I. (2010). Control and interference in task switching—A review. Psychological Bulletin, 136, 849–874. doi:https://doi.org/10.1037/a0019842
Kiesel, A., Wendt, M., & Peters, A. (2007). Task switching: On the origin of response congruency effects. Psychological Research, 71, 117–125.
Koch, I. (2001). Automatic and intentional activation of task sets. Journal of Experimental Psychology: Learning, Memory, and Cognition, 27, 1474–1486.
Koch, I., & Allport, A. (2006). Cue-based preparation and stimulus-based priming of tasks in task switching. Memory & Cognition, 34, 433–444. doi:https://doi.org/10.3758/BF03193420
Koch, I., Gade, M., Schuch, S., & Philipp, A. M. (2010). The role of inhibition in task switching: A review. Psychonomic Bulletin & Review, 17, 1–14. doi:https://doi.org/10.3758/PBR.17.1.1
Koch, I., Poljac, E., Müller, H., & Kiesel, A. (in press). Cognitive structure, flexibility, and plasticity in human multitasking—An integrative review of dual-task and task-switching research. Psychological Bulletin.
Kool, W., & Botvinick, M. (2014). A labor/leisure tradeoff in cognitive control. Journal of Experimental Psychology: General, 143, 131–141. doi:https://doi.org/10.1037/a0031048
Kool, W., McGuire, J. T., Rosen, Z. B., & Botvinick, M. M. (2010). Decision making and the avoidance of cognitive demand. Journal of Experimental Psychology: General, 139, 665–682. doi:https://doi.org/10.1037/a0020198
Kray, J., & Lindenberger, U. (2000). Adult age differences in task switching. Psychology and Aging, 15, 126–147.
Kushleyeva, Y., Salvucci, D. D., & Lee, F. J. (2005). Deciding when to switch tasks in time-critical multitasking. Cognitive Systems Research, 6, 41–49.
Lawo, V., Philipp, A. M., Schuch, S., & Koch, I. (2012). The role of task preparation and task inhibition in age-related task-switching deficits. Psychology and Aging, 27, 1130–1137.
Leonhard, T., Ruiz Fernández, S., Ulrich, R., & Miller, J. (2011). Dual-task processing when task 1 is hard and task 2 is easy: Reversed central processing order? Journal of Experimental Psychology: Human Perception and Performance, 37, 115–136.
Liefooghe, B., Demanet, J., & Vandierendonck, A. (2009). Is advance reconfiguration in voluntary task switching affected by the design employed? Quarterly Journal of Experimental Psychology, 62, 850–857.
Lien, M.-C., & Ruthruff, E. (2008). Inhibition of task set: Converging evidence from task choice in the voluntary task-switching paradigm. Psychonomic Bulletin & Review, 15, 1111–1116. doi:https://doi.org/10.3758/PBR.15.6.1111
Masson, M. E. J., & Carruthers, S. (2014). Control processes in voluntary and explicitly cued task switching. Quarterly Journal of Experimental Psychology, 67, 1944–1958.
Mayr, U. (2006). What matters in the cued task-switching paradigm: Tasks or cues? Psychonomic Bulletin & Review, 13, 794–799. doi:https://doi.org/10.3758/BF03193999
Mayr, U., & Bell, T. (2006). On how to be unpredictable evidence from the voluntary task-switching paradigm. Psychological Science, 17, 774–780.
Mayr, U., & Bryck, R. L. (2007). Outsourcing control to the environment: Effects of stimulus/response locations on task selection. Psychological Research, 71, 107–116.
Mayr, U., & Keele, S. W. (2000). Changing internal constraints on action: The role of backward inhibition. Journal of Experimental Psychology: General, 129, 4–26. doi:https://doi.org/10.1037/0096-3445.129.1.4
Mayr, U., Kuhns, D., & Rieter, M. (2013). Eye movements reveal dynamics of task control. Journal of Experimental Psychology: General, 142, 489–502.
Meiran, N. (1996). Reconfiguration of processing mode prior to task performance. Journal of Experimental Psychology: Learning, Memory, and Cognition, 22, 1423–1442. doi:https://doi.org/10.1037/0278-7393.22.6.1423
Meiran, N., Chorev, Z., & Sapir, A. (2000). Component processes in task switching. Cognitive Psychology, 41, 211–253.
Meiran, N., & Kessler, Y. (2008). The task rule congruency effect in task switching reflects activated long-term memory. Journal of Experimental Psychology: Human Perception and Performance, 34, 137–157. doi:https://doi.org/10.1037/0096-1523.34.1.137
Miller, J., Ulrich, R., & Rolke, B. (2009). On the optimality of serial and parallel processing in the psychological refractory period paradigm: Effects of the distribution of stimulus onset asynchronies. Cognitive Psychology, 58, 273–310. doi:https://doi.org/10.1016/j.cogpsych.2006.08.003
Mittelstädt, V., Dignath, D., Schmidt-Ott, M., & Kiesel, A. (2018). Exploring the repetition bias in voluntary task switching. Psychological Research, 82, 78–91. doi:https://doi.org/10.1007/s00426-017-0911-5
Mittelstädt, V., & Miller, J. (2017). Separating limits on preparation versus online processing in multitasking paradigms: Evidence for resource models. Journal of Experimental Psychology: Human Perception and Performance, 43, 89–102.
Monsell, S. (2003). Task switching. Trends in Cognitive Sciences, 7, 134–140. doi:https://doi.org/10.1016/S1364-6613(03)00028-7
Monsell, S., & Mizon, G. A. (2006). Can the task-cuing paradigm measure an endogenous task-set reconfiguration process? Journal of Experimental Psychology: Human Perception and Performance, 32, 493–516.
Monsell, S., Yeung, N., & Azuma, R. (2000). Reconfiguration of task-set: Is it easier to switch to the weaker task? Psychological Research, 63, 250–264. doi:https://doi.org/10.1007/s004269900005
Navon, D., & Miller, J. (2002). Queuing or sharing? A critical evaluation of the single-bottleneck notion. Cognitive Psychology, 44, 193–251.
Nickerson, R. S. (2002). The production and perception of randomness. Psychological Review, 109, 330–357. doi:https://doi.org/10.1037/0033-295X.109.2.330
Orr, J. M., & Weissman, D. H. (2011). Succumbing to bottom-up biases on task choice predicts increased switch costs in the voluntary task switching paradigm. Frontiers in Psychology, 2, 31. doi:https://doi.org/10.3389/fpsyg.2011.00031
Pashler, H. (1984). Processing stages in overlapping tasks: Evidence for a central bottleneck. Journal of Experimental Psychology: Human Perception and Performance, 10, 358–377. doi:https://doi.org/10.1037/0096-1523.10.3.358
Pashler, H. (2000). Task switching and multitask performance. In S. Monsell & J. Driver (Eds.), Control of cognitive processes: Attention and performance XVIII (pp. 277–309). Cambridge, MA: MIT Press.
Payne, S. J., Duggan, G. B., & Neth, H. (2007). Discretionary task interleaving: Heuristics for time allocation in cognitive foraging. Journal of Experimental Psychology: General, 136, 370–388.
Plessow, F., Kiesel, A., & Kirschbaum, C. (2012). The stressed prefrontal cortex and goal-directed behaviour: Acute psychosocial stress impairs the flexible implementation of task goals. Experimental Brain Research, 216, 397–408.
Plessow, F., Kiesel, A., Petzold, A., & Kirschbaum, C. (2011). Chronic sleep curtailment impairs the flexible implementation of task goals in new parents. Journal of Sleep Research, 20, 279–287.
Poljac, E., Simon, S., Ringlever, L., Kalcik, D., Groen, W. B., Buitelaar, J. K., & Bekkering, H. (2010). Impaired task switching performance in children with dyslexia but not in children with autism. Quarterly Journal of Experimental Psychology, 63, 401–416.
Poljac, E., & Yeung, N. (2012). Dissociable neural correlates of intention and action preparation in voluntary task switching. Cerebral Cortex, 24, 465–478.
Questienne, L., Atas, A., Burle, B., & Gevers, W. (2018). Objectifying the subjective: Building blocks of metacognitive experiences in conflict tasks. Journal of Experimental Psychology: General, 147, 125–131.
Questienne, L., van Dijck, J.-P., & Gevers, W. (2018). Introspection of subjective feelings is sensitive and specific. Journal of Experimental Psychology: Human Perception and Performance, 44, 215–225. doi:https://doi.org/10.1037/xhp0000437
Rapoport, A., & Budescu, D. V. (1997). Randomization in individual choice behavior. Psychological Review, 104, 603–617. doi:https://doi.org/10.1037/0033-295X.104.3.603
Redick, T. S., Shipstead, Z., Meier, M. E., Montroy, J. J., Hicks, K. L., Unsworth, N., … Engle, R. W. (2016). Cognitive predictors of a common multitasking ability: Contributions from working memory, attention control, and fluid intelligence. Journal of Experimental Psychology: General, 145, 1473–1492.
Reuss, H., Kiesel, A., Kunde, W., & Hommel, B. (2011). Unconscious activation of task sets. Consciousness and Cognition, 20, 556–567.
Rogers, R. D., & Monsell, S. (1995). Costs of a predictible switch between simple cognitive tasks. Journal of Experimental Psychology: General, 124, 207–231. doi:https://doi.org/10.1037/0096-3445.124.2.207
Rubinstein, J. S., Meyer, D. E., & Evans, J. E. (2001). Executive control of cognitive processes in task switching. Journal of Experimental Psychology: Human Perception and Performance, 27, 763–797.
Salvucci, D. D., & Taatgen, N. A. (2010). The multitasking mind. New York, NY: Oxford University Press.
Schneider, D. W. (2015a). Attentional control of response selection in task switching. Journal of Experimental Psychology: Human Perception and Performance, 41, 1315–1324.
Schneider, D. W. (2015b). Isolating a mediated route for response congruency effects in task switching. Journal of Experimental Psychology: Learning, Memory, and Cognition, 41, 235–245. doi:https://doi.org/10.1037/xlm0000049
Schneider, D. W. (2016). Perceptual and conceptual priming of cue encoding in task switching. Journal of Experimental Psychology: Learning, Memory, and Cognition, 42, 1112–1126.
Schneider, D. W. (2017). Categorization difficulty modulates the mediated route for response selection in task switching. Psychonomic Bulletin & Review. Advance online publication. doi:https://doi.org/10.3758/s13423-017-1416-3
Schneider, D. W., & Anderson, J. R. (2010). Asymmetric switch costs as sequential difficulty effects. Quarterly Journal of Experimental Psychology, 63, 1873–1894.
Schneider, D. W., & Logan, G. D. (2006a). Hierarchical control of cognitive processes: Switching tasks in sequences. Journal of Experimental Psychology: General, 135, 623–640.
Schneider, D. W., & Logan, G. D. (2006b). Priming cue encoding by manipulating transition frequency in explicitly cued task switching. Psychonomic Bulletin & Review, 13, 145–151. doi:https://doi.org/10.3758/BF03193826
Schuch, S., & Koch, I. (2003). The role of response selection for inhibition of task sets in task shifting. Journal of Experimental Psychology: Human Perception and Performance, 29, 92–105. doi:https://doi.org/10.1037/0096-1523.29.1.92
Sohn, M.-H., & Anderson, J. R. (2001). Task preparation and task repetition: Two-component model of task switching. Journal of Experimental Psychology: General, 130, 764–7728.
Stevenson, L. M., & Carlson, R. A. (2003). Information acquisition strategies and the cognitive structure of arithmetic. Memory & Cognition, 31, 1249–1259.
Strayer, D. L., & Drews, F. A. (2007). Cell-phone–induced driver distraction. Current Directions in Psychological Science, 16, 128–131. doi:https://doi.org/10.1111/j.1467-8721.2007.00489.x
Teuchies, M., Demanet, J., Sidarus, N., Haggard, P., Stevens, M. A., & Brass, M. (2016). Influences of unconscious priming on voluntary actions: Role of the rostral cingulate zone. NeuroImage, 135, 243–252.
Tombu, M., & Jolicœur, P. (2003). A central capacity sharing model of dual-task performance. Journal of Experimental Psychology: Human Perception and Performance, 29, 3–18. doi:https://doi.org/10.1037/0096-1523.29.1.3
Umemoto, A., & Holroyd, C. B. (2016). Exploring individual differences in task switching: Persistence and other personality traits related to anterior cingulate cortex function. Progress in Brain Research, 229, 189–212. doi:https://doi.org/10.1016/bs.pbr.2016.06.003
Vandamme, K., Szmalec, A., Liefooghe, B., & Vandierendonck, A. (2010). Are voluntary switches corrected repetitions? Psychophysiology, 47, 1176–1181.
Vandierendonck, A., Demanet, J., Liefooghe, B., & Verbruggen, F. (2012). A chain-retrieval model for voluntary task switching. Cognitive Psychology, 65, 241–283.
Vandierendonck, A., Liefooghe, B., & Verbruggen, F. (2010). Task switching: Interplay of reconfiguration and interference control. Psychological Bulletin, 136, 601–626. doi:https://doi.org/10.1037/a0019791
Waszak, F., Hommel, B., & Allport, A. (2003). Task-switching and long-term priming: Role of episodic stimulus–task bindings in task-shift costs. Cognitive Psychology, 46, 361–413. doi:https://doi.org/10.1016/S0010-0285(02)00520-0
Welford, A. T. (1952). The “psychological refractory period” and the timing of high-speed performance—A review and a theory. British Journal of Psychology, 43, 2–19.
Wisniewski, D., Reverberi, C., Tusche, A., & Haynes, J. D. (2015). The neural representation of voluntary task-set selection in dynamic environments. Cerebral Cortex, 25, 4715–4726.
Yeung, N. (2010). Bottom-up influences on voluntary task switching: The elusive homunculus escapes. Journal of Experimental Psychology: Learning, Memory, and Cognition, 36, 348–362.
Yeung, N., & Monsell, S. (2003). Switching between tasks of unequal familiarity: The role of stimulus-attribute and response-set selection. Journal of Experimental Psychology: Human Perception and Performance, 29, 455–469. doi:https://doi.org/10.1037/0096-1523.29.2.455
Author note
This research was supported by a grant within the Priority Program, SPP 1772, from the German Research Foundation, Grant No. KI1388/8-1. J.M. was further supported by a grant from the Alexander von Humboldt Foundation. We thank Jonathan Bercher for his assistance with data collection. The raw data are available via the Open Science Framework, at https://osf.io/5kbmg/.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mittelstädt, V., Miller, J. & Kiesel, A. Trading off switch costs and stimulus availability benefits: An investigation of voluntary task-switching behavior in a predictable dynamic multitasking environment. Mem Cogn 46, 699–715 (2018). https://doi.org/10.3758/s13421-018-0802-z
Published:
Issue Date:
DOI: https://doi.org/10.3758/s13421-018-0802-z