Abstract
Outcome monitoring is crucial for subsequent adjustments in behavior and is associated with a specific electrophysiological response, the feedback-related negativity (FRN). Besides feedback generated by one’s own action, the performance of others may also be relevant for oneself, and the observation of outcomes for others’ actions elicits an observer FRN (oFRN). To test how these components are influenced by social setting and predictive value of feedback information, we compared event-related potentials, as well as their topographies and neural generators, for performance feedback generated by oneself and others in a cooperative versus competitive context. Our results show that (1) the predictive relevance of outcomes is crucial to elicit an FRN in both players and observers, (2) cooperation increases FRN and P300 amplitudes, especially in individuals with high traits of perspective taking, and (3) contrary to previous findings on gambling outcomes, oFRN components are generated for both cooperating and competing observers, but with smaller amplitudes in the latter. Neural source estimation revealed medial prefrontal activity for both FRN and oFRN, but with additional generators for the oFRN in the dorsolateral and ventral prefrontal cortex, as well as the temporoparietal junction. We conclude that the latter set of brain regions could mediate social influences on action monitoring by representing agency and social relevance of outcomes and are, therefore, recruited in addition to shared prediction error signals generated in medial frontal areas during action outcome observation.
Similar content being viewed by others
Introduction
Monitoring one’s own performance is fundamental for successful adjustments in behavior and for learning and guidance of future actions (Rabbitt, 1966). In the presence of other people, monitoring one’s own actions might be even more salient (Festinger, 1954): Imagine that you play tennis in a doubles game and fail to catch a ball just given over by your partner. Conversely, we also keep track of actions and their consequences for other persons (e.g., our teammates or rivals), especially when they are relevant for our own goals—for example because we cooperate or compete with them.
According to the reinforcement learning theory (Holroyd & Coles, 2002), action monitoring can be based either directly on the internal representation of the action or on external feedback information, which is usually available sometime after action execution (Gentsch, Ullsperger, & Ullsperger, 2009; Heldmann, Rüsseler, & Münte, 2008; Holroyd & Coles, 2002). Monitoring is mainly based on internal representation of one’s own or observed motor action, when the information about the correctness or goal conduciveness of a given action is available already at the time of or even before the onset of the motor response. On the other hand, monitoring has to rely on external feedback when there is no reliable internal knowledge about the correctness or incorrectness at the time of action execution (Bediou, Koban, Rosset, Pourtois, & Sander, 2012; Holroyd & Coles, 2002). When both types of information are available, later occurring external feedback might be redundant and low in predictive relevance and, thus, not elicit a prediction error signal (Heldmann et al., 2008; Holroyd & Coles, 2002).
Feedback on one’s own action signals the need for behavioral adjustment and heightened cognitive control, but it is also associated with emotional or motivational significance. Positive performance feedback is generally rewarding, whereas negative feedback elicits negative emotions (Hajcak, Moser, Holroyd, & Simons, 2006). However, the affective appraisal of feedback information might depend on situational factors like social context: In comparison with feedback given in private, receiving negative feedback in the presence of other people may enhance its impact (Radke, de Lange, Ullsperger, & de Bruijn, 2011), especially when the other person is perceived as similar and close (Festinger, 1954) or when the relationship fosters interdependence or reciprocity (Van Meel & Van Heijningen, 2010). Alternatively, external feedback might become more relevant in a competitive social context.
In the present study, we sought to clarify such influences of social context on action monitoring by investigating brain responses to feedback about self-performance versus others’ performance in cooperative, as opposed to competitive, interpersonal settings. Our paradigm allowed us to disentangle effects related to agency (self- vs other-generated actions) from effects related to two types of personal significance by manipulating the predictive relevance (i.e., the informative value or nonredundancy) of feedback information and social context separately (for actions generated either by oneself or by the other). Brain systems underlying action monitoring generate early and rapid responses to undesirable outcomes, characterized by typical event-related potentials (ERPs) at the time of action execution: Already, 0–100 ms after error commission, an error-related negativity (ERN) occurs over frontocentral electrodes, followed by an error positivity (Pe; see Falkenstein, Hohnsbein, Hoormann, & Blanke, 1991; Gehring, Goss, Coles, Meyer, & Donchin, 1993), with putative sources in medial cortical regions, especially the anterior cingulate cortex (ACC; see Herrmann, Römmler, Ehlis, Heidrich, & Fallgatter, 2004; O'Connell et al., 2007; Van Veen & Carter, 2002; Vocat, Pourtois, & Vuilleumier, 2008). Whereas the ERN reflects a generic and automatic response to errors, the Pe may be related to higher-order evaluative processing, posterror adjustments, and error awareness (Falkenstein, Hoormann, Christ, & Hohnsbein, 2000; Overbeek, Nieuwenhuis, & Ridderinkhof, 2005).
The feedback-related negativity (FRN; also referred to as medial frontal negativity, or MFN) is another negative component in ERPs, arising 200–300 ms after the presentation of negative, as compared with positive, outcome feedback. It is time-locked to feedback cues, while the ERN is time-locked to action execution. However, the FRN shows a similar topographical distribution as the ERN and is thought to have similar generators in the medial prefrontal cortex (MPFC) and ACC (Gehring & Willoughby, 2002; Gentsch et al., 2009; Miltner, Braun, & Coles, 1997). When external information is redundant (i.e., low in predictive relevance), because erroneous responses can easily be detected via internal representations (thus eliciting an ERN), negative feedback does not produce an FRN (Heldmann et al., 2008; Holroyd & Coles, 2002), suggesting that both components reflect a prediction error signal with common mechanisms (Holroyd & Coles, 2002). Despite these commonalities, ERN and FRN also differ in important features, especially in terms of the source of information (internal vs. external) that is used to compute a reward prediction error. Furthermore, while the ERN is typically followed by the Pe, the FRN is sometimes followed by a P300-like positivity, although the latter is inconstant and not fully understood. Similar to the Pe, this P300 response might also be related to higher-order outcome evaluation and affective significance of the feedback, since it is sensitive to absolute reward magnitude (Yeung & Sanfey, 2004) and expectation (Bellebaum & Daum, 2008; Hajcak, Holroyd, Moser, & Simons, 2005; Núñez Castellar, Kühn, Fias, & Notebaert, 2010; Wu & Zhou, 2009).
Interestingly, the representation of others’ actions may recruit partly similar processes as the representation of our own actions (Jeannerod, 2001; Rizzolatti, Fogassi, & Gallese, 2001; Sebanz, Knoblich, & Prinz, 2003). Thus, observing errors made by others also elicits a negative ERP, the observer ERN (oERN), whose topography and sources resemble the ERN, but with delayed latencies (Bates, Patel, & Liddle, 2005; Carp, Halenar, Quandt, Sklar, & Compton, 2009; Koban, Pourtois, Vocat, & Vuilleumier, 2010; Miltner, Brauer, Hecht, Trippe, & Coles, 2004; van Schie, Mars, Coles, & Bekkering, 2004). Likewise, an observer FRN (oFRN) can be measured during the same time window as the classical FRN (around 200–300 ms), but with smaller amplitudes for observers than for agents of actions (Fukushima & Hiraki, 2009; Leng & Zhou, 2010; Yu & Zhou, 2006), possibly reflecting a decreased saliency of other-generated errors relative to one’s own (Bellebaum, Kobza, Thiele, & Daum, 2010). Unlike the oERN (to the best of our knowledge), the neural sources of the oFRN have not been investigated yet.
Recently, a growing number of studies have examined how action-monitoring systems are influenced by social factors. For instance, brain responses to observed errors (oERN) are modulated by the relationship between agent and observer, with larger effects when perceived similarity is high (Carp et al., 2009), and during cooperation, as compared with competition (Koban et al., 2010). Besides this research on internal error monitoring, a few other studies also investigated the influence of social relationship on processing external feedback information about outcomes (Mobbs et al., 2009). However, an important limitation of these studies is that they focused either on self-generated actions only or on the observation of others’ actions only (but see de Bruijn, de Lange, von Cramon, & Ullsperger, 2009). For example, a recent study (Rigoni, Polezzi, Rumiati, Guarino, & Sartori, 2009) reported a reduced amplitude of the FRN/MFN in social (comparative and competitive) versus individual conditions during a gambling task, but because the outcomes for self and other were presented at the same time, both types of information could not be disentangled. Another recent study (Van Meel & Van Heijningen, 2010) found an FRN for negative, as compared with positive, feedback during a learning task only in a competitive setting, not in an individual condition, indicating that the mere presence of other people may enhance the processing of (external) feedback, perhaps due to social comparison effects (Boksem, Ruys, & Aarts, 2011). Accordingly, fMRI results indicate that the dorsomedial prefrontal cortex, a region typically recruited by mentalizing tasks (Kelley et al., 2002; Mitchell, Banaji, & Macrae, 2005), shows higher activation during error monitoring in social (cooperative or competitive) context, relative to individual settings (Radke et al., 2011).
Results concerning social effects on ERPs to others’ feedback are also mixed: In a gambling task, the oFRN/oMFN was larger when participants thought that they were observing other humans rather than a computer (Fukushima & Hiraki, 2009). The oFRN was also larger for friends than for strangers in one study (Kang, Hirsh, & Chasteen, 2010), but not in another (Leng & Zhou, 2010). Interestingly, cooperative versus antagonistic relationships between the participant and another virtual player during a gambling task produced no effects on ERPs to own losses, but the oFRN/oMFN was amplified for losses of cooperating partners and gains of antagonistic partners, indicating an evaluation of outcomes based on self-interest (Itagaki & Katayama, 2008; Marco-Pallares, Kramer, Strehl, Schroder, & Munte, 2010). However, gambling outcomes are directly related not to performance accuracy but, rather, to probabilistic factors. Given previous evidence that observation effects depend on perceived agency of the observed player, as well as interpersonal relationships (Fukushima & Hiraki, 2006; Kang et al., 2010), it is critical to investigate the effects of cooperative and competitive context on performance monitoring in real two-participant settings. In an elegant fMRI study, de Bruijn et al. (2009) showed that feedback indicating erroneous responses activated the ACC irrespective of social context. However, they found that a reward-dependent activity in striatal areas was modulated by cooperation versus competition, indicating a possible dissociation between observed error and reward processing.
Here, we investigate—in contrast to previous studies—the time course of brain responses to feedback in both the agent and observer of actions, to determine whether similar effects of social context are observed for positive and negative outcomes based on performance (in contrast to gambling, which is less relevant for self-monitoring) and to identify the possible neural circuits underlying these modulations of scalp ERPs. We measured the feedback-locked ERPs to positive versus negative outcomes during a speeded go/no-go task and compared pairs of real participants in either cooperative or competitive conditions. Results for the response-locked ERPs following own and observed commission errors (ERN and oERN, respectively) of the same data set, using this go/no task, were reported elsewhere (Koban et al., 2010) and revealed earlier and enhanced error-related components in cooperating versus competing observers. Contrary to error observation, the processing of own errors (as indicated by ERN and Pe) was not influenced by social context, and we reasoned that this might reflect the generic nature of early brain responses to commission errors (based on internal signals). However, the feedback-related ERPs provide a different window into action monitoring, and feedback processing relies on the integration of different (external) information, where social and motivational influences can be more easily observed. In the present study, we reanalyzed the same experimental data set, including the same participants, but we focused on different trial types (fast and slow hits on go trials), completely different time windows (corresponding to brain activity elicited by the visual feedback given 1 s after the response), and different components (FRN and feedback P300 instead of ERN and Pe), unlike our previous report (Koban et al., 2010).
In our go/no-go task, positive feedback was given following hits made within an adaptive time limit on go trials (fast hits) and after correctly withheld responses on no-go trials, whereas negative feedback was given following hits made beyond the time limit on go trials (slow hits) and following commission errors on no-go trials. We reasoned that feedback on no-go trials would have low predictive relevance (i.e., could provide only redundant information about actual outcome), since these actions (commission errors and correct withholding) could already be monitored (and generate an ERN) through internal motor representations for the player and mirroring motor representations for the observer. In contrast, feedback on go trials (fast and slow hits) was relevant throughout the experiment, since the time limit was adapted online on the basis of individual reaction times (RTs), so that actual performance outcome was difficult to anticipate for player and observer at the time of the response. Consequently, our first hypothesis was that, if both FRN and oFRN reflect a prediction error signal, they should manifest only on go , but not on no-go, trials—even though commission errors on no-go trials are more discordant with the actual task goals than are slow responses on go trials.
More critically, our second and major prediction concerned the influence of interpersonal context (cooperation vs. competition). For feedback about own actions, cooperation (but not competition) should increase the social pressure to perform well (Festinger, 1954; Van Meel & Van Heijningen, 2010) and amplify the motivational impact of negative performance (which also affects the co-player), leading to larger FRNs. On the other hand, competitive settings could make negative performance feedback more salient as well (cf. Van Meel & Van Heijningen, 2010), so the direction of this effect is open to empirical investigation. For the observation condition, if others’ performance feedback is mainly processed according to self-centered reward outcome (Hajcak et al., 2006; Holroyd & Coles, 2002), as previously suggested (Itagaki & Katayama, 2008; Marco-Pallares et al., 2010), the oFRN should be larger in cooperating observers for negative, as compared with positive, outcomes, whereas an inverse effect may be found in competing observers. However, because an oFRN has also been reported for neutral observers (Fukushima & Hiraki, 2009; Leng & Zhou, 2010; Marco-Pallares et al., 2010; Yu & Zhou, 2006) and attributed to simulation processes, a smaller oFRN might also arise during competition, where self- and other-based evaluations might interfere with each other. Indeed, recent fMRI data suggested a possible dissociation between error monitoring and reward processing in competing observers (de Bruijn et al., 2009). Hence, if the oFRN reflects a reward-based signal originating in the striatum, feedback effects should be reversed for competition. On the other hand, if the oFRN is driven by mirrored error monitoring, as observed in medial prefrontal areas with fMRI (de Bruijn et al., 2009), it should be similar in cooperation and competition.
Finally, the third goal of our study was to identify the putative neural sources of these effects using a topographical analysis (Michel, Seeck, & Landis, 1999; Pourtois, Delplanque, Michel, & Vuilleumier, 2008), combined with a distributed linear inverse solution model (Michel et al., 2004; Michel et al., 2001; Pascual-Marqui, 2002). While the FRN is attributed to generators in the MPFC and ACC (Gehring & Willoughby, 2002; Miltner et al., 1997), to our knowledge, no study has explored the neural sources of the oFRN component and their modulation by social factors. Moreover, because changes in the amplitude of ERP components at specific electrodes on the scalp may reflect either a modulation of a single generator or a recruitment of partly distinct regions, a full understanding of these electrophysiological markers and their modulations requires additional analysis beyond waveform amplitude measures (Pourtois et al., 2008).
Materials and method
Participants
Participants were described in detail in our previous article (Koban et al., 2010). In brief, 34 healthy volunteers (16 men) with normal or corrected-to-normal vision participated and constituted 17 pairs of partners, which were familiar with each other (e.g., friends or colleagues). Data from one pair of participants (2 women) had to be excluded from analyses due to excessive movement artifacts. Participants were randomly assigned to one of two social context conditions (cooperation vs. competition), as described in detail in Koban et al.: Cooperation was induced by rewarding the participants on the basis of their joint performance after every two blocks of the task (one block for each player), whereas in competition, only the better of the 2 performing players was rewarded after every two blocks. These rules were given to both participants prior to the actual EEG recordings. The proportion of male and female participants and same-sex versus opposite-sex pairs was exactly matched across both experimental groups. Familiarity and liking between the 2 players was measured with custom-designed items regarding the duration and closeness of acquaintanceship or friendship and did not differ between social context conditions (for details, see Koban et al., 2010).
Because the processing of others’ feedback (oFRN) in noncompetitive situations has previously been associated to individual traits of empathy (Fukushima & Hiraki, 2009), we also obtained questionnaire data on empathy, using the Interpersonal Reactivity Index (IRI; Davis, 1983). The IRI measures four different dimensions of empathy (perspective taking [PT], emotional compassion [EC], personal distress, fantasy scale), which were submitted to correlation analyses with feedback-monitoring activity in cooperative and competitive situations. Additionally, trait aggressiveness was assessed with the Aggressiveness Questionnaire (TAQ; Buss & Perry, 1992).
Stimuli and task
We used a modified version of a go/no-go task used in previous studies (Aarts & Pourtois, 2010; Dhar & Pourtois, 2011; Pourtois, 2011; Vocat et al., 2008) to generate a high number of errors with maintained motivation. Participants in every pair took turns in performing and observing the go/no-go task in a total of eight blocks (four blocks as player, four as observer), each comprising 60 trials (40 go trials and 20 no-go trials). Every trial started with a cue (black arrow pointing up or down, random duration of 1–2 s), which then turned green (go trial), turned cyan (no-go trial), or changed direction (no-go trial). The player had to respond as quickly as possible to go trials by a keypress. The time limit was initially set to 350 ms and then adapted online during the experimental blocks (arithmetic mean value between 350 ms and the RTs of the last correct hit within the time limit), in order to increase speed pressure and make the outcome success on go trials difficult to predict for the participants (see Aarts & Pourtois, 2010; Dhar & Pourtois, 2011; Koban et al., 2010; Pourtois, 2011; Vocat et al., 2008). Importantly, participants were not aware of this manipulation.
Feedback was presented 1,000 ms after response onset and consisted of either a green dot for a correct keypress within the time limit (fast hits) and correct no-go trials or a red dot for a keypress beyond the time limit (slow hits) and commission errors on no-go trials. Red and green dots were equiluminant. To ensure a sufficient amount of attention and involvement of the participants during the observation blocks, we instructed them to silently count the correct no-go trials of their partner (i.e., the player), as has been done for observed errors in previous studies on action observation (e.g., van Schie et al., 2004).
EEG recording and analysis
EEG was recorded using 64 electrodes from both participants in each pair, using two synchronized Biosemi Active Systems. Data were online high-pass filtered with 0.1 Hz and sampled at 2048 Hz. For offline preprocessing, the signal was filtered with 0.5-Hz high-pass and corrected for eye blink artifacts, using an algorithm as implemented in BESA software (Berg & Scherg, 1994). Response-locked ERPs on fast hits and error trials of this data set were reported in our previous article (Koban et al., 2010). We selected epochs from −500 ms before to 1,000 ms after feedback presentation for fast and slow hits (go trials), as well as after correct and incorrect no-go trials, corresponding to positive and negative performance feedback after own and observed trials. Segments were baseline corrected using the prestimulus interval (−500–0 ms). Epochs containing large nonneurophysiological artifacts were excluded before averaging. Individual average waveforms were then filtered with 1-Hz high-pass and 30-Hz low-pass filter and downsampled to 512-Hz sampling rate for calculation of ERP components and for subsequent topographical and source analyses.
The amplitude of the FRN components (observer and player FRN) was defined, following standard practice and using an unbiased a priori defined time window, as the mean voltage between 200 and 300 ms at electrodes Fz, FCz, and Cz. For the analysis of the P300, the mean voltage from 300 to 400 ms after feedback presentation was calculated at the same electrode sites along the midline. Statistical analysis on ERP components was performed using a mixed model ANOVA with experimental context (cooperation vs. competition) as a between-subjects factor and trial type (go vs. no-go trial), electrode position (Fz, FCz, and Cz), agency (player vs. observer), and feedback (positive vs. negative feedback) as within-subjects factors.
Topographical analysis
Complementary to classical ERP analysis, we performed a topographical segmentation algorithm based on K-mean spatial cluster analysis (Pascual-Marqui, Michel, & Lehmann, 1995) and implemented in Cartool software by Denis Brunet (http://brainmapping.unige.ch/Cartool.htm). These analyses also allowed us to obtain additional and unbiased information about differences in neurophysiological response between conditions, not available with standard ERP analyses, which typically focus on only one or a few preselected electrodes.
This method has been detailed elsewhere (Murray, Brunet, & Michel, 2008; Pourtois et al., 2008). In brief, the aim of microstate segmentation is to identify periods of temporal stability (and by extension, changes) in the distribution of the global electric field (i.e., topography) over the scalp surface and over successive time points, by using a formal statistical approach applied to the whole topography information, rather than to amplitude values from single electrodes. These stable topographic periods are usually referred to as “microstates” (Michel et al., 1999). Following standard practice (Murray et al., 2008; Pourtois, 2011; Pourtois et al., 2008; Pourtois, Thut, Grave de Peralta, Michel, & Vuilleumier, 2005), segmentation into microstates was first performed on the grand average ERP waveforms of slow and fast hits in both agency conditions (player and observer) and both social contexts (cooperation and competition), for the time window of 0–500 ms after feedback presentation, so as to cover all stimulus-locked (feedback-locked) components of interest. The optimal number of different topographies (maps) explaining at least 90 % of variance was selected on the basis of an objective cross-validation criterion (Pascual-Marqui et al., 1995).
To statistically assess the validity of these maps, a back-fitting procedure was then applied. The template maps identified by the clustering algorithm in the group-averaged ERPs were spatially fitted to the individual-participant ERP data to provide estimates of their representation across time and task conditions. This procedure provides fine-grained quantitative values, such as global explained variance (GEV, or goodness of fit), which is a critical index of the significance of a given topography at particular time-points, not available in a classical component analysis (Picton et al., 2000). GEV represents the sum of the explained variance weighted by the global field power at each moment in time. Because we were especially interested in the processing of negative feedback as indicated by the FRN component (Itagaki & Katayama, 2008), we assessed whether the topography of the ERP signal was reliably altered during this time interval (200–300 ms) separately as a function of agency (player vs. observer) and social context (cooperation vs. competition). GEV values obtained after fitting were then submitted to mixed model ANOVAs.
Source analyses
We used standardized low-resolution brain electromagnetic tomography (Pascual-Marqui, 2002) to estimate the configuration of neural generators underlying the dominant topographies described by the previous clustering analysis. sLORETA assumes a maximal “smoothness” of the distribution of standardized current density in order to restrict the three-dimensional inverse solutions given by the topographical potential distribution on the scalp. It uses a three-shell spherical head model that is co-registered to the MNI152 template (Mazziotta et al., 2001) in standardized stereotactic space (Talairach & Tournoux, 1988) and restricts source dipoles to 6,239 cortical gray-matter voxels with a 5-mm resolution.
We used a regularization parameter of SNR = 100 to estimate the source activity for individual ERPs (0–500 ms after feedback onset) in the four different feedback conditions on go trials: player fast hits, player slow hits, observer fast hits, and observer slow hits, for both experimental groups (i.e., social contexts). Because, to our knowledge, no prior study has investigated the neural sources of the observer FRN, as compared with the FRN, we first determined the neural generators of the FRN and the oFRN separately, irrespective of (i.e., collapsing across) the two social context conditions (cooperation and competition). We statistically assessed the differences in source activity for negative (slow hits), as compared with positive, (fast hits), feedback, using paired t-tests. Regions that showed significantly higher current source density for negative, as compared with positive, feedback (slow vs. fast hits) were submitted to a region of interest (ROI) approach in order to compare activations in the different experimental conditions in more detail (cooperation vs. competition, player vs. observer). Following the logic of an internal localizer (Friston, Rotshtein, Geng, Sterzer, & Henson, 2006), this ROI definition based on feedback correctness is orthogonal to the experimental factors that were of main interest in the subsequent analysis (social context).
Results
Behavioral results
No differences in questionnaire measures of aggressiveness or empathy were found between the experimental groups, but the perceived competitiveness was reliably influenced by experimental context in the predicted way, as found in debriefing questionnaires (see Koban, et al., 2010), suggesting that our manipulation was effective.
The number of correct fast hits was not significantly different during cooperation (M = 61.1 trials, SD = ±12.9) and competition (53.3 trials ± 15.4), t(30) = −1.56, p = .13, nor was the number of slow hits (i.e., go responses made after the correct time limit; 93.9 ± 12.3 trials in cooperation, 102.8 ± 17.8 in competition), t(30) = 1.64, p = .11, or the number of errors on no-go trials (32.2 ± 15.8 in cooperation, 28.3 ± 17.4 in competition), t(30) = 0.66, p = .51. As was expected, mean RTs were significantly shorter for fast hits (238 ± 31 ms in cooperation, 251 ± 37 ms in competition) and errors (269 ± 32 ms in cooperation, 271 ± 50 ms in competition) than for slow hits (342 ± 23 ms in cooperation, 357 ± 41 ms in competition), F(2, 60) = 184.7, p < .001, but were comparable for the two social context groups, F(1, 30) = 0.7, p = .39. There was no significant interaction between these factors [feedback type × social context, F(2, 60) = 0.8, p = .45]. The behavioral data therefore ensure a similar distribution of events and response latencies in the critical experimental conditions and no difference in strategy between conditions regarding emphasis on speed versus accuracy (cf. Koban et al., 2010).
We next looked at whether slow (negative) and fast (positive) feedback had a differential impact on behavioral adjustments, such as changes in postfeedback RTs. If participants used the performance feedback as relevant information in order to adjust behavior, they should speed up after “too slow” feedback. Therefore, we compared RT changes on trials following fast hits versus trials following slow hits (difference between RTs in trial n − trial n − 1) in the two social context conditions. A highly significant main effect of previous trial type was found, F(1, 30) = 146.1, p < .001, indicating that participants slowed down following fast hits (+52.5 ms in cooperation, +62.2 ms in competition), whereas they became faster following slow hits (−41.0 ms in cooperation, −44.1 ms in competition). Thus, people did adjust their effort and behavior to improve performance in response to feedback. However, there was no effect of social context and no significant interaction (both Fs < 1), suggesting that increased feedback components in cooperation did not lead to observable behavioral adjustment differences.
Electrophysiological results: FRN
Average ERPs time-locked to feedback presentation (red negative vs. green positive isoluminant dots) showed a conspicuous relative negativity on fronto-central electrodes that was specific to the go trials (Fig. 1a). No differences in ERP waveforms were apparent on no-go trials (see supplementary Fig. S1). An ANOVA performed on the mean amplitude of the FRN (200–300 ms) disclosed a highly significant main effect for trial type (go vs. no-go trial), F(1, 30) = 44.9, p < .001, and an interaction effect of trial type and feedback (negative vs. positive), F(1, 30) = 24.3, p < .001, but no three-way-interaction with electrode (please see supplementary Table S2 for all mean amplitudes and standard deviations).
ERPs elicited by feedback on go trials at electrode FCz (a), mean evoked potential amplitude differences (slow − fast hits) during the FRN time window (200–300 ms) at electrodes Fz, FCz, and Cz (b), and mean evoked potential amplitude differences during the P300 time window (300–400 ms) at electrodes Fz, FCz, and Cz (c). Vertical bars denote standard errors
Separate statistical analyses (ANOVAs) were carried out to confirm that no effect on FRN was observed when the feedback was weak in predictive relevance (i.e. no-go trials, in which participants could monitor their own and their partner’s performance using internal motor signals or observed motor responses; see supplementary material S6), in contrast to feedback presented after go trials (which provided participants with relevant information about their speed performance—i.e., fast vs. slow hits). Thus, when comparing commission errors and correct withholds on no-go trials, we found no effect of feedback (positive vs. negative) during the FRN time window (200–300 ms), F(1, 30) = 0.26, p = .74, and no interaction effects. By contrast, the amplitude of the FRN on go trials (fast and slow hits) was reliably modulated by experimental factors (see Fig. 1a). A mixed model ANOVA on the mean FRN amplitude (200–300 ms) with feedback type (positive/fast hits vs. negative/slow hits), electrode position (Fz, FCz, Cz), and agency (player vs. observer) as within-subjects factors, plus social context (cooperation vs. competition) as a between-subjects factor, revealed a significant main effect for agency, F(1, 30) = 15.3, p < .001, and electrode, F(2, 60) = 6.3, p = .003, as well as for feedback, F(1, 30) = 47.7, p < .001. This reflected a more negative FRN component for negative, as compared with positive, outcomes (after slow vs. fast hits, respectively) over all experimental conditions (see also Fig. 1b). More important, the interaction between agency and feedback was significant, F(1, 30) = 11.6, p = .002, indicating stronger FRN differences in players than in observers, as was also the interaction between social context and feedback, F(1, 30) = 5.8, p = .023, indicating larger differences in the cooperation, as compared with the competition, context. In addition, the interaction between electrode and feedback was also significant, F(2, 60) = 11.5, p < .001. Furthermore, there was a significant three-way interaction between feedback, electrode, and social context, F(2, 60) = 8.6, p < .001, pointing at topographical differences of the feedback-related effects between the two conditions (see below). Finally, the three-way interaction between feedback, agency, and social context approached significance, F(1, 30) = 3.2, p = .082, suggesting that the effect of social context on FRN was slightly stronger in players than in observers. To verify this, we performed Tukey tests on the FRN amplitude differences (negative–positive), collapsed across all three electrodes: This analysis revealed that only the player in the cooperation condition showed a larger FRN effect (i.e., enhanced FRN for negative, as compared with positive, feedback on go trials), relative to all other conditions—that is, player competition (p = .028), observer cooperation (p = .016), and observer competition (p = .006). None of the other pairwise comparisons was significant (all ps > .6).
Although there were only slight differences in the frequency of negative feedback across trial types, we also formally ruled out the possibility that different FRN amplitudes might reflect subtle effects of feedback likelihood (cf. Hajcak, Moser, Holroyd, & Simons, 2007) by repeating these analyses, but now including the proportion of negative feedback as a continuous covariate in our general linear model. However, this did not change the significance of all the effects reported above. To further rule out the possibility that latency differences could have biased the analyses of mean amplitude, we extracted the latency of maximal negative difference between the ERPs following negative versus positive feedback, using a generous time window of 200–350 ms to cover possible differences in FRN peak latency. A mixed-model ANOVA yielded neither significant latency differences for agency, context, or electrode position nor any interaction effect.
In sum, these ERP results confirm that participants differentially processed negative (slow hits) versus positive (fast hits) feedbacks during go trials, leading to larger FRN amplitude for the former than for the latter condition. Critically, this effect was significantly modulated by agency and social context, as demonstrated by larger amplitude differences for players than for observers and for cooperators than for competitors.
Electrophysiological results: P300
Although we had no specific prediction for later feedback-evoked components, a large positive deflection following negative feedback was apparent in the cooperation player condition, resembling a P300 (although with rather frontal maximum over FCz). This accords with a few previous studies reporting P300 effects evoked by feedback information (e.g., Hajcak et al., 2005; Yeung & Sanfey, 2004). To better characterize this effect, we performed similar analyses for the P300 time window (300–400 ms; see Fig. 1). The mixed-effects ANOVA showed only a trend for the main effect of feedback, F(1, 30) = 3.1, p = .087, but the interaction between feedback and social context was significant, F(1, 30) = 7.1, p = .012. Similarly, the interactions between feedback and agency, F(1, 30) = 6.3, p = .018, and between feedback and electrode position, F(2, 60) = 17.7, p < .001, were both significant. The ANOVA also revealed a significant three-way interaction between feedback, electrode, and social context, F(2, 60) = 5.9, p = .005, as well as between feedback, electrode, agency, F(2, 60) = 5.5, p = .006, both of which imply topographical differences in the feedback effect depending on social context and agency. Planned comparisons (based on Tukey tests) between positive and negative feedback were also performed for this P300 effect in each of the four main experimental conditions (play and observe in both social contexts). This revealed that the P300 modulation (i.e., larger amplitude for negative than for positive feedback) was actually significant only in the play cooperation condition (p = .0003), but in none of the other three conditions (all ps > .70; see also Fig. 1c).
Finally, we assessed whether these amplitude modulations of feedback-related activity (FRN and P300) might be related to interindividual variations in empathy and PT (see Fukushima & Hiraki, 2009). To this aim, we calculated Spearman rank order correlations between the mean amplitude differences for each component (slow − fast hit feedback collapsed across Fz, FCz, and Cz) and the scores from subscales of the IRI. The PT subscale correlated significantly with the player’s FRN, r = −.41, p < .05, and the P300, r = .36, p < .05, whereas the EC scale correlated positively with the observer’s oP300, r = .36, p < .05 (see Fig. 2). Notably, these correlations found for the whole sample of participants were driven mainly by participants in the cooperation condition, which showed generally higher correlations when considered alone (FRN ~ PT, r = −.51, p < .05; P300 ~ PT, r = .44, n.s.; and oP300 ~ EC, r = .54, p < .05), relative to participants in the competition condition (r = −.25, r = .27, and r = .25; all n.s.). These selective correlations further underscore the specific effects of cooperation on both the player’s and the observer’s responses. Relationship between feedback-specific components and response-/error-locked components reported in our previous article (Koban et al., 2010) can be found in the supplementary material (S5).
Scatterplots for the correlations of three different feedback-locked ERP components (ΔFRN, ΔP300, and ΔoP300) with different subscales of the Interpersonal Reactivity Index (Davis, 1983)
Topographic results
The standard mean amplitude analyses performed above revealed several significant interactions between feedback and electrode position, suggesting possible changes in the electric field configuration (i.e., topography) of the FRN as a function of condition. To gain further insight into these topographic changes, which suggest changes in neural generators rather than just amplitude modulations, we performed a standard spatiotemporal cluster analysis based on K-means (Pascual-Marqui et al., 1995; Pourtois et al., 2008), followed by source analysis. This segmentation avoids a priori selection of specific electrodes or time windows. To this aim, we used the eight grand average ERP waveforms (2 context × 2 feedback × 2 agency) during a large time interval (0–500 ms post-feedback-onset) that encompassed all ERP components of interest.
Results from this segmentation analysis disclosed a spatiotemporal solution with 19 dominant topographical mapsFootnote 1 (see Fig. 3a, b), which explained 90.7 % of variance. In agreement with the conventional ERP analysis, qualitative differences in topography were observed mainly in the time window of the FRN between 250 and 300 ms. The P300 component (map 12) was expressed mainly in the following time interval, between 300 and 400 ms.
Results of the K-means segmentation procedure. a The average global field power (GFP) per condition and sequence of the 19 distinct topography solutions identified in the grand average ERPs are shown for all experimental conditions. Same numbers denote the occurrence of the same map; order of map number is arbitary. b Duration of each map in each condition was determined by the clustering algorithm as implemented in Cartool. Topographical properties of the 19 different maps identified by the microstate segmentation (blue denote negative and red positive scalp potentials). c Results of the fitting procedure: Global explained variance (GEV) for maps 9 and 11 corresponding to the FRN topography following negative feedback during the time window 200–300 ms and for the different social and agency conditions. Vertical lines denote error bars
In the cooperation context, a distinctive topography (map 11) was uniquely present for negative feedback and similarly arose in both the player and the observer (Fig. 3a). Another distribution of the electric field (map 8) was elicited by positive feedback during the same time window; this map occurred only briefly and later (after map 11) following negative feedback.
In the competition context, the player also showed distinctive maps during the FRN time window, but with a very different configuration for negative feedback (map 9) and positive feedback (map 10). Both of these maps were again followed by map 8. Remarkably, this differentiation of feedback type was specific to the player. In the competing observer, map 9 was not specific to errors but was elicited by both positive and negative feedback. Map 10 was not observed.
To confirm the statistical significance of these different topographic segments following negative feedback in competition and cooperation conditions, the two FRN-specific maps (9 and 11) were fitted back on the ERP data of each individual participant, using a noncompetitive spatial-fitting procedure (Murray et al., 2008). This enabled us to test for potential significant differences in the GEV (corresponding to the goodness of fit) of these dominant topographies for negative performance feedback across experimental conditions. The ANOVA revealed significant interaction effects for map and context, F(1, 30) = 6.3, p = .017, map and agency, F(1, 30) = 4.4, p = .044, and map, agency, and context, F(1, 39) = 10.7, p = .003 (see Fig. 3c), confirming differential FRN topographies during competition and cooperation, as well as a modulation depending on agency (own vs. observed performance feedback).
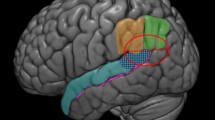
Results of the source localization, as obtained with sLORETA. Clusters with significantly higher CSD for negative versus positive performance feedback when playing were found mainly in medial prefrontal regions (a). When observing, activity in response to negative feedback emerged in the medial prefrontal cortex (MPFC), dorsal prefrontal cortex (dPFC), orbitofrontal cortex (OFC), and right superior temporal lobe/temporo-parietal junction (rSTL/TPG) (b, from top to bottom). Graphs plot the activity pattern in the different ROIs for the different experimental conditions (c). Vertical lines denote error bars
Taken together, the topography data suggest that the modulation of brain responses arising during the FRN time window as a function of feedback relevance and social context were associated with a recruitment of partly different networks, leading to a partly different distribution of neural activity over electrodes. Such differences were further explored by a distributed source localization analysis (see the next section).
Inverse solution results
We used sLORETA to estimate the possible neural generators underlying the feedback ERP topographies and their modulation by social context and agency. In a first step, we collapsed the two social context conditions (cooperation and competition) to determine sources of the FRN (player) and oFRN (observer). To this aim, we statistically compared negative (slow hits) and positive (fast hits) feedback in the inverse solution space during the time interval corresponding to the FRN (i.e., average for 250- to 300-ms interval post-stimulus-onset, during which the topography was stable).
For the player condition (FRN), this analysis showed significantly higher current source densities for negative than for positive feedbacks in a large cluster encompassing the medial frontal gyrus (MFG) and ACC (see Fig. 4a and Table 1).
For the observer condition (oFRN), neural sources were also found in the MFG and ACC, similar to the player condition, although extending slightly to more ventral areas (Fig. 4b). However, additional sources were found in several other regions, including the middle and superior frontal gyri and orbital and rectal gyri, as well as the right posterior superior temporal gyrus in the temporo-parietal junction (TPJ; see Fig. 4b, c, Table 2).
Finally, we tested how activity in these regions varied as a function of our main experimental conditions (social context and agency), by conducting an ROI analysis on the mean current activity extracted from each source. For this purpose, we created spheres (with a radius of 12 mm) around the maxima identified in the inverse solution analysis (negative vs. positive feedback), for each of four different ROIs: MPFC (x-, y-, z-coordinates 0, 45, 10), right dorsal prefrontal cortex (dPFC; 20, 60, 25), right orbitofrontal cortex (OFC; 5, 50, −20), and right posterior superior temporal gyrus (rSTL/TPJ; 35, −60, 30). We then submitted the mean source activity extracted during the 250- to 300-ms postfeedback time interval to a mixed-model ANOVA, with context as a between-subjects and agency and feedback as within-subjects factors (see Fig. 4).
An overview of significant main and interaction effects is given in Table 3. Critically, we found a main effect of agency in all regions, which showed that playing relative to observing tended to increase activity. The latter effect is likely to reflect globally higher involvement and feedback processing when acting, as compared with observing (in line with our waveform analysis; see above). More interesting, a consistent pattern of higher activity depending on agency × context (see interaction in Table 3) was found across all frontal regions, reflecting the fact that source activity was always the strongest for the cooperating player condition, in line with the hypothesis that cooperation might increase the level of feedback monitoring and cognitive control in the player (cf. ERP results). In the STG/TPJ ROI, which is an important region for social cognition and PT (Blakemore & Decety, 2001; Ruby and Decety 2004), activity was modulated not only by agency and context, but additionally by feedback valence. Accordingly, only this ROI showed a significant three-way interaction (see Table 3), with increased current source density for negative feedback in the cooperating player, but the opposite trend in the competition context.
For completeness, we also explored the possible sources of the P300 effect, which was not predicted in our initial hypotheses. We therefore probed for a significant interaction between feedback type and social context in the playing condition, using the 300- to 400-ms time interval and the contrast: (Negative Coop Play > Positive Coop Play) > (Negative Comp Play > Positive Comp Play). Significant interaction effects were found in a large cluster comprising the ACC and MPFC, plus smaller clusters in the dorsolateral prefrontal cortex, anterior insula, and inferior frontal gyri bilaterally. Additional activations were observed in the right and left precentral gyrus (see Table 4).
Discussion
The goal of our study was to characterize the interactive effects of agency, predictive relevance, and interpersonal setting on brain systems responsible for monitoring one’s own versus others’ performance. In accord with our first hypothesis, we found that feedback-related activity was influenced by the predictive value of feedback information about outcomes: The FRN effect was abolished on no-go trials, because participants could rely on internal representations to detect whether an error had been committed or not (see Koban et al., 2010). On the other hand, since the outcome of any given go trial was difficult to predict on the basis of internal monitoring mechanisms alone (due to the adaptive time limit), the performance feedback (negative vs. positive) following (slow vs. fast) go responses was highly relevant and informative throughout the task (for both participants in a pair) and, hence, significantly modulated the FRN as well as the behavioral postfeedback adjustment in RTs. Remarkably, the effect on the FRN was observed in both the player and the observer conditions. These findings not only extend previous results for error and feedback monitoring during one’s own performance (Heldmann et al., 2008; Holroyd & Coles, 2002; Stahl, 2010), but also provide the first evidence, to the best of our knowledge, that predictive relevance also influences the processing of performance feedback in the observer (oFRN), providing further support for partly shared mechanisms underlying own and observed action monitoring.
Whereas predictive relevance appears to be a necessary condition for the generation of FRN, as well as oFRN, our results also show that these prediction error signals are amplified as a function of agency and context, two factors that determine the social relevance of information. With regard to the role of agency in performance monitoring, we found a larger FRN for one’s own than for observed performance feedback. This effect corroborates previous results (Bellebaum et al., 2010; Fukushima & Hiraki, 2009; Leng & Zhou, 2010; Yu & Zhou, 2006). As was proposed by Bellebaum et al. (2010), it is likely that such agency-related enhancement results from the higher personal relevance of feedback about one’s own performance and is consistent with a greater importance of such feedback in terms of subsequent behavioral adjustment (Holroyd & Coles, 2002; Ridderinkhof, Ullsperger, Crone, & Nieuwenhuis, 2004).
Most important, classical ERP, but also topographical and source analysis, results showed a strong modulation of feedback processing as a function of social context. In the cooperative condition, the difference between responses to negative versus positive performance feedback was greater than in the competitive condition, across the two agency conditions (observer and player). This finding suggests that, in cooperative settings, one’s own performance information might be more relevant and salient because the agent is responsible for the outcome of both players. Similarly, the performance of the observed partner might be more salient during cooperation. In the following sections, we discuss these findings in detail.
Topographies and underlying neural generators of player and observer FRN
Classical ERP analyses based on waveforms at a few electrodes are not sufficient to characterize all quantitative and, especially, qualitative changes in neural responses between conditions (Michel et al., 1999; Pourtois et al., 2008). Therefore, in addition to the quantitative amplitude differences for the FRN and oFRN waveforms at frontocentral electrodes, our unbiased spatiotemporal analysis also revealed significant changes in the distribution and temporal properties of the electric field (i.e., topography) depending on both agency and social context, which could not readily be identified by waveform analysis alone. Importantly, these different topographical configurations presumably imply changes in the neural generators activated during feedback processing (Lehmann & Skrandies, 1980; Michel et al., 2001). Source reconstructions of the FRN and oFRN demonstrated that the neural sources of the player’s FRN were located mainly in medial prefrontal structures (MPFC), including the dorsal ACC, consistent with previous source localization results for feedback processing during various tasks (e.g., Gehring & Willoughby, 2002; Miltner et al., 1997). The MPFC and, especially, ACC are involved in cognitive control and action monitoring (Holroyd & Coles, 2002), but also in the evaluation of the motivational or affective significance of events, particularly for the more ventral parts of the ACC (Aarts & Pourtois, 2010; Bush, Luu, & Posner, 2000; Gehring & Willoughby, 2002). Our source localization results also pointed to the involvement of the MPFC in the generation of the observer’s FRN (oFRN), but in a slightly more ventral location, as compared with the MPFC cluster found for the player’s FRN. This further accords with the notion of shared neural substrates for monitoring actions performed by oneself and those performed by social partners.
Critically, however, during observation, the processing of negative feedback additionally recruited more dorsal and orbital frontal areas, as well as the right STG/TPJ. The dPFC, especially the superior frontal gyrus, is implicated in higher-order executive functions (Koechlin & Summerfield, 2007), including attention allocation, episodic retrieval, and working memory (Overwalle, 2009; Petrides, 2005). The cluster found in the superior frontal gyrus resembles activation patterns seen during introspection or mentalizing tasks (Goldberg, Harel, & Malach, 2006; Schmitz, Kawahara-Baccus, & Johnson, 2004) and moral reasoning (Greene, Nystrom, Engell, Darley, & Cohen, 2004), as well as joint attention (Williams, Waiter, Perra, Perrett, & Whiten, 2005). A recent study (Radke et al., 2011) similarly showed an increase in dorsomedial PFC activity (although left lateralized) during negative feedback processing in social, but not in nonsocial (private), task performance. Given such overlap of executive control functions with social and self-related reasoning, this region could implement a system for the “social control” of actions—that is, the allocation of attention to performance feedback as a socially relevant event. In social contexts, such evaluation could take place for both self-executed and observed actions.
By contrast, the selective involvement of the OFC may be related to the representation of emotional or motivational factors, such as the reward value of feedback and action outcome (Dhar, Wiersema, & Pourtois, 2011; Kringelbach, 2005; Peters & Büchel, 2010; Rushworth, Behrens, Rudebeck, & Walton, 2007; Wallis, 2007). The OFC is also critically involved in the social guidance of actions (Bechara, Damasio, & Damasio, 2000; Damasio, Tranel, & Damasio, 1990), including the evaluation of their social consequences and the experience of guilt in case of wrongdoing (Wagner, N'Diaye, Ethofer, & Vuilleumier, 2011). Such a role of the OFC in the social representation of action values would be consistent with its selective response to the interaction between self-agency and social cooperation.
Finally, the additional source in the right STS/TPJ might reflect PT and attribution of agency when processing the other’s feedback, since this region has an important function in mental state attribution (e.g., Decety & Grèzes, 1999; Mitchell, Macrae, & Banaji, 2006; Saxe & Kanwisher, 2003). Besides its general role in social cognition, the right TPJ is also activated by attentional reorienting in spatial cuing tasks and might implement more domain-general processes of reorienting (Mitchell, 2008), which could also contribute to social PT. Interestingly, both the anterior MPFC and TPJ are important for the inhibition of shared representations and the differentiation between self- and other-generated actions (Brass, Ruby, & Spengler, 2009; Spengler, von Cramon, & Brass, 2009), a function that might be crucial during action and feedback observation.
In sum, our results show that some neural mechanisms (especially in the MPFC) may partly be shared for processing one’s own (FRN) versus other’s (oFRN) performance feedback, in agreement with previous fMRI findings (de Bruijn et al., 2009) and with theoretical accounts suggesting a role for covert simulation during action understanding (Gallese, Keysers, & Rizzolatti, 2004; Rizzolatti et al., 2001), but are modulated by the social context of actions. On the other hand, specific mechanisms may also exist to allow an efficient differentiation between monitoring other-generated or self-generated action outcomes but rely on other regions (especially the ventral ACC and TPJ), which were found to be involved in the generation of the oFRN in our study.
Cooperation and perspective taking enhance feedback processing
In line with our third prediction, we found that feedback processing was enhanced in the cooperative, as opposed to the competitive, context. This was verified by our ERP waveform results, as well as the topographical and source localization analyses, showing differential responses to the player’s own performance feedback as a function of social context. First, the FRN amplitude was increased in cooperation, as compared with competition, and was accompanied with enhanced current sources for negative feedback in the MPFC and right TPJ in the former condition. As outlined above, these regions are crucially involved in social information processing and mental state attribution (Mitchell et al., 2006; Saxe & Kanwisher, 2003). Furthermore, we found a significant correlation between FRN amplitude and PT in the cooperation condition, which provides additional support to our hypothesis that cooperation may enhance the saliency and the affective evaluation of feedback information. The monitoring of one’s own performance might be more emotionally relevant in the cooperative condition, presumably due to the social pressure associated with the co-player. An increase in prefrontal activation was seen for both negative and positive performance feedback in the dPFC and OFC and may reflect a general social facilitation effect—that is, an increase of effort and performance in social, relative to individual, settings (Carver & Scheier, 1981; Triplett, 1898; Zajonc, 1965). As predicted by previous behavioral studies (Johnson, 1981), this effect of social monitoring should be amplified in cooperative settings, as we found here, but did not translate into behavioral performance differences. Alternatively, it is possible that the FRN might be modulated by more general motivational effects and that individuals might differ in the subjective saliency or motivational value of feedback information, but we did not obtain individual scores to assess such individual differences. Furthermore, increased saliency or relevance of the feedback stimuli could also mediate the effects of cooperative context on enhanced feedback processing in our experiment. Future research is needed to disentangle the mechanistic processes by which social factors modulate cognitive processing.
On the other hand, social effects in the ERPs were not paralleled by differences in postfeedback adjustment between the two social context conditions (for similar findings, see De Bruijn, Mars, Bekkering, & Coles, 2012). Both cooperators and competitors showed a lengthening of RTs after fast hits and a speeding-up after slow hits. However, there was a (nonsignificant) tendency for generally shorter RTs in cooperation than in competition, and average RT changes might not be sensitive enough to reflect trial-by-trial adjustments in cognitive control and effort. It would be interesting to relate single-trial electrophysiological responses following errors (ERN) or negative feedback (FRN) to subsequent behavioral adjustments, but the signal-to-noise ratio of scalp EEG might not be sufficient for such analysis. Future studies could use intracranial EEG, single-trial time frequency, or single-trial topographical analyses to address these questions. We also note that due to our block design, we could not investigate behavioral changes following error observation, which might also differ across contexts (De Bruijn et al., 2012; Núñez Castellar, Notebaert, Van den Bossche, & Fias, 2011).
Similarly striking as the FRN effects, a P300 effect (larger for negative than for positive feedback) was found to be significant in the cooperation context only, and its magnitude correlated with PT across participants. This modulation of the P300 by the valence and social context of feedback is unlikely to be caused by a simple oddball effect (Duncan-Johnson & Donchin, 1977; Squires, Squires, & Hillyard, 1975), since we collected a larger number of slow than of fast hits, leading to a slightly higher frequency of negative than of positive feedback. Hence, if this P300 effect merely reflected the detection of rare/deviant events, this component should be larger for positive than for negative feedback, which is exactly the opposite of what we have found. Instead, we surmise that this P300 effect might at least partly relate to the emotional and/or social impact of negative feedback for the player (Yeung & Sanfey, 2004), in keeping with its significant correlation with the PT scale. Indeed, in the cooperative context, being slow could affect the team performance (and payoff for both participants), such that negative feedback (for the player) might evoke negative social emotions such as guilt or shame, especially for participants who tend to take the perspective of the other person into account (for relationships between guilt and measures of empathy and perspective taking, see Tangney, 1991). The sources of this component in the dACC and bilateral anterior insula, as well as other prefrontal regions, accord well with previous results for error- and feedback-related activations in fMRI (for reviews, see Klein et al., 2007; Taylor, Stern, & Gehring, 2007). Especially, the anterior insula has been related to error awareness (Klein et al., 2007; Ullsperger, Harsay, Wessel, & Ridderinkhof, 2010), but given its role in emotion processing and interoception (Critchley, Wiens, Rotshtein, Ohman, & Dolan, 2004; Singer, Critchley, & Preuschoff, 2009), it might also be involved in the affective evaluation of negative performance outcomes (Brass & Haggard, 2010). However, since we had no a priori prediction about P300, our interpretation of this effect remains tentative. Further ERP research is needed to corroborate these conclusions and verify whether variations of the P300 and activations in the anterior insula might index the subjective experience of social and self-evaluative emotions when processing negative feedback in (cooperative) social context during other experimental paradigms.
Importantly, we note that social context effects were not observed during the response-locked action monitoring in the player (Koban et al., 2010), since neither ERN nor Pe amplitudes were significantly different between cooperation and competition. This points to a dissociation between error and feedback monitoring, which are based on different types of information (internal motor representation vs. external visual feedback). The input from different sensory or motor areas may lead to similar prediction error signals, but subsequent evaluation processes might integrate additional signals from social or motivational context.
Our fourth and last prediction concerned the effect of social context on the observer when processing feedback information about the other person’s performance. On the basis of previous findings (Itagaki & Katayama, 2008; Marco-Pallares et al., 2010), we expected opposite FRN effects for cooperation versus competition—that is, a classic oFRN to negative feedback during cooperation but a greater response to positive feedback during competition. However, our results did not fulfill this prediction. The oFRN amplitude was enhanced when observing negative versus positive outcomes in both the cooperators and competitors (although this effect was smaller in the latter). Furthermore, neural activity extracted from source ROIs appeared very similar in both feedback conditions for the observers, despite changes in topography across social contexts. These findings therefore suggest that the oFRN was primarily sensitive to negative outcomes, irrespective of the social relationships between partners, or to feedback cues as an action error signal, rather than a reward error signal (de Bruijn et al., 2009). However, given changes in topography, we cannot entirely rule out the possibility that neural activity partly differed between these two social contexts in brain regions that were not included in our ROI analysis.
This divergent finding for oFRN across studies is likely explained by differences in the task and experimental settings. In particular, it seems to accord with recent hypotheses (Fukushima & Hiraki, 2006; Marco-Pallares et al., 2010) suggesting that two different processes might lead to the generation of an oFRN and influence the direction of its amplitude changes: a first mechanism based on the evaluation of outcome for oneself, and a second simulation or mirroring mechanism based on the observed action outcome. Some evidence for two separate mechanisms involved in action outcome observation comes from the fMRI study by de Bruijn et al. (2009), who showed dissociable reward- versus error-related signals. Striatal reward versus medial prefrontal error detection mechanisms could be differentially recruited depending on task (e.g., gambling vs. performance-based feedback) and social context (e.g., computer vs. real human opponent, familiarity, and spatial proximity of participants, etc.). For instance, in the study by Itagaki and Katayama (2008), participants played against a virtual partner, whereas here we invited 2 participants who had to sit next to each other during the whole experiment, a procedure that should enhance the attribution of agency and simulation processes. In another study (Marco-Pallares et al., 2010), participants who did not know each other were assigned to a fixed role of being either the performer or the observer, whereas our participants switched between these two roles several times, which might also promote PT or simulation processes. Taken together, these differences might account for a stronger contribution of action simulation but a weaker contribution of self- and reward-centered appraisal to the oFRN.
Another important consideration that could account for divergent findings between studies concerns a crucial difference between gambling outcomes and performance feedback (as obtained during our go/no-go task, for example). Here, feedback was based on a continuous adjustment of criteria directly reflecting individual performance, including moment-to-moment fluctuations, such that it really provided relevant information about optimal/desired response speed. In the case of random gains or losses (gambling; see, e.g., Itagaki & Katayama 2008), the outcome is not really controllable by the participant, even if it depends on a self-generated action (e.g., choice between two boxes). In contrast, performance feedback provided during simple RT tasks not only may reflect positive or negative outcomes (like losing or winning point), but additionally may incorporate information about actual performance and goals and, thus, have implications for motivating behavioral adjustments (as evidenced by the trial-by-trial changes in RTs). Therefore, it is likely that the monitoring of gambling versus performance outcome may at least partly diverge (Elliott, Frith, & Dolan, 1997), especially in their relative recruitment of action- and reward-monitoring systems (de Bruijn, et al., 2009). It would be interesting to test more directly in future work whether ERP correlates of performance monitoring (FRN and oFRN) may differ between these two different task settings (gambling vs. performance outcome).
Finally, we note that our crossed factorial design comparing competition versus cooperation did not include a neutral control condition—for example, with a noninteractive context. Although this might potentially make it difficult to attribute some effects to competition or cooperation specifically, our primary aim was to compare these two conditions as opposite points along the same continuum of outcome-monitoring processes reflected by the FRN and oFRN. Moreover, whereas a control condition with a single player performing alone would be easy to implement, a noninteractive observer condition might be harder to construct without confound in attention and motivation. Nevertheless, future research should further explore neural processes that might be uniquely or commonly engaged in these different social contexts, relative to nonsocial situations.
Conclusions
The aim of our study was to investigate the influence of personal relevance (based on internal prediction and agency), as well as social relevance, on the neural response to feedback about actions produced by oneself or others. We show that the generation of both FRN and oFRN depends on whether the feedback is relevant—that is, contains new information about the adequacy of the just performed action. In addition, the FRN amplitude is larger than the oFRN, due to the increased personal relevance of own feedback and/or to qualitative changes in the configuration of the underlying neural generators (FRN vs. oFRN), as suggested by topographical analyses and source estimations. Furthermore, contrary to the ERN and Pe, the FRN and P300 amplitudes to own feedback and its topography are modulated by social context, with larger responses for cooperators than for competitors. Negative feedback leads to speeding-up in following trials, which confirms that this external information was indeed relevant to adjust behavior and effort. Although the adjustment in RTs was similar across contexts, we suggest that the negative feedback has an increased motivational impact or social relevance for players during cooperation, because negative outcomes may decrease the team’s performance and evoke negative self-conscious emotions such as guilt or shame. Consistent with this view, we found that this social effect is more pronounced for individuals with higher PT abilities. By contrast, observers showed an oFRN to negative, as compared with positive, feedback that was similar across both social contexts, which might reflect a predominant recruitment of simulation processes, rather than self-centered evaluations regarding the reward value of action outcomes for the observer. Taken together, these results highlight the important influences of social and motivational factors on the monitoring of behavior, for both self-generated and other-generated actions, and provide new insights into their underlying neural substrates.
Notes
Note that numbering of maps results from the clustering and the relative time of their appearance but does not depend on topographic characteristics and is, therefore, arbitrary.
References
Aarts, K., & Pourtois, G. (2010). Anxiety not only increases, but also alters early error-monitoring functions. Cognitive, Affective, & Behavioral Neuroscience, 10, 479–492.
Bates, A. T., Patel, T. P., & Liddle, P. F. (2005). External behavior monitoring mirrors internal behavior monitoring: Error-related negativity for observed errors. Journal of Psychophysiology, 19, 281–288.
Blakemore, S. J., & Decety, J. (2001). From the perception of action to the understanding of intention. Nature Reviews. Neuroscience, 2(8), 561–567.
Bechara, A., Damasio, H., & Damasio, A. R. (2000). Emotion, decision making and the orbitofrontal cortex. Cerebral Cortex, 10, 295–307.
Bediou, B., Koban, L., Rosset, S. B., Pourtois, G., & Sander, D. (2012). Delayed monitoring of accuracy errors compared to commission errors in ACC. NeuroImage, 60, 1925–1936.
Bellebaum, C., & Daum, I. (2008). Learning-related changes in reward expectancy are reflected in the feedback-related negativity. European Journal of Neuroscience, 27, 1823–1835.
Bellebaum, C., Kobza, S., Thiele, S., & Daum, I. (2010). It was not MY fault: Event-related brain potentials in active and observational learning from feedback. Cerebral Cortex, 20, 2874–2883.
Berg, P., & Scherg, M. (1994). A multiple source approach to the correction of eye artifacts. Electroencephalography and Clinical Neurophysiology, 90, 229–241.
Boksem, M. A. S., Ruys, K. I., & Aarts, H. (2011). Facing disapproval: Performance monitoring in a social context. Social Neuroscience, 6, 360–368.
Brass, M., & Haggard, P. (2010). The hidden side of intentional action: The role of the anterior insular cortex. Brain Structure & Function, 214, 603–610.
Brass, M., Ruby, P., & Spengler, S. (2009). Inhibition of imitative behaviour and social cognition. Philosophical Transactions of the Royal Society B, 364, 2359–2367.
Bush, G., Luu, P., & Posner, M. I. (2000). Cognitive and emotional influences in anterior cingulate cortex. Trends in Cognitive Sciences, 4, 215–222.
Buss, A. H., & Perry, M. (1992). The aggression questionnaire. Journal of Personality and Social Psychology, 63, 452–459.
Carp, J., Halenar, M. J., Quandt, L. C., Sklar, A., & Compton, R. J. (2009). Perceived similarity and neural mirroring: Evidence from vicarious error processing. Social Neuroscience, 4, 85–96.
Carver, C. S., & Scheier, M. F. (1981). The self-attention-induced feedback loop and social facilitation. Journal of Experimental Social Psychology, 17, 545–568.
Critchley, H. D., Wiens, S., Rotshtein, P., Ohman, A., & Dolan, R. J. (2004). Neural systems supporting interoceptive awareness. Nature Neuroscience, 7, 189–195.
Damasio, A. R., Tranel, D., & Damasio, H. (1990). Individuals with sociopathic behavior caused by frontal damage fail to respond autonomically to social stimuli. Behavioural Brain Research, 41, 81–94.
Davis, M. H. (1983). Measuring individual differences in empathy: Evidence for a multidimensional approach. Journal of Personality and Social Psychology, 44, 113–126.
de Bruijn, E. R. A., de Lange, F. P., von Cramon, D. Y., & Ullsperger, M. (2009). When errors are rewarding. Journal of Neuroscience, 29, 12183–12186.
de Bruijn, E. R. A., Mars, R. B., Bekkering, H., & Coles, M. G. H. (2012). Your mistake is my mistake. .. or is it? Behavioural adjustments following own and observed actions in cooperative and competitive contexts. Quarterly Journal of Experimental Psychology, 65, 317–325.
Decety, J., & Grèzes, J. (1999). Neural mechanisms subserving the perception of human actions. Trends in Cognitive Sciences, 3, 172–178.
Dhar, M., & Pourtois, G. (2011). Early error detection is generic, but subsequent adaption to errors is not: Evidence from ERPs. Neuropsychologia, 49, 1236–1245.
Dhar, M., Wiersema, J. R., & Pourtois, G. (2011). Cascade of neural events leading from error commission to subsequent awareness revealed using EEG source imaging. PLoS One, 6, e19578–e19578.
Duncan-Johnson, C. C., & Donchin, E. (1977). On quantifying surprise: The variation of event-related potentials with subjective probability. Psychophysiology, 14, 456–467.
Elliott, R., Frith, C. D., & Dolan, R. J. (1997). Differential neural response to positive and negative feedback in planning and guessing tasks. Neuropsychologia, 35, 1395–1404.
Falkenstein, M., Hohnsbein, J., Hoormann, J., & Blanke, L. (1991). Effects of crossmodal divided attention on ERP components: Error processing in choice reaction tasks. Electroencephalography and Clinical Neurophysiology, 78, 447–455.
Falkenstein, M., Hoormann, J., Christ, S., & Hohnsbein, J. (2000). ERP components on reaction errors and their functional significance: A tutorial. Biological Psychology, 51, 87–107.
Festinger, L. (1954). A theory of social comparison processes. Human Relations, 7, 117–140.
Friston, K. J., Rotshtein, P., Geng, J. J., Sterzer, P., & Henson, R. N. (2006). A critique of functional localisers. NeuroImage, 30, 1077–1087.
Fukushima, H., & Hiraki, K. (2006). Perceiving an opponent's loss: Gender-related differences in the medial-frontal negativity. Social Cognitive and Affective Neuroscience, 1, 149–157.
Fukushima, H., & Hiraki, K. (2009). Whose loss is it? Human electrophysiological correlates of non-self reward processing. Social Neuroscience, 4, 261–275.
Gallese, V., Keysers, C., & Rizzolatti, G. (2004). A unifying view of the basis of social cognition. Trends in Cognitive Sciences, 8, 396–403.
Gehring, W. J., Goss, B., Coles, M. G. H., Meyer, D. E., & Donchin, E. (1993). A neural system for error-detection and compensation. Psychological Science, 4, 385–390.
Gehring, W. J., & Willoughby, A. R. (2002). The medial frontal cortex and the rapid processing of monetary gains and losses. Science, 295, 2279–2282.
Gentsch, A., Ullsperger, P., & Ullsperger, M. (2009). Dissociable medial frontal negativities from a common monitoring system for self- and externally caused failure of goal achievement. NeuroImage, 47, 2023–2030.
Goldberg, I. I., Harel, M., & Malach, R. (2006). When the brain loses its self: Prefrontal inactivation during sensorimotor processing. Neuron, 50, 329–339.
Greene, J. D., Nystrom, L. E., Engell, A. D., Darley, J. M., & Cohen, J. D. (2004). The neural bases of cognitive conflict and control in moral judgment. Neuron, 44, 389–400.
Hajcak, G., Holroyd, C. B., Moser, J. S., & Simons, R. F. (2005). Brain potentials associated with expected and unexpected good and bad outcomes. Psychophysiology, 42, 161–170.
Hajcak, G., Moser, J. S., Holroyd, C. B., & Simons, R. F. (2006). The feedback-related negativity reflects the binary evaluation of good versus bad outcomes. Biological Psychology, 71, 148–154.
Hajcak, G., Moser, J. S., Holroyd, C. B., & Simons, R. F. (2007). It's worse than you thought: The feedback negativity and violations of reward prediction in gambling tasks. Psychophysiology, 44, 905–912.
Heldmann, M., Rüsseler, J., & Münte, T. F. (2008). Internal and external information in error processing [Abstract]. BMC Neuroscience, 9, 33.
Herrmann, M. J., Römmler, J., Ehlis, A.-C., Heidrich, A., & Fallgatter, A. J. (2004). Source localization (LORETA) of the error-related-negativity (ERN/Ne) and positivity (Pe). Cognitive Brain Research, 20, 294–299.
Holroyd, C. B., & Coles, M. G. H. (2002). The neural basis of human error processing: Reinforcement learning, dopamine, and the error-related negativity. Psychological Review, 109, 679–709.
Itagaki, S., & Katayama, J. (2008). Self-relevant criteria determine the evaluation of outcomes induced by others. NeuroReport, 19, 383–387.
Jeannerod, M. (2001). Neural simulation of action: A unifying mechanism for motor cognition. NeuroImage, 14, S103–S109.
Johnson, D. W. (1981). Effects of cooperative, competitive, and individualistic goal structures on achievement: A meta-analysis. Psychological Bulletin, 89, 47–62.
Kang, S. K., Hirsh, J. B., & Chasteen, A. L. (2010). Your mistakes are mine: Self–other overlap predicts neural response to observed errors. Journal of Experimental Social Psychology, 46, 229–232.
Kelley, W. M., Macrae, C. N., Wyland, C. L., Caglar, S., Inati, S., & Heatherton, T. F. (2002). Finding the self? An event-related fMRI study. Journal of Cognitive Neuroscience, 14, 785–794.
Klein, T. A., Endrass, T., Kathmann, N., Neumann, J., von Cramon, D. Y., & Ullsperger, M. (2007). Neural correlates of error awareness. NeuroImage, 34, 1774–1781.
Koban, L., Pourtois, G., Vocat, R., & Vuilleumier, P. (2010). When your errors make me lose or win: Event-related potentials to observed errors of cooperators and competitors. Social Neuroscience, 5, 360–374.
Koechlin, E., & Summerfield, C. (2007). An information theoretical approach to prefrontal executive function. Trends in Cognitive Sciences, 11, 229–235.
Kringelbach, M. L. (2005). The human orbitofrontal cortex: Linking reward to hedonic experience. Nature Reviews Neuroscience, 6, 691–702.
Lehmann, D., & Skrandies, W. (1980). Reference-free identification of components of checkerboard-evoked multichannel potential fields. Electroencephalography and Clinical Neurophysiology, 48, 609–621.
Leng, Y., & Zhou, X. (2010). Modulation of the brain activity in outcome evaluation by interpersonal relationship: An ERP study. Neuropsychologia, 48, 448–455.
Marco-Pallares, J., Kramer, U., Strehl, S., Schroder, A., & Munte, T. (2010). When decisions of others matter to me: An electrophysiological analysis [Abstract]. BMC Neuroscience, 11, 86.
Mazziotta, J., Toga, A., Evans, A., Fox, P., Lancaster, J., Zilles, K., . . . Mazoyer, B. (2001). A probabilistic atlas and reference system for the human brain: International Consortium for Brain Mapping (ICBM). Philosophical Transactions of the Royal Society B, 356, 1293–1322.
Michel, C. M., Murray, M. M., Lantz, G., Gonzalez, S., Spinelli, L., & Grave de Peralta, R. (2004). EEG source imaging. Clinical Neurophysiology, 115, 2195–2222.
Michel, C. M., Seeck, M., & Landis, T. (1999). Spatiotemporal dynamics of human cognition. News in Physiological Sciences, 14, 206–214.
Michel, C. M., Thut, G., Morand, S., Khateb, A., Pegna, A. J., Grave de Peralta, R., . . . Landis, T. (2001). Electric source imaging of human brain functions. Brain Research Reviews, 36, 108–118.
Miltner, W. H. R., Brauer, J., Hecht, H., Trippe, R., & Coles, M. G. H. (2004). Parallel brain activity for self-generated and observed errors. In M. Ullsperger & M. Falkenstein (Eds.), Errors, conflicts, and the brain: Current opinions on performance monitoring (pp. 124–129). Leipzig: MPI of Cognitive Neuroscience.
Miltner, W. H. R., Braun, C. H., & Coles, M. G. H. (1997). Event-related brain potentials following incorrect feedback in a time-estimation task: Evidence for a “generic” neural system for error detection. Journal of Cognitive Neuroscience, 9, 788–798.
Mitchell, J. P. (2008). Activity in right temporo-parietal junction is not selective for theory-of-mind. Cerebral Cortex, 18, 262–271.
Mitchell, J. P., Banaji, M. R., & Macrae, C. N. (2005). The link between social cognition and self-referential thought in the medial prefrontal cortex. Journal of Cognitive Neuroscience, 17, 1306–1315.
Mitchell, J. P., Macrae, C. N., & Banaji, M. R. (2006). Dissociable medial prefrontal contributions to judgments of similar and dissimilar others. Neuron, 50, 655–663.
Mobbs, D., Yu, R., Meyer, M., Passamonti, L., Seymour, B., Calder, A. J., . . . Dalgleish, T. (2009). A key role for similarity in vicarious reward. Science, 324, 900.
Murray, M. M., Brunet, D., & Michel, C. M. (2008). Topographic ERP analyses: A step-by-step tutorial review. Brain Topography, 20, 249–264.
Núñez Castellar, E., Kühn, S., Fias, W., & Notebaert, W. (2010). Outcome expectancy and not accuracy determines posterror slowing: ERP support. Cognitive, Affective, & Behavioral Neuroscience, 10, 270–278.
Núñez Castellar, E., Notebaert, W., Van den Bossche, L., & Fias, W. (2011). How monitoring other's actions influences one's own performance. Experimental Psychology, 58, 499–508.
O'Connell, R. G., Dockree, P. M., Bellgrove, M. A., Kelly, S. P., Hester, R., Garavan, H., . . . Foxe, J. J. (2007). The role of cingulate cortex in the detection of errors with and without awareness: A high-density electrical mapping study. European Journal of Neuroscience, 25, 2571–2579.
Overbeek, J. M., Nieuwenhuis, S., & Ridderinkhof, K. R. (2005). Dissociable components of error processing. Journal of Psychophysiology, 19, 319–329.
Overwalle, F. V. (2009). Social cognition and the brain: A meta-analysis. Human Brain Mapping, 30, 829–858.
Pascual-Marqui, R. D. (2002). Standardized low-resolution brain electromagnetic tomography (sLORETA): Technical details. Methods and Findings in Experimental and Clinical Pharmacology, 24(Suppl. D), 5–12.
Pascual-Marqui, R. D., Michel, C. M., & Lehmann, D. (1995). Segmentation of brain electrical activity into microstates: model estimation and validation. IEEE Transactions on Bio-Medical Engineering, 42, 658–665.
Peters, J., & Büchel, C. (2010). Neural representations of subjective reward value. Behavioural Brain Research, 213, 135–141.
Petrides, M. (2005). Lateral prefrontal cortex: Architectonic and functional organization. Philosophical Transactions of the Royal Society B, 360, 781–795.
Picton, T. W., Bentin, S., Berg, P., Donchin, E., Hillyard, S. A., Johnson, R., . . . Taylor, M. J. (2000). Guidelines for using human event-related potentials to study cognition: Recording standards and publication criteria. Psychophysiology, 37, 127–152.
Pourtois, G. (2011). Early error detection predicted by reduced pre-response control process: An ERP topographic mapping study. Brain Topography, 23, 403–422.
Pourtois, G., Delplanque, S., Michel, C., & Vuilleumier, P. (2008). Beyond conventional event-related brain potential (ERP): Exploring the time-course of visual emotion processing using topographic and principal component analyses. Brain Topography, 20, 265–277.
Pourtois, G., Thut, G., Grave de Peralta, R., Michel, C., & Vuilleumier, P. (2005). Two electrophysiological stages of spatial orienting towards fearful faces: Early temporo-parietal activation preceding gain control in extrastriate visual cortex. NeuroImage, 26, 149–163.
Rabbitt, P. M. (1966). Errors and error correction in choiceresponse tasks. Journal of Experimental Psychology, 71(2), 264–272.
Radke, S., de Lange, F. P., Ullsperger, M., & de Bruijn, E. R. A. (2011). Mistakes that affect others: An fMRI study on processing of own errors in a social context. Experimental Brain Research, 211, 405–413.
Ridderinkhof, K. R., Ullsperger, M., Crone, E. A., & Nieuwenhuis, S. (2004). The role of the medial frontal cortex in cognitive control. Science, 306, 443–447.
Rigoni, D., Polezzi, D., Rumiati, R., Guarino, R., & Sartori, G. (2009). When people matter more than money: An ERPs study. Brain Research Bulletin, 81, 445–452.
Rizzolatti, G., Fogassi, L., & Gallese, V. (2001). Neurophysiological mechanisms underlying the understanding and imitation of action. Nature Reviews Neuroscience, 2, 661–670.
Ruby, P., & Decety, J. (2004). How would you feel versus how do you think she would feel? A neuroimaging study of perspective-taking with social emotions. Journal of Cognitive Neuroscience, 16(6), 988–999.
Rushworth, M. F. S., Behrens, T. E. J., Rudebeck, P. H., & Walton, M. E. (2007). Contrasting roles for cingulate and orbitofrontal cortex in decisions and social behaviour. Trends in Cognitive Sciences, 11, 168–176.
Saxe, R., & Kanwisher, N. (2003). People thinking about thinking people: The role of the temporo-parietal junction in “theory of mind. NeuroImage, 19, 1835–1842.
Schmitz, T. W., Kawahara-Baccus, T. N., & Johnson, S. C. (2004). Metacognitive evaluation, self-relevance, and the right prefrontal cortex. NeuroImage, 22, 941–947.
Sebanz, N., Knoblich, G., & Prinz, W. (2003). Representing others' actions: Just like one's own? Cognition, 88, B11–B21.
Singer, T., Critchley, H. D., & Preuschoff, K. (2009). A common role of insula in feelings, empathy and uncertainty. Trends in Cognitive Sciences, 13, 334–340.
Spengler, S., von Cramon, D. Y., & Brass, M. (2009). Control of shared representations relies on key processes involved in mental state attribution. Human Brain Mapping, 30, 3704–3718.
Squires, N. K., Squires, K. C., & Hillyard, S. A. (1975). Two varieties of long-latency positive waves evoked by unpredictable auditory stimuli in man. Electroencephalography and Clinical Neurophysiology, 38, 387–401.
Stahl, J. (2010). Error detection and the use of internal and external error indicators: An investigation of the first-indicator hypothesis. International Journal of Psychophysiology, 77, 43–52.
Talairach, J., & Tournoux, P. (1988). Co-planar stereotaxic atlas of the human Brain. New York: Thieme Medical Publishers.
Tangney, J. P. (1991). Moral affect: The good, the bad, and the ugly. Journal of Personality and Social Psychology, 61, 598–607.
Taylor, S. F., Stern, E. R., & Gehring, W. J. (2007). Neural systems for error monitoring: Recent findings and theoretical perspectives. The Neuroscientist, 13, 160–172.
Triplett, N. (1898). The dynamogenic factors in pacemaking and competition. The American Journal of Psychology, 9, 507–533.
Ullsperger, M., Harsay, H. A., Wessel, J. R., & Ridderinkhof, K. R. (2010). Conscious perception of errors and its relation to the anterior insula. Brain Structure & Function, 214, 629–643.
Van Meel, C. S., & Van Heijningen, C. A. A. (2010). The effect of interpersonal competition on monitoring internal and external error feedback. Psychophysiology, 47, 213–222.
van Schie, H. T., Mars, R. B., Coles, M. G. H., & Bekkering, H. (2004). Modulation of activity in medial frontal and motor cortices during error observation. Nature Neuroscience, 7, 549–554.
Van Veen, V., & Carter, C. S. (2002). The timing of action-monitoring processes in the anterior cingulate cortex. Journal of Cognitive Neuroscience, 14, 593–602.
Vocat, R., Pourtois, G., & Vuilleumier, P. (2008). Unavoidable errors: A spatio-temporal analysis of time-course and neural sources of evoked potentials associated with error processing in a speeded task. Neuropsychologia, 46, 2545–2555.
Wagner, U., N'Diaye, K., Ethofer, T., & Vuilleumier, P. (2011). Guilt-specific processing in the prefrontal cortex. Cerebral Cortex, 21(11), 2461–2470.
Wallis, J. D. (2007). Orbitofrontal cortex and its contribution to decision-making. Annual Review of Neuroscience, 30, 31–56.
Williams, J. H. G., Waiter, G. D., Perra, O., Perrett, D. I., & Whiten, A. (2005). An fMRI study of joint attention experience. NeuroImage, 25, 133–140.
Wu, Y., & Zhou, X. (2009). The P300 and reward valence, magnitude, and expectancy in outcome evaluation. Brain Research, 1286, 114–122.
Yeung, N., & Sanfey, A. G. (2004). Independent coding of reward magnitude and valence in the human brain. Journal of Neuroscience, 24, 6258–6264.
Yu, R., & Zhou, X. (2006). Brain potentials associated with outcome expectation and outcome evaluation. NeuroReport, 17, 1649–1653.
Zajonc, R. B. (1965). Social facilitation. Science, 149, 269–274.
Author Note
This research was supported by a research award from the Evens Foundation and by the National Center of Competence in Research (NCCR) Affective Sciences financed by the Swiss National Science Foundation (51NF40-104897) and hosted by the University of Geneva. Cartool software has been programmed by Denis Brunet, from the Functional Brain Mapping Laboratory, Geneva, Switzerland (http://brainmapping.unige.ch/Cartool.htm) and is supported by the Center for Biomedical Imaging (CIBM) of Geneva and Lausanne. G.P. is supported by grants from the European Research Council (Starting Grant 200758) and Ghent University (BOF Grant 05Z01708).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 830 kb)
Rights and permissions
About this article
Cite this article
Koban, L., Pourtois, G., Bediou, B. et al. Effects of social context and predictive relevance on action outcome monitoring. Cogn Affect Behav Neurosci 12, 460–478 (2012). https://doi.org/10.3758/s13415-012-0091-0
Published:
Issue Date:
DOI: https://doi.org/10.3758/s13415-012-0091-0