Abstract
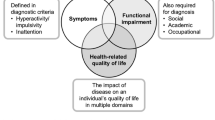
Background: Attempts to estimate the cost effectiveness of attention-deficit hyperactivity disorder (ADHD) treatments in the past have relied on classifying ADHD patients as responders or non-responders to treatment. Responder status has been associated with a small gain in health-related quality of life (HR-QOL) [or utility, as measured using the generic QOL measure EQ-5D] of 0.06 (on a scale from 0 being dead to 1.0 being full health).
Objectives: The goal of the present study was to develop and validate several ADHD-related health states, and to estimate utility values measured amongst the general public for those states and to re-estimate utility values associated with responder status.
Methods: Detailed qualitative interview data were collected from 20 young ADHD patients to characterize their HR-QOL. In addition, item-by-item clinical and HR-QOL data from a clinical trial were used to define and describe four health states (normal; borderline to mildly ill; moderately to markedly ill; and severely ill). ADHD experts assessed the content validity of the descriptions. The states were rated by 100 members of the UK general public using the time trade-off (TTO) interview and visual analog scale. Statistical mapping was also undertaken to estimate Clinical Global Impression-Improvement (CGI-I) utilities (i.e. response status) from Clinical Global Impression-Severity (CGI-S) defined states. The mapping work estimated changes in utilities from study baseline to last visit for patients with a CGI-I score of ≤2 or ≤3.
Results: The validity of the four health states developed in this study was supported by in-depth interviews with ADHD experts and patients, and clinical trial data. TTO-derived utilities for the four health states ranged from 0.839 (CGI-S state ‘normal’) to 0.444 (CGI-S state ‘severely ill’). From the mapping work, the change in utility for treatment responders was 0.19 for patients with a CGI-I score of ≤2 and 0.15 for patients with a CGI-I score of ≤3.
Conclusions: The present study provides utilities for different severity levels of ADHD estimated in a TTO study. This approach provides a more granular assessment of the impact of ADHD on HR-QOL than binary approaches employed in previous economic analyses. Change in utility for responders and non-responders at different levels of CGI-I was estimated, and thus these utilities may be used to compare health gains of different ADHD interventions.





Similar content being viewed by others
References
American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 4th ed. Washington, DC: APA, 1994
National Institute for Health and Clinical Excellence. Methylphenidate, atomoxetine and dexamfetamine for attention deficit hyperactivity disorder (ADHD) in children and adolescents. (TA98). London: NICE, 2006 Mar [online]. Available from URL: http://www.nice.org.uk/TA098 [Accessed 2009 May 6]
Pliszka SR, Carlson C, Swanson JM. ADHD with comorbid disorders: clinical assessment and management. New York: Guilford, 1999
Harpin VA. The effect of ADHD on the life of an individual, their family, and community from preschool to adult life. Arch Dis Child 2005; 90: 2–7
Biederman J, Newcom J, Sprich S. Comorbidity of attention-deficit hyperactivity disorder with conduct, depressive, anxiety and other disorders. Am J Psychiatry 1991; 148: 564–77
Barbaresi WJ, Katusic SK, Colligan RC, et al. Long-term school outcomes for children with attention-deficit/hyperactivity disorder: a population-based perspective. J Dev Behav Pediatr 2007; 28: 265–73
Kendall J, Hatton D, Beckett A, et al. Children’s accounts of attention-deficit/hyperactivity disorder. Adv Nurs Science 2003; 26: 114–30
Schatz DB, Rostain AL. ADHD with comorbid anxiety: a review of the current literature. J Atten Disord 2006; 10: 141–9
Levy F. Synaptic gating and ADHD: a biological theory of comorbidity of ADHD and anxiety. Neuropsychopharmacology 2004; 29: 1589–96
Treuting JJ, Hinshaw SP. Depression and self-esteem in boys with attention-deficit/hyperactivity disorder: associations with comorbid aggression and explanatory attribution mechanisms. J Abnorm Child Psychol 2001; 29: 23–39
Ralston SJ, Lorenzo MJM. ADORE — Attention-Deficit hyperactivity disorder Observational Research in Europe. Eur Child Adolesc Psychiatry 2004; 13 (1 Suppl.): i36–42
Nijmeijer JS, Minderaa RB, Buitelaar JK, et al. Attention-deficit/hyperactivity disorder and social dysfunctioning. Clin Psychol Rev 2008; 28: 692–708
Coghill D, Danckaerts M, Sonuga-Barke E, et al. Practitioner review: quality of life in child mental health — conceptual challenges and practical choices. J Child Psychol Psychiatry 2009; 50(5): 544–61
Klassen AF, Miller A, Fine S. Agreement between parent and child report of quality of life in children with attentiondeficit/hyperactivity disorder. Child Care Health Dev 2006; 32: 397–406
Escobar R, Soutullo CA, Hervas A, et al. Worse quality of life for children with newly diagnosed attention-deficit/hyperactivity disorder, compared with asthmatic and healthy children. Pediatrics 2005; 116(3): e364–9
Topolski TD, Edwards DC, Patrick DL, et al. Quality of life of adolescent males with attention-deficit hyperactivity disorder. J Atten Disord 2004; 7: 163–73
Buitelaar JK, Wilens TE, Zhang S, et al. Comparison of symptomatic versus functional changes in children and adolescents with ADHD during randomized, double-blind treatment with psychostimulants, atomoxetine, or placebo. J Child Psychol Psychiatry 2009; 50: 335–42
Bukstein OG, Arnold LE, Landgraf JM, et al. Does switching from oral extended-release methylphenidate to the methylphenidate transdermal system affect health-related quality-of-life and medication satisfaction for children with attention-deficit/hyperactivity disorder? Child Adolesc Psychiatry Ment Health 2009; 3(1): 39
Manos M, Frazier TW, Landgraf JM, et al. HRQL and medication satisfaction in children with ADHD treated with the methylphenidate transdermal system. Curr Med Res Opin 2009; 25(12): 3001–10
Landgraf JM, Rich M, Rappaport L. Measuring quality of life in children with attention-deficit hyperactivity disorder and their families. Arch Pediatr Adolesc Med 2002; 156:384–91
Meaux JB, Hester C, Smith B, et al. Stimulant medications: a trade-off? The lived experience of adolescents with ADHD. J Spec Pediatr Nurs 2006; 11: 214–26
National Institute for Health and Clinical Excellence. Guide to the methods of technology appraisal. London: NICE, 2008 June [online]. Available from URL: http://www.nice. org.uk/media/B52/A7/TAMethodsGuideUpdatedJune2008. pdf [Accessed 2009 May 6]
Coghill D, Spender Q, Barton J, et al. Measuring quality of life in children with attention-deficit-hyperactivity disorder in the United Kingdom. 16th World Congress of the International Association of Child and Adolescent Psychiatry and Allied Professions (IACAPAP); 2004 Aug 23–26; Berlin
Joffe H, Yardley L. Content and thematic analysis. In: Marks D, Yardley L, editors. Research methods for clinical and health psychology. London: Sage, 2004: 56–68
Arnold LE, Bozzolo DR, Hodgkins P, et al. Switching from oral extended-release methylphenidate to the methylphenidate transdermal system: continued attention-deficit/hyperactivity disorder symptom control and tolerability after abrupt conversion. Curr Med Res Opin 2010; 26: 129–37
Guy W. ECDEU assessment manual for psychopharmacology. Rockville (MD): US Department of Health, Education, and Welfare, 1976: 218-22. DHEW publication no.: ADM 76-338
DuPaul GJ, Reid R, Power TJ, et al. ADHD rating scale -IV: checklists, norms, and clinical interpretation. New York: Guildford Publications, 1998
Bennett KJ, Torrance GW. Measuring health status preferences and utilities: rating scale, time trade-off, and standard gamble techniques. In: Spilker B, editor. Quality of life and pharmacoeconomics in clinical trials. 2nd ed. Philadelphia (PA): Lippincott-Raven, 1996: 575–86
Torrance GW. Measurement of health state utilities for economic appraisal. J Health Econ 1986; 5: 1–30
McGough JJ, Wigal SB, Abikoff J, et al. A randomized, double-blind, placebo-controlled, laboratory classroom assessment of methylphenidate transdermal system in children with ADHD. J Atten Disord 2006; 9: 476–85
Conners KC. Conners’ rating scales — revised: technical manual. North Tonawanda, NY: Multi-Health Systems Inc., 2007
Lloyd AJ, van Hanswijck de Jonge P, Doyle S, et al. Health state utility scores for cancer-related anaemia through societal and patient valuations. Value Health 2008; 11(7): 1178–85
Kind P, Dolan P, Gudex C, et al. Variations in population health status: results from a United Kingdom national questionnaire survey. BMJ 1998; 316: 736–41
Matza LS, Secnik K, Rentz AM, et al. Assessment of health state utilities for attention-deficit/hyperactivity disorder in children using parent proxy report. Qual Life Res 2005; 14: 735–47
Kostopoulou O. The transient nature of utilities and health preferences. Med Decis Making 2006; 26: 304–6
The study was funded by Shire Pharmaceuticals, which develops and markets treatment for ADHD. PH and HE are employees and stockholders of Shire Pharmaceuticals. RS was an employee of Shire Pharmaceuticals when the research was undertaken. All three contributed to the conduct of the study, including interpretation of the data, and preparation, review, and approval of the manuscript. AL and AN are employees of Oxford Outcomes, and this company was paid a fixed fee to undertake the study. PF, JB, and RA are all employees of the University of Sheffield who were also paid a fixed fee for their contribution to the study. ESB has received consultancy payments and honoraria from Shire Pharmaceuticals.
AL directed the study, developed the methodology, and took the lead in writing the manuscript. PH conceived the need for the study, contributed to the study throughout, and co-wrote the manuscript. RS contributed to the study methodology throughout and contributed to the manuscript. RA provided a considerable degree of conceptual input at the start of the study and helped to interpret the study findings. ESB provided expert insight into how HR-QOL is affected in ADHD. He also co-wrote large parts of the paper. AN was instrumental in the development of the qualitative study design and planning for analysis. She also co-wrote parts of the manuscript. PF undertook the statistical mapping work. JB contributed to the conception of the study, helped develop the health states, supported the mapping work, and helped co-write parts of the paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Key points for decision makers
• Previous assessments of the cost effectiveness of treatments for ADHD have often relied upon a binary classification of patients as responders or non-responders
• The current work was designed to provide utility estimates reflecting different levels of severity of the patient’s symptoms
• The estimated utility values may offer improved sensitivity and specificity for economic evaluations
• Utilities for responder and non-responder health states were also estimated to provide compatibility with previous economic evaluations
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Lloyd, A., Hodgkins, P., Sasane, R. et al. Estimation of Utilities in Attention-Deficit Hyperactivity Disorder for Economic Evaluations. Patient-Patient-Centered-Outcome-Res 4, 247–257 (2011). https://doi.org/10.2165/11592150-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11592150-000000000-00000