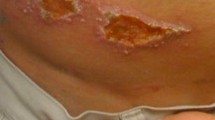
Abstract
Skin ulcers are a commonly encountered problem at departments of tropical dermatology in the Western world. Furthermore, the general dermatologist is likely to be consulted more often for imported chronic skin ulcers because of the ever-increasing travel to and from tropical countries. The most common cause of chronic ulceration throughout the world is probably pyoderma. However, in some parts of the world, cutaneous leishmaniasis is one of the most prevalent causes. Mycobacterium ulcerans is an important cause of chronic ulcers in West Africa.
Bacterial infections include pyoderma, mycobacterial infections, diphtheria, and anthrax. Pyoderma is caused by Staphylococcus aureus and/or β-hemolytic streptococci group A. This condition is a common cause of ulcerative skin lesions in tropical countries and is often encountered as a secondary infection in travelers. The diagnosis is often made on clinical grounds. Antibacterial treatment for pyoderma should preferably be based on culture outcome. Floxacillin is generally active against S. aureus and β-hemolytic streptococci. Infection with Mycobacterium ulcerans, M. marinum, and M. tuberculosis may cause ulcers. Buruli ulcers, which are caused by M. ulcerans, are endemic in foci in West Africa and have been reported as an imported disease in the Western world. Treatment is generally surgical, although a combination of rifampin (rifampicin) and streptomycin may be effective in the early stage. M. marinum causes occasional ulcerating lesions in humans. Treatment regimens consist of combinations containing clarithromycin, rifampin, or ethambutol. Cutaneous tuberculosis is rare in travelers but may be encountered in immigrants from developing countries. Treatment is with multiple drug regimens consisting of isoniazid, ethambutol, pyrazinamide, and rifampin. Cutaneous diphtheria is still endemic in many tropical countries. Cutaneous diphtheria ulcers are nonspecific and erythromycin and penicillin are both effective antibacterials. Antitoxin should be administered intramuscularly in suspected cases. Anthrax is caused by spore-forming Bacillus anthracis. This infection is still endemic in many tropical countries. Eschar formation, which sloughs and leaves behind a shallow ulcer at the site of inoculation, characterizes cutaneous anthrax. Penicillin and doxycycline are effective antibacterials.
Cutaneous leishmaniasis is caused by different species belonging to the genus Leishmania. The disorder is one of the ten most frequent causes of skin diseases in travelers returning from (sub)tropical countries. The clinical picture is diverse, ranging from a painless papule or nodule to an ulcer with or without a scab. Treatment depends on the clinical manifestations and the species involved.
Sporotrichosis, chromo(blasto)mycosis, and mycetoma are the most common mycoses that may be accompanied by ulceration. Infections are restricted to certain regions and often result from direct penetration of the fungus into the skin. Anti-mycotic treatment depends on the microorganism involved.
The most common causes of infectious skin ulceration encountered in patients from tropical countries who present at a department of tropical dermatology are reviewed in this article.


Similar content being viewed by others
References
Ryan ET, Wilson ME, Kain KC. Illness after international travel. N Engl J Med 2002; 347: 505–16
Wilson ME. Skin problems in the traveler. Infect Dis Clin North Am 1998; 12: 471–88
Wilson ME, Chen LH. Dermatologic infectious diseases in international travelers. Curr Infect Dis Rep 2004; 6: 54–62
Lucchina LC, Wilson ME, Drake LA. Dermatology and the recently returned traveler: infectious diseases with dermatologic manifestations. Int J Dermatol 1997; 36: 167–81
Freedman DO, Weld LH, Kozarsky PE,et al. Spectrum of disease and relation to place of exposure among ill returned travelers. N Engl J Med 2006; 354: 119–30
Zeegelaar JE, Steketee WH, van Thiel PP. Changing pattern of imported cutaneous leishmaniasis in the Netherlands. Clin Exp Dermatol 2005; 30: 1–5
Lupi O, Madkan V, Tyring SK. Tropical dermatology: bacterial tropical diseases. J Am Acad Dermatol 2006; 54: 559–78
Gupta SK, Shukla VK. Leg ulcers in the tropics. Int J Low Extrem Wounds 2002; 1: 58–61
Oluwasanmi JO, Alao MO, Ofodile FA. Tropical ulcers. Plast Reconstr Surg 1979; 64: 41–6
Maddox JS, Ware JC, Dillon Jr HC. The natural history of streptococcal skin infection: prevention with topical antibiotics. J Am Acad Dermatol 1985; 13: 207–12
Greene SL, Su WP, Muller SA. Ecthyma gangrenosum: report of clinical, histopathologic, and bacteriologic aspects of eight cases. J Am Acad Dermatol 1984; 11: 781–7
Dillon Jr HC. Post-streptococcal glomerulonephritis following pyoderma. Rev Infect Dis 1979; 1: 935–45
Alausa OK, Montefiore D. Bacterial infections, sensitivity patterns, and chemotherapy among hospital patients in the tropics. Scand J Infect Dis 1978; 10: 295–302
Rotta J, Tikhomirov E. Streptococcal diseases worldwide: present status and prospects. Bull World Health Organ 1987; 65: 769–77
Mahe A, Prual A, Konate M,et al. Skin diseases of children in Mali: a public health problem. Trans R Soc Trop Med Hyg 1995; 89: 467–70
Brahmadathan KN, Koshi G. Epidemiology of streptococcal pyoderma in an orphanage community of a tropical country. J Trop Med Hyg 1988; 91: 306–14
Caumes E, Carriere J, Guermonprez G,et al. Dermatoses associated with travel to tropical countries: a prospective study of the diagnosis and management of 269 patients presenting to a tropical disease unit. Clin Infect Dis 1995; 20: 542–8
Naafs B. Tropical holiday memories. Eur J Dermatol 1999; 9: 500–5
Brahmadathan KN, Koshi G. Importance of group G streptococci in human pyogenic infections. J Trop Med Hyg 1989; 92: 35–8
Suite M. Cutaneous infections in Trinidad. Int J Dermatol 1990; 29: 31–4
Allen AM, Taplin D, Twigg L. Cutaneous streptococcal infections in Vietnam. Arch Dermatol 1971; 104: 271–80
Sheagren JN. Staphylococcal infections of the skin and skin structures. Cutis 1985; 36: 2–6
Bisno AL, Stevens DL. Streptococcal infections of skin and soft tissues. N Engl J Med 1996; 334: 240–5
Lowy FD. Staphylococcus aureus infections. N Engl J Med 1998; 339: 520–32
Martin JM, Green M. Group A streptococcus. Semin Pediatr Infect Dis 2006; 17: 140–8
Basak S, Dutta SK, Gupta S,et al. Bacteriology of wound infection: evaluation by surface swab and quantitative full thickness wound biopsy culture. J Indian Med Assoc 1992; 90: 33–4
Slater RA, Lazarovitch T, Boldur I,et al.Swab cultures accurately identify bacterial pathogens in diabetic foot wounds not involving bone. Diabet Med 2004; 21: 705–9
Bill TJ, Ratliff CR, Donovan AM,et al. Quantitative swab culture versus tissue biopsy: a comparison in chronic wounds. Ostomy Wound Manage 2001; 47: 34–7
Perry CR, Pearson RL, Miller GA. Accuracy of cultures of material from swabbing of the superficial aspect of the wound and needle biopsy in the preoperative assessment of osteomyelitis. J Bone Joint Surg Am 1991; 73: 745–9
Nagaraju U, Bhat G, Kuruvila M,et al. Methicillin-resistant Staphylococcus aureus in community acquired pyoderma. Int J Dermatol 2004; 43: 412–4
Zeegelaar IE, Langenberg W, Hu R,et al. Tolerability and efficacy of hydrocolloid dressings in the treatment of venous leg ulcers under tropical conditions: an open prospective study. J Eur Acad Dermatol Venereol 2001; 15: 234–7
Price JD, Harding JW. Flucloxacillin in the treatment of infectious conditions in children. Curr Med Res Opin 1975; 3: 77–82
Duncan JT. A clinical appraisal of flucloxacillin in the management of skin and soft tissue infections in Nigeria. J Int Med Res 1984; 12: 210–5
Crofts HG. Flucloxacillin (Floxapen) in the treatment of skin and upper respiratory tract infections. N Z Med J 1978; 87: 308–11
Korting HC, Neubert U, Abeck D. Current antimicrobial susceptibility of cutaneous bacteria to firstline antibiotics. Int J Antimicrob Agents 1998; 10: 165–8
Moellering JrRC. Past, present, and future of antimicrobial agents. Am J Med 1995; 99: 11–8S
Fung HB, Chang JY, Kuczynski S. A practical guide to the treatment of complicated skin and soft tissue infections. Drugs 2003; 63: 1459–80
Staphylococcus aureus resistant to vancomycin: United States, 2002. MMWR Morb Mortal Wkly Rep 2002; 51: 565–7
Winter GD, Scales JT. Effect of air drying and dressings on the surface of a wound. Nature 1963; 197: 91–2
Hautmann G, Lotti T. Atypical mycobacterial infections of the skin. Dermatol Cli 1994; 12: 657–68
Kostman JR, DiNubile MJ. Nodular lymphangitis: a distinctive but often unrecognized syndrome. Ann Intern Med 1993; 118: 883–8
van der Werf TS, van der Graaf WT, Tappero JW,. Mycobacterium ulcerans infection. Lancet 1999; 354: 1013–8
Dodiuk-Gad R, Dyachenko P, Ziv M, et al. Nontuberculous mycobacterial infections of the skin: a retrospective study of 25 cases. J Am Acad Dermatol 2007; 57 (3): 413–20
Portaels F. Epidemiology of mycobacterial diseases. Clin Dermatol 1995; 13: 207–22
Wayne LG, Sramek HA. Agents of newly recognized or infrequently encountered mycobacterial diseases. Clin Microbiol Rev 1992; 5: 1–25
Street ML, Umbert-Millet IJ, Roberts GD,et al. Nontuberculous mycobacterial infections of the skin: report of fourteen cases and review of the literature. J Am Acad Dermatol 1991; 24: 208–15
Clancey JK. Mycobacterial skin ulcers in Uganda: description of a new mycobacterium (Mycobacterium buruli). J Pathol Bacteriol 1964; 88: 175–87
Portaels F, Chemlal K, Elsen P,et al. Mycobacterium ulcerans in wild animals. Rev Sci Tech 2001; 20: 252–64
Eddyani M, Ofori-Adjei D, Teugels G,et al. Potential role for fish in transmission of Mycobacterium ulcerans disease (Buruli ulcer): an environmental study. Appl Environ Microbiol 2004; 70: 5679–81
Portaels F, Elsen P, Guimaraes-Peres A, et al. Insects in the transmission of Mycobacterium ulcerans infection [letter]. Lancet 1999; 353: 986
Marston BJ, Diallo MO, Horsburgh Jr CR, et al. Emergence of Buruli ulcer disease in the Daloa region of Cote d’Ivoire. Am J Trop Med Hyg 1995; 52: 219–24
Debacker M, Aguiar J, Steunou C, et al. Buruli ulcer recurrence, Benin. Emerg Infect Dis 2005; 11: 584–9
Amofah G, Bonsu F, Tetteh C, et al. Buruli ulcer in Ghana: results of a national case search. Emerg Infect Dis 2002; 8: 167–70
Barker DJ. Epidemiology of Mycobacterium ulcerans infection. Trans R Soc Trop Med Hyg 1973; 67: 43–50
Semret M, Koromihis G, MacLean JD,et al. Mycobacterium ulcerans infection (Buruli ulcer): first reported case in a traveler. Am J Trop Med Hyg 1999; 61: 689–93
Faber WR, Arias-Bouda LM, Zeegelaar JE,et al. First reported case of Mycobacterium ulcerans infection in a patient from China. Trans R Soc Trop Med Hyg 2000; 94: 277–9
Evans MR, Mawdsley J, Bull R,et al. Buruli ulcer in a visitor to London. Br J Dermatol 2003; 149: 907–9
Ross BC, Johnson PD, Oppedisano F, et al. Detection of Mycobacterium ulcerans in environmental samples during an outbreak of ulcerative disease. Appl Environ Microbiol 1997; 63: 4135–8
Portaels F, Meyers WM. Buruli ulcer. In: Faber WR, Hay RJ, Naafs B, editors. Imported skin diseases. Maarssen: Elsevier, 2006
Hayman J. Out of Africa: observations on the histopathology of Mycobacterium ulcerans infection. J Clin Pathol 1993; 46: 5–9
Meyers WM, Shelly WM, Connor DH. Heat treatment of Mycobacterium ulcerans infections without surgical excision. Am J Trop Med Hyg 1974; 23: 924–9
Palomino JC, Obiang AM, Realini L,et al. Effect of oxygen on growth of Mycobacterium ulcerans in the BACTEC system. J Clin Microbiol 1998; 36: 3420–2
Roberts B, Hirst R. Immunomagnetic separation and PCR for detection of Mycobacterium ulcerans. J Clin Microbiol 1997; 35: 2709–11
Ross BC, Marino L, Oppedisano F, et al. Development of a PCR assay for rapid diagnosis of Mycobacterium ulcerans infection. J Clin Microbiol 1997; 35: 1696–700
Stienstra Y, van der Werf TS, Guarner J,et al. Analysis of an IS2404-based nested PCR for diagnosis of Buruli ulcer disease in regions of Ghana where the disease is endemic. J Clin Microbiol 2003; 41: 794–7
Buruli ulcer disease. Wkly Epidemiol Rec 2004; 79: 194–9
Dega H, Bentoucha A, Robert J,et al. Bactericidal activity of rifampin-amikacin against Mycobacterium ulcerans in mice. Antimicrob Agents Chemother 2002; 46: 3193–6
Gluckman SJ. Mycobacterium marinum. Clin Dermatol 1995; 13: 273–6
Lewis FM, Marsh BJ, von Reyn CF. Fish tank exposure and cutaneous infections due to Mycobacterium marinum: tuberculin skin testing, treatment, and prevention. Clin Infect Dis 2003; 37: 390–7
Chemlal K, Portaels F. Molecular diagnosis of nontuberculous mycobacteria. Curr Opin Infect Dis 2003; 16: 77–83
Bayer-Garner IB, Cox MD, Scott MA,et al. Mycobacteria other than Mycobacterium tuberculosis are not present in erythema induratum/nodular vasculitis: a case series and literature review of the clinical and histologic findings. J Cutan Pathol 2005; 32: 220–6
Braback M, Riesbeck K, Forsgren A. Susceptibilities of Mycobacterium marinum to gatifloxacin, gemifloxacin, levofloxacin, linezolid, moxifloxacin, telithromycin, and quinupristin-dalfopristin (Synercid) compared to its susceptibilities to reference macrolides and quinolones. Antimicrob Agents Chemother 2002; 46: 1114–6
Gimenez GR, Sanchez RS, Iban Ochoa RM, et al. Fish tank granuloma: response to treatment with clarithromycin. J Eur Acad Dermatol Venereol 1999; 12: 81–2
Yates VM, Ormerod LP. Cutaneous tuberculosis in Blackburn district (U.K.): a 15-year prospective series, 1981-95. Br J Dermatol 1997; 136: 483–9
Beutner EH. Tuberculosis of the skin: historical perspectives on tuberculin and Bacille Calmette Guerin. Int J Dermatol 1997; 36: 73–7
MacGregor RR. Cutaneous tuberculosis. Clin Dermatol 1995; 13: 245–55
Barbagallo J, Tager P, Ingleton R,et al. Cutaneous tuberculosis: diagnosis and treatment. Am J Clin Dermatol 2002; 3: 319–28
Seckin D, Akpolat T, Ceyhan M,et al. Polymerase chain reaction in cutaneous tuberculosis. Int J Dermatol 1997; 36: 51–4
Hofler W. Cutaneous diphtheria. Int J Dermatol 1991; 30: 845–7
Wagner J, Ignatius R, Voss S, et al. Infection of the skin caused by Corynebacterium ulcerans and mimicking classical cutaneous diphtheria. Clin Infect Dis 2001; 33: 1598–600
Pandit N, Yeshwanth M. Cutaneous diphtheria in a child. Int J Dermatol 1999; 38: 298–9
Koopman JS, Campbell J. The role of cutaneous diphtheria infections in a diphtheria epidemic. J Infect Dis 1975; 131: 239–44
Kjeldsen K, Simonsen O, Heron I. Immunity against diphtheria 25-30 years after primary vaccination in childhood. Lancet 1985; I: 900–2
de Benoist AC, White JM, Efstratiou A,et al. Imported cutaneous diphtheria, United Kingdom. Emerg Infect Dis 2004; 10: 511–3
Van den Enden E, Van Gompel A, Van Esbroeck M. Cutaneous anthrax, Belgian traveler. Emerg Infect Dis 2006; 12: 523–5
Swartz MN. Recognition and management of anthrax: an update. N Engl J Med 2001; 345: 1621–6
Dixon TC, Meselson M, Guillemin J,et al. Anthrax. N Engl J Med 1999; 341: 815–26
Sternbach G. The history of anthrax. J Emerg Med 2003; 24: 463–7
Shlyakhov E, Rubinstein E. Evaluation of the anthraxin skin test for diagnosis of acute and past human anthrax. Eur J Clin Microbiol Infect Dis 1996; 15: 242–5
Desjeux P. Leishmaniasis: public health aspects and control. Clin Dermatol 1996; 14: 417–23
Uzun S, Uslular C, Yucel A, et al. Cutaneous leishmaniasis: evaluation of 3,074 cases in the Cukurova region of Turkey. Br J Dermatol 1999; 140: 347–50
Rowland M, Munir A, Durrani N,et al. An outbreak of cutaneous leishmaniasis in an Afghan refugee settlement in north-west Pakistan. Trans R Soc Trop Med Hyg 1999; 93: 133–6
Brooker S, Mohammed N, Adil K, et al. Leishmaniasis in refugee and local Pakistani populations. Emerg Infect Dis 2004; 10: 1681–4
Cupolillo E, Aguiar AF, Brahim LR, et al. Recent advances in the taxonomy of the New World leishmanial parasites. Med Microbiol Immunol (Berl) 2001; 190: 57–60
Cupolillo E, Brahim LR, Toaldo CB, et al. Genetic polymorphism and molecular epidemiology of Leishmania (Viannia) braziliensis from different hosts and geographic areas in Brazil. J Clin Microbiol 2003; 41: 3126–32
Marfurt J, Niederwieser I, Makia ND,et al. Diagnostic genotyping of Old and New World Leishmania species by PCR-RFLP. Diagn Microbiol Infect Dis 2003; 46: 115–24
Cupolillo E, Momen H, Grimaldi Jr G. Genetic diversity in natural populations of New World Leishmania. Mem Inst Oswaldo Cruz 1998; 93: 663–8
Lainson R, Shaw JJ. The laboratory mix-up of Leishmania strains [letter]. Trans R Soc Trop Med Hyg 1990; 84: 753
Shaw JJ. Taxonomy of the genus Leishmania: present and future trends and their implications. Mem Inst Oswaldo Cruz 1994; 89: 471–8
Rioux JA, Lanotte G, Serres E,et al. Taxonomy of Leishmania: use of isoenzymes. Suggestions for a new classification. Ann Parasitol Hum Comp 1990; 65: 111–25
Killick-Kendrick R. Phlebotomine vectors of the leishmaniases: a review. Med Vet Entomol 1990; 4: 1–24
Killick-Kendrick R. The biology and control of phlebotomine sand flies. Clin Dermatol 1999; 17: 279–89
Desjeux P. Leishmaniasis: current situation and new perspectives. Comp Immunol Microbiol Infect Dis 2004; 27: 305–18
Bauer IL. Knowledge and behavior of tourists to Manu National Park, Peru, in relation to leishmaniasis. J Travel Med 2002; 9: 173–9
El Hajj L, Thellier M, Carriere J,et al. Localized cutaneous leishmaniasis imported into Paris: a review of 39 cases. Int J Dermatol 2004; 43: 120–5
Harms G, Schonian G, Feldmeier H. Leishmaniasis in Germany. Emerg Infect Dis 2003; 9: 872–5
Schwartz E, Hatz C, Blum J. New World cutaneous leishmaniasis in travellers. Lancet Infect Dis 2006; 6: 342–9
Guiguemde RT, Sawadogo OS, Bories C, et al. Leishmania major and HIV co-infection in Burkina Faso. Trans R Soc Trop Med Hyg 2003; 97: 168–9
Maingon RD, Ward RD, Hamilton JG,et al. The Lutzomyia longipalpis species complex: does population sub-structure matter to leishmania transmission? Trends Parasitol 2008 Jan; 24 (1): 12–7
Uthman MA, Satir AA, Tabbara KS. Clinical and histopathological features of zoonotic cutaneous leishmaniasis in Saudi Arabia. J Eur Acad Dermatol Venereol 2005; 19: 431–6
Jacobson RL, Eisenberger CL, Svobodova M,et al. Outbreak of cutaneous leish-maniasis in northern Israel. J Infect Dis 2003; 188: 1065–73
Al Tawfiq JA, AbuKhamsin A. Cutaneous leishmaniasis: a 46-year study of the epidemiology and clinical features in Saudi Arabia (1956-2002). Int J Infect Dis 2004; 8: 244–50
Sharma NL, Mahajan VK, Kanga A,et al. Localized cutaneous leishmaniasis due to Leishmania donovani and Leishmania tropica: preliminary findings of the study of 161 new cases from a new endemic focus in Himachal Pradesh, India. Am J Trop Med Hyg 2005; 72: 819–24
Barnetson RS, Ridley RS, Wheate HW. A form of muco-cutaneous leishmaniasis in the Old World. Trans R Soc Trop Med Hyg 1978; 72: 516–8
Morsy TA, Khalil NM, Salama MM,et al. Mucosal leishmaniasis caused by Leishmania tropica in Saudi Arabia. J Egypt Soc Parasitol 1995; 25: 73–9
Oliveira CC, Lacerda HG, Martins DR,et al. Changing epidemiology of American cutaneous leishmaniasis (ACL) in Brazil: a disease of the urban-rural interface. Acta Trop 2004; 90: 155–62
Romero GA, Ishikawa E, Cupolillo E,et al. The rarity of infection with Leishmania (Viannia) braziliensis among patients from the Manaus region of Amazonas state, Brazil, who have cutaneous leishmaniasis. Ann Trop Med Parasitol 2002; 96: 131–6
Corredor A, Kreutzer RD, Tesh RB,et al. Distribution and etiology of leishmaniasis in Colombia. Am J Trop Med Hyg 1990; 42: 206–14
Velez I, Agudelo S, Robledo S,et al. Diffuse cutaneous leishmaniasis with mucosal involvement in Colombia, caused by an enzymatic variant of Leishmania panamensis. Trans R Soc Trop Med Hyg 1994; 88: 199
Turetz ML, Machado PR, Ko AI,et al. Disseminated leishmaniasis: a new and emerging form of leishmaniasis observed in northeastern Brazil. J Infect Dis 2002; 186: 1829–34
Silveira FT, Lainson R, Corbett CE. Clinical and immunopathological spectrum of American cutaneous leishmaniasis with special reference to the disease in Amazonian Brazil: a review. Mem Inst Oswaldo Cruz 2004; 99: 239–51
Silveira FT, Lainson R, Corbett CE. Further observations on clinical, histopathological, and immunological features of borderline disseminated cutaneous leishmaniasis caused by Leishmania (Leishmania) amazonensis. Mem Inst Oswaldo Cruz 2005; 100: 525–34
Reithinger R, Aadil K, Hami S,et al. Cutaneous leishmaniasis, northern Afghanistan. Emerg Infect Dis 2004; 10: 966–7
Magill AJ. Epidemiology of the leishmaniases. Dermatol Clin 1995; 13: 505–23
Navin TR, Arana FE, de Merida AM,et al. Cutaneous leishmaniasis in Guatemala: comparison of diagnostic methods. Am J Trop Med Hyg 1990; 42: 36–42
De Bruijn MH, Barker DC. Diagnosis of New World leishmaniasis: specific detection of species of the Leishmania braziliensis complex by amplification of kinetoplast DNA. Acta Trop 1992; 52: 45–58
Weigle KA, de Davalos M, Heredia P,et al. Diagnosis of cutaneous and mucocutaneous leishmaniasis in Colombia: a comparison of seven methods. Am J Trop Med Hyg 1987; 36: 489–96
Kalter DC. Laboratory tests for the diagnosis and evaluation of leishmaniasis. Dermatol Clin 1994; 12: 37–50
Faber WR, Oskam L, van Gool T,et al. Value of diagnostic techniques for cutaneous leishmaniasis. J Am Acad Dermatol 2003; 49: 70–4
Sarantopoulos GP, Binder S, Wortmann G,et al. Old World cutaneous leishmaniasis in Los Angeles: a case report, overview of the current literature, and guide for the treating dermatopathologist. Am J Dermatopathol 2003; 25: 321–6
Herwaldt BL, Berman JD. Recommendations for treating leishmaniasis with sodium stibogluconate (Pentostam) and review of pertinent clinical studies. Am J Trop Med Hyg 1992; 46: 296–306
World Health Organization. The leishmaniases. WHO technical report series. Vol 701. Geneva: WHO, 1984
Romero GA, Guerra MV, Paes MG,et al. Comparison of cutaneous leishmaniasis due to Leishmania (Viannia) braziliensis and L. (V.) guyanensis in Brazil: therapeutic response to meglumine antimoniate. Am J Trop Med Hyg 2001; 65: 456–65
Oliveira-Neto MP, Schubach A, Mattos M,et al. Treatment of American cutaneous leishmaniasis: a comparison between low dosage (5 mg/kg/day) and high dosage (20 mg/kg/day) antimony regimens. Pathol Biol (Paris) 1997; 45: 496–9
Oliveira-Neto MP, Schubach A, Mattos M,et al. Intralesional therapy of American cutaneous leishmaniasis with pentavalent antimony in Rio de Janeiro, Brazil: an area of Leishmania (V.) braziliensis transmission. Int J Dermatol 1997; 36: 463–8
Blum J, Desjeux P, Schwartz E,et al. Treatment of cutaneous leishmaniasis among travellers. J Antimicrob Chemother 2004; 53: 158–66
Lai A Fat EJ, Vrede MA, Soetosenojo RM,et al. Pentamidine, the drug of choice for the treatment of cutaneous leishmaniasis in Surinam. Int J Dermatol 2002; 41: 796–800
Tuon FF, Amato VS, Graf ME,et al. Treatment of New World cutaneous leishmaniasis: a systematic review with a meta-analysis. Int J Dermatol 2008 Feb; 47 (2): 109–24
Soto J, Rea J, Balderrama M,et al. Efficacy of miltefosine for Bolivian cutaneous leishmaniasis. Am J Trop Med Hyg 2008 Feb; 78 (2): 210–1
Berman J. Miltefosine to treat leishmaniasis. Expert Opin Pharmacother 2005; 6: 1381–8
Soto J, Soto P. Miltefosine: oral treatment of leishmaniasis. Expert Rev Anti Infect Ther 2006; 4: 177–85
Soto J, Toledo J, Valda L,et al. Treatment of Bolivian mucosal leishmaniasis with miltefosine. Clin Infect Dis 2007; 44: 350–6
El Safi SH, Murphy AG, Bryceson AD,et al. A double-blind clinical trial of the treatment of cutaneous leishmaniasis with paromomycin ointment. Trans R Soc Trop Med Hyg 1990; 84: 690–1
El On J, Halevy S, Grunwald MH,et al. Topical treatment of Old World cutaneous leishmaniasis caused by Leishmania major: a double-blind control study. J Am Acad Dermatol 1992; 27: 227–31
Gurei MS, Tatli N, Ozbilge H,et al. Efficacy of cryotherapy and intralesional Pentostam in treatment of cutaneous leishmaniasis. J Egypt Soc Parasitol 2000; 30: 169–76
Faris RM, Jarallah JS, Khoja TA,et al. Intralesional treatment of cutaneous leishmaniasis with sodium stibogluconate antimony. Int J Dermatol 1993; 32: 610–2
Asilian A, Sadeghinia A, Faghihi G,et al. Comparative study of the efficacy of combined cryotherapy and intralesional meglumine antimoniate (Glucantime) vs. cryotherapy and intralesional meglumine antimoniate (Glucantime) alone for the treatment of cutaneous leishmaniasis. Int J Dermatol 2004; 43: 281–3
Alrajhi AA, Ibrahim EA, De Vol EB,et al. Fluconazole for the treatment of cutaneous leishmaniasis caused by Leishmania major. N Engl J Med 2002; 346: 891–5
Weinrauch L, Livshin R, El On J. Ketoconazole in cutaneous leishmaniasis. Br J Dermatol 1987; 117: 666–8
Minodier P, Parola P. Cutaneous leishmaniasis treatment. Travel Med Infect Dis 2007 May; 5 (3): 150–8
Reithinger R, Mohsen M, Wahid M,et al. Efficacy of thermotherapy to treat cutaneous leishmaniasis caused by Leishmania tropica in Kabul, Afghanistan: a randomized, controlled trial. Clin Infect Dis 2005; 40: 1148–55
De Araujo T, Marques AC, Kerdel F. Sporotrichosis. Int J Dermatol 2001; 40: 737–42
Barros MB, Schubach AO, do Valle AC, et al. Cat-transmitted sporotrichosis epidemic in Rio de Janeiro, Brazil: description of a series of cases. Clin Infect Dis 2004; 38: 529–35
Schubach A, Schubach TM, Barros MB, et al. Cat-transmitted sporotrichosis, Rio de Janeiro, Brazil. SEmerg Infect Dis 2005; 11: 1952–4
da Rosa AC, Scroferneker ML, Vettorato R, et al. Epidemiology of sporotrichosis: a study of 304 cases in Brazil. J Am Acad Dermatol 2005; 52: 451–9
Neyra E, Fonteyne PA, Swinne D, et al. Epidemiology of human sporotrichosis investigated by amplified fragment length polymorphism. J Clin Microbiol 2005; 43: 1348–52
Bustamante B, Campos PE. Endemic sporotrichosis. Curr Opin Infect Dis 2001; 14: 145–9
Quintal D. Sporotrichosis infection on mines of the Witwatersrand. J Cutan Med Surg 2000; 4: 51–4
Werner AH, Werner BE. Sporotrichosis in man and animal. Int J Dermatol 1994; 33: 692–700
Carvalho MT, de Castro AP, Baby C, et al. Disseminated cutaneous sporotrichosis in a patient with AIDS: report of a case. Rev Soc Bras Med Trop 2002; 35: 655–9
Bullpitt P, Weedon D. Sporotrichosis: a review of 39 cases. Pathology 1978; 10: 249–56
Winn RE, Anderson J, Piper J, et al. Systemic sporotrichosis treated with itraconazole. Clin Infect Dis 1993; 17: 210–7
Conti Diaz IA, Civila E, Gezuele E, et al. Treatment of human cutaneous sporotrichosis with itraconazole. Mycoses 1992; 35: 153–6
Chapman SW, Pappas P, Kauffmann C, et al. Comparative evaluation of the efficacy and safety of two doses of terbinafine (500 and 1000 mg day(-1)) in the treatment of cutaneous or lymphocutaneous sporotrichosis. Mycoses 2004; 47: 62–8
Cabezas C, Bustamante B, Holgado W, et al. Treatment of cutaneous sporotrichosis with one daily dose of potassium iodide. Pediatr Infect Dis J 1996; 15: 352–4
Kauffman CA. Old and new therapies for sporotrichosis. Clin Infect Dis 1995; 21: 981–5
Bonifaz A, Carrasco-Gerard E, Saul A. Chromoblastomycosis: clinical and mycologic experience of 51 cases. Mycoses 2001; 44: 1–7
Lupi O, Tyring SK, McGinnis MR. Tropical dermatology: fungal tropical diseases. J Am Acad DermatolS 2005; 53: 931–51, quiz
Rivitti EA, Aoki V. Deep fungal infections in tropical countries. SClin Dermatol 1999; 17: 171–90
Howles JK, Kennedy CB, Garvin WH, et al. Chromoblastomycosis: report of nine cases from a single area in Louisiana. AMA Arch Derm Syphilol 1954; 69: 83–90
Bonifaz A, Paredes-Solis V, Saul A. Treating chromoblastomycosis with systemic antifungals. SExpert Opin Pharmacother 2004; 5: 247–54
Magana M, Magana-Garcia M. Mycetoma. Dermatol Clin 1989; 7: 203–17
McGinnis MR. Mycetoma. Dermatol Clin 1996; 14: 97–104
Lichon V, Khachemoune A. Mycetoma: a review. Am J Clin Dermatol 2006; 7: 315–21
Fahal AH. Mycetoma: a thorn in the flesh. Trans R Soc Trop Med Hyg 2004; 98: 3–11
Fahal AH, Sharfi AR, Sheik HE, et al. Internal fistula formation: an unusual complication of mycetoma. Trans R Soc Trop Med Hyg 1996; 90: 550–2
Fahal AH, Hassan MA. Mycetoma. Br J Surg 1992; 79: 1138–41
Welsh O, Salinas MC, Rodriguez MA. Treatment of eumycetoma and actino- mycetoma. Curr Top Med Mycol 1995; 6: 47–71
Welsh O. Mycetoma: current concepts in treatment. Int J Dermatol 1991; 30: 387–98
Bonifaz A, Flores P, Saul A, et al. Treatment of actinomycetoma due to Nocardia spp. with amoxicillin-clavulanate. Br J Dermatol 2007; 156: 308–11
Acknowledgments
No sources of funding were used to assist in the preparation of this review. The authors have no conflicts of interest that are directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zeegelaar, J.E., Faber, W.R. Imported Tropical Infectious Ulcers in Travelers. Am J Clin Dermatol 9, 219–232 (2008). https://doi.org/10.2165/00128071-200809040-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00128071-200809040-00002