Abstract
Background: Recent studies have reported abnormal functional connectivity patterns in the brains of people with autism that may be accompanied by decreases in white matter integrity. Since autism is a developmental disorder, we aim to investigate the nature and location of decreases in white and grey matter integrity in an adolescent sample while accounting for age.
Methods: We used structural (T1) imaging to study brain volumetrics and diffusion tensor imaging (DTI) to investigate white and grey matter integrity in people with autism. We obtained magnetic resonance images for adolescents aged 12–18 years with high-functioning autism and from matched controls. Fractional anisotropy and mean diffusivity, as well as grey and white matter volumetrics were analyzed.
Results: There were 17 participants with autism and 25 matched controls included in this study. Participants with autism had lower fractional anisotropy in the left and right superior and inferior longitudinal fasciculus, but this effect was not significant after adjusting for age and intelligence quotient (IQ). The kurtosis of the white matter fractional anisotropy probability distribution was higher in this participant group, with and without adjustment for age and IQ. Most notably, however, the mean diffusivity levels were markedly increased in the autism group throughout the brain, and the mean diffusivity probability distributions of both grey and white matter were shifted toward a higher value, particularly with age and IQ adjustment. No volumetric differences in grey and white matter were found.
Limitations: We corrected for age and IQ using a linear model. The study was also limited by its sample size, investigated age range and cross-sectional design.
Conclusion: The findings suggest that autism is characterized by a generalized reduction of white matter integrity that is associated with an increase of interstitial space. The generalized manifestation of the white matter abnormalities provides an important new perspective on autism as a connectivity disorder.
Introduction
Recently, various researchers have proposed that impaired integration of information underlies autism, and that this impairment results from abnormal neural connectivity.1–3 Converging evidence has shown deficient connectivity in tasks involving language comprehension,2,4 working memory5 and action planning6 using functional magnetic resonance imaging (fMRI); listening using magnetoencephalography;7 mentalizing using positron emission tomography,8 and also in resting state networks using fMRI.9 These studies, however, address functional connectivity between grey matter areas, which should be differentiated from structural (anatomic) connectivity associated with the intermediating white matter tracts. How or to what extent functional and structural connectivity are related often remains unclear. Yet, in general, it is believed that abnormal functional connectivity does not necessitate abnormal structural connectivity, but rather that deficient structural connectivity commonly results in a lack of functional connectivity.10,11 A comprehensive understanding of neural connectivity in people with autism therefore requires clear evidence as to whether structural connectivity is affected in these individuals.
Currently, most evidence is still indirect and merely describes global white matter properties. A volumetric MRI study found an abnormal developmental trajectory of white matter in children with autism: they had normal head circumference at birth and increased cerebral and cerebellar white matter volume at 2–3 years of age that normalized again in later years.12 This pattern was confirmed in additional analyses of head circumferences, postmortem findings and MRI measurements,13,14 which led the authors to argue that the early overgrowth interferes with the normal developmental trajectory of cortical connectivity. In a qualitative review, data were combined from 6 studies on the development of white matter volume from 2 to 20 years of age.15 Again, the same developmental trajectory was found, with a white matter increase in early childhood that normalized at around 12 years of age. This developmental finding underlines the importance of using samples with a narrow age range when studying autism, and it raises the question as to whether the integrity of white matter at the age of 12 years and above is affected. Morphologic studies have shown that in typical populations, brain structures mature over a posterior–anterior axis, from primary to higher association areas.16 It is therefore conceivable that as the brain in people with autism develops abnormally, the late developing pre-frontal and superior-temporal areas show the most pronounced defects in adolescence.17
A more direct and informative method for investigating in vivo white matter integrity is diffusion tensor imaging (DTI), an MRI-based technique that measures the directional diffusion profile of water molecules, which manifests the axonal architecture of the brain at the micrometer level. Fractional anisotropy and mean diffusivity are 2 measures derived from diffusion tensor data.18,19 Fractional anisotropy and mean diffusivity provide an index for the integrity of neural tissue, and more specifically, fractional anisotropy provides an indication of the directionality of white matter microstructural architecture and mean diffusivity of the interstitial space in both white and grey matter. So far, a limited number of studies have used DTI for the study of autism (Table 1). Barnea-Goraly and colleagues20 were the first to apply DTI to a small number of autistic children and controls. Using a voxel-based approach that excluded the cerebellum, they found reduced fractional anisotropy in the corpus callosum and in the white matter of the ventromedial prefrontal cortices, anterior cingulate gyri and temporoparietal junctions, indicating a reduction in white matter integrity in the autism group. A more recent study that used a voxel-based approach in a large sample of autistic children and adults showed a fractional anisotropy reduction within and near the corpus callosum and internal capsule.21 The authors argued that these reductions did not reflect slowing of white matter development but rather that they persist into adulthood. The effect size did not allow for correction for multiple comparisons, necessitating confirmation of the findings. Findings of reduced fractional anisotropy in the frontal lobe and left temporal lobe were reported in an exploratory study on a small group of Chinese children with high-functioning autism.22 Neither of these studies reported on mean diffusivity. A DTI study of the corpus callosum in a large sample of autistic children and adults found a reduction of fractional anisotropy and increase of mean diffusivity, indicating a reduced integrity of the genu, body and splenium of the corpus callosum.23 Analysis of the superior temporal gyrus and temporal stem in the same participant group also showed a fractional anisotropy reduction and mean diffusivity increase.24 Another recent DTI study used tractography of the frontal lobe25 and found reduced fractional anisotropy and increased mean diffusivity along the short frontal association fibres and reduced fractional anisotropy along the long frontal fibres. Although the patient group was large, the study sample was also heterogeneous since it included children with autism, pervasive developmental disorder not otherwise specified and Asperger disorder. A cerebellar DTI study in adults with Asperger syndrome also found reduced fractional anisotropy, but mean diffusivity did not differ between the patient and control groups.26 Contrary to the other DTI findings in populations with autism, a high b-value, diffusion-weighted imaging study of 7 toddlers (aged 1.8–3.3 yr) with autism compared with 41 healthy controls found an increase in fractional anisotropy as well as increased probability for zero displacement and a reduced mean displacement probability that was most prominent in the left frontal lobe.27 Measures of mean diffusivity were not reported, but the displacement findings imply a decrease in mean diffusivity in these areas. It should be noted, however, that high b-value imaging primarily depicts intracellular diffusion properties, whereas conventional DTI is believed to mostly reflect extra-cellular diffusion.28 The authors interpreted their findings as evidence for accelerated white matter maturation in young children with autism.
Diffusion tensor imaging studies on autism
There is a need to add whole-brain DTI analyses in a homogeneous sample (both in age and diagnosis) on both the fractional anisotropy and mean diffusivity measures to the autism literature. Furthermore, as autism is a developmental disorder, differences in brain structures change over time. Therefore, it is interesting to relate these to DTI findings and to make group inferences on the neuroanatomy of autism without developmental changes as a confounder. We set out to address these questions in the current DTI study by extracting the main moments of a participant’s fractional anisotropy and mean diffusivity probability distribution in grey and white matter and compare these moments at the group level using a homogeneous adolescent sample of participants with autism and well-matched controls. Further, we investigated the spatial location of voxel-wise differences in fractional anisotropy and mean diffusivity between these groups by using whole-brain, search volume–corrected t statistics. In addition, we looked into the spatial location of voxel-wise volumetric differences in white matter using voxel-based morphometry (VBM)29 in T1 images and correlated the outcome with the voxel-wise DTI results. We hypothesized that in our adolescent sample, people with autism would show a decrease in white matter integrity (i.e., abnormalities in fractional anisotropy or mean diffusivity), whereas white matter volumes would not differ between groups, as indicated by previous studies (see a review15). Absent volumetric abnormalities in the presence of white matter integrity differences would suggest that volumetric differences cannot account for differences in integrity. Since cortical maturation follows an abnormal developmental trajectory in autism, we also hypothesized that the effects would be most pronounced in the late-developing higher-association areas such as the prefrontal and superior-temporal areas. As the development of grey and white matter is closely related, we predicted that we would find greater fractional anisotropy and mean diffusivity differences in the temporal and frontal regions, and smaller or no differences at all in the parietal and occipital regions.
Methods
Participants
We included typically developing adolescents (controls) and adolescents with autism aged 12–18 years in the study. We obtained written informed consent from all participants and their parents. The local medical ethics committee (CMO regio Arnhem-Nijmegen) approved our study.
We recruited the participants with autism through Karakter, Child and Adolescent Psychiatry University Center, Nijmegen. Diagnostic assignment followed DSM-IV criteria for autistic disorder.30 Diagnostic characterization included the Autism Diagnostic Interview — Revised (ADI-R)31 as assessed by a trained clinician and a series of clinical assessments, which included detailed developmental history, clinical observation, medical work-up and cognitive testing. The participants with autism were tested with the full Wechsler Intelligence Scale for Children III.32 Only participants with an intelligence quotient (IQ) of 80 or higher were included. To screen for the presence of comorbid psychiatric disorders or learning problems in the controls, Child Behavior Checklist (CBCL) questionnaires33 were completed by the parents or guardians of the controls and Teacher Report Form (TRF) questionnaires34 were completed by a teacher at school. Exclusion criteria were any medical condition affecting central nervous system function, neurologic disorders, substance abuse and a family history of psychiatric disorders.
For the control group, we assessed more than 200 children from local high schools for verbal IQ, performance IQ and full-scale IQ using a short form of the Wechsler Intelligence Scale for Children III (WISC-III) including vocabulary, similarities, block design and picture completion32,35 to find suitable matches. We matched the groups for age, sex, handedness,36 total IQ, performance IQ and verbal IQ.
Data acquisition and procedure
We acquired neuroimaging data on a 1.5 T Siemens Sonata scanner at the Donders Institute for Brain, Cognition and Behaviour in Nijmegen. For each participant, a T1-weighted whole-brain scan was collected (magnetization-prepared rapid acquisition with gradient echo [MPRAGE], TI 850 ms, repetition time [TR] 2250 ms, echo time [TE] 3.68 ms, flip angle 15°, field of view [FOV] 256 × 256 × 176 mm3, voxel size 1.0 × 1.0 × 1.0 mm3), as well as a set of whole-brain diffusion-weighted images (twice-refocused spin-echo echo-planar imaging [TRSE-EPI]; TR 10100 ms, TE 93 ms, diffusion directions 30, b-value 900 s/mm2, unweighted images 4, FOV 320 × 320 × 160 mm3, voxel size 2.5 × 2.5 × 2.5 mm3). The T1 image served as a high-resolution anatomic reference image for the DTI data and was used for assessing grey and white matter volume differences between participant groups. The participants were familiarized with the set-up and normal scanning procedures before the actual image acquisition by means of a rehearsal in a separate replica (dummy) scanner.
The set of diffusion images was first carefully corrected for imaging artifacts from head and cardiac motion using robust tensor estimation software developed in house.37 In short, this consisted of iteratively reweighted least-squares tensor estimation with Welsch-type weighting functions and dedicated image processing to robustly detect and eliminate cardiac and head motion artifacts. Subsequently, the images were realigned and eddy current–corrected by minimization of the diffusion tensor residual errors.38 The diffusion tensors and their derivative fractional anisotropy and mean diffusivity measures were then normally computed using linear regression as implemented in a diffusion toolbox (http://sourceforge.net/projects/spmtools) of SPM5 (Wellcome Department of Cognitive Neurology). Next, the DTI results were spatially normalized to the ICBM152 reference template. To this end, the participant’s anatomic image was coregistered to the set of diffusion images using SPM’s mutual information-based rigid-body transformation routine. We always visually inspected this coregistration step to ensure that it was not notably biased toward the magnetic susceptibility–induced geometric distortions in the diffusion-weighted images. Subsequently, we computed the spatial normalization parameters describing the nonlinear transformation of the anatomic image to the reference template (all using SPM5 functionality and standard settings) and applied them to the fractional anisotropy and mean diffusivity images. These normalized images were smoothed with an 8 × 8 × 8 mm3 full-width at half-maximum 3-dimensional (3-D) Gaussian kernel and used for voxel-wise comparisons across all participants.
For investigation of white and grey matter volume differences, VBM of the T1-weighted images was performed in SPM5 using a VBM5 toolbox (http://dbm.neuro.uni-jena.de). The computed white matter probability map39 was modulated with the Jacobian determinants of the normalization parameters to allow interpretation of the results in terms of volumetric differences. The modulated white matter partitions were smoothed with an 8 × 8 × 8 mm3 full-width at half-maximum 3-D Gaussian kernel. We performed the same analysis for grey matter. We computed total white and grey matter volume by summation of the probability maps, thresholded at 0.5.
Statistical analysis
To characterize the fractional anisotropy and mean diffusivity probability distributions in each participant for white and grey matter, we computed the raw and first central moments (i.e., the sample mean, the sample standard deviation [SD], the sample skewness and the sample kurtosis). We compared these moments at the group level using 2-sided 2-sample Student t tests once without and once with inclusion of age and total IQ as covariates. An α-level of 0.05 was used to denote a result statistically significant.
We performed regionally specific group analysis of the grey matter, white matter, fractional anisotropy and mean diffusivity measures using whole-brain voxel-wise 1-sided 2-sample Student t tests in SPM5 in both directions. Although the control group was closely matched, we performed the voxel-wise analyses once without and once with inclusion of age and total IQ as covariates. The fractional anisotropy analyses were explicitly restricted to the computed white matter mask, whereas the mean diffusivity analyses were restricted to a constructed whole-brain mask. Voxel-wise analyses were evaluated at a false discovery rate (FDR) of 0.05, so that for every reported voxel, the probability of true-positive discovery was 95%. The results were normalized to Z scores.
Results
Participants
We included 17 adolescents with autism and 25 controls in the study. All participants were between 12 and 18 years, and right-handed. Average ADI-R scores for the autism group were social 18.2 (standard deviation [SD] 4.9), verbal 13.4 (SD 4.6), nonverbal 8.5 (SD 3.2), stereotypy 4.1 (SD 2.7) and onset 2.2 (SD 1.2). None of the controls was within the CBCL or TRF clinical range. There were no significant differences in age, sex, handedness,36 total IQ, performance IQ and verbal IQ between the groups. In the autism group, nobody was on psychotropic medication, but 2 participants had previously used risperidone (Table 2).
Study participant demographics and general characteristics
Microstructural differences
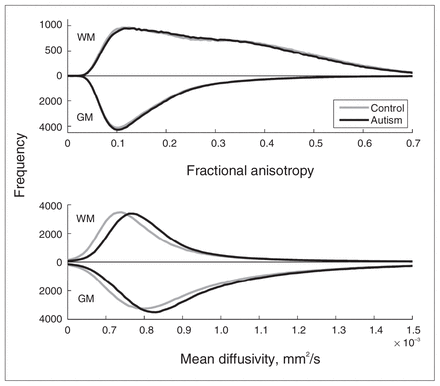
We first examined whether we could find evidence at the whole-brain level for pervasive differences in brain microstructure between the autism and control groups. To this end, we calculated the fractional anisotropy and mean diffusivity histograms for white and grey matter per participant and compared the group-averaged distributions (Fig. 1, upper panel). The figure shows virtually equal fractional anisotropy distributions for both groups in both grey and white matter. The white matter distributions have 2 maxima, 1 at the position of the grey matter maximum and 1 around the expected position. Two-sample Student t tests on the main moments of the distributions (Table 3) indicate that for fractional anisotropy there was no significant difference in the mean, SD and skewness between the autism and control groups, but that the kurtosis in white matter is larger in the autism group. This indicates that there are no prominent fractional anisotropy differences at the whole-brain level but that white matter fractional anisotropy is somewhat more outlier-prone in the autism group. The mean diffusivity distributions in white and grey matter (Fig. 1, lower panel), however, were both notably shifted toward greater mean diffusivity for the autism group, whereas the global shape of the distribution appeared to be unaffected. Two-sample Student t tests on the main moments of the distributions (Table 3) indicate that the average mean diffusivity was significantly larger in the autism group, but that there was no difference in the other moments (i.e., in the shape of the distribution). To confirm the validity of this finding, we examined whether this effect was similarly present in cerebral spinal fluid (CSF) by selecting voxels with mean diffusivity values above 1.5 ×10−3 mm2/s. We did not find a difference in average mean diffusivity for these voxels between the groups (p = 0.94).
Fractional anisotropy and mean diffusivity histograms. The figure shows the group-averaged histograms of the fractional anisotropy (upper panel; bin size 0.005) and mean diffusivity (lower panel; bin size 0.01 × 10−3 mm2/s) for grey and white matter. Results for grey matter are depicted downwards for clarity. Upper panel: As expected, fractional anisotropy in grey matter has the largest number of low fractional anisotropy values and peaks at around 0.1. (It deviates from its expectation value zero owing to acquisition noise.) The fractional anisotropy distribution of white matter is much broader, ranging up to values of 0.7. Note that there are 2 peaks, one around 0.1 and the other around 0.35. Lower panel: As expected, the distributions of mean diffusivity are moderately greater in grey matter than white matter. Note, however, that in the autism group the distribution of mean diffusivity is shifted toward higher values in both grey and white matter. Mean diffusivity values above 1.5 × 10−3 mm2/s are not depicted; they do not differ between groups and reflect cerebral spinal fluid outside the brain tissue. GM = grey matter; WM = white matter.
Main moments of the fractional anisotropy and mean diffusivity probability distributions
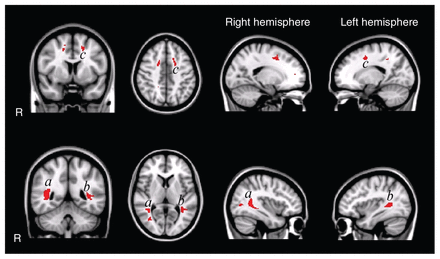
Second, we analyzed microstructural differences using voxel-wise comparisons to allow for fine grained regional inferences. In 3 confined regions, the fractional anisotropy was significantly decreased (Fig. 2) for the autism group compared with the control group. The largest region was located in the left superior and inferior longitudinal fasciculus on the border of the temporal and occipital lobe. In the right hemisphere, the superior and inferior longitudinal fasciculi were affected as well. In the frontal lobe, we found regions with decreased fractional anisotropy in the left and right corona radiata, but only the left was statistically significant. We found no regions with increased fractional anisotropy in the autism group compared with the control group. However, when we included age and total IQ as covariates, these 3 regions no longer showed significant differences between groups. This suggests that the controls’ greater fractional anisotropy values can, at least partially, be explained by the variance in age and IQ.
Voxel-wise fractional anisotropy group comparisons uncorrected for age and total intelligence quotient (IQ). The highlighted patches in the figure indicate regions with significantly decreased fractional anisotropy (p < 0.05, false discovery rate [FDR]–corrected) in white matter in the autism group (overlaid on the Montreal Neurological Institute coordinate 152 T1 brain). Fractional anisotropy is decreased in the left and right superior and inferior longitudinal fasciculus in temporal/occipital regions and in the left corona radiata in frontal regions. We found no regions with increased fractional anisotropy in the autism group. Of note: when corrected for age and total IQ, the group differences no longer reach significance at p < 0.05 FDR–corrected for multiple comparisons.
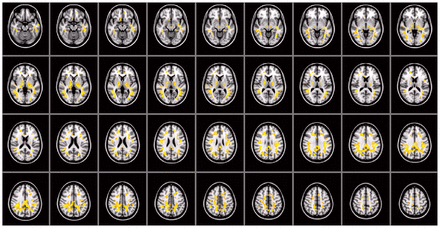
Compared with fractional anisotropy, differences in mean diffusivity were not limited to small regions, but rather were significantly increased in most of the brain (including frontal, temporal, parietal and occipital regions and the cerebellum) in the autism group compared with the control group, even after including age and total IQ as covariates (Fig. 3). The left and right anterior, superior and posterior corona radiata, and the anterior and posterior limb of the internal capsule, middle cerebellar peduncle, thalamus and thalamic radiations and inferior and superior longitudinal and fronto-occipital fasciculus showed an increased mean diffusivity. Parts of the genu, body and splenium of corpus callosum also showed a significant increase in mean diffusivity. We found no mean diffusivity decreases in the autism group. As for the histogram analyses, the mean diffusivity findings did not seem artifactual (e.g., because of more head motion during scanning), as no mean diffusivity difference was found in voxels containing CSF.
Voxel-wise mean diffusivity group comparisons corrected for age and total intelligence quotient. As indicated by the large highlighted areas, mean diffusivity is increased throughout the brain (overlaid on the Montreal Neurological Institute coordinate 152 T1 brain). We found no regions with decreased mean diffusivity in the autism group.
Volumetric differences and covariates
We found no differences in total white or grey matter volume. At the voxel level, the VBM analyses showed that there were no region-specific differences in grey or white matter volumetrics between groups.
Discussion
We found a generalized deficit in the integrity of white matter in adolescents with autism. More specifically, we found an increase in mean diffusivity throughout the white matter of the cerebrum and cerebellum, even after correcting for age and IQ. This is in line with and extends the findings of an overall mean diffusivity increase in the frontal lobe of people with autism25 and is also in line with mean diffusivity increase in the corpus callosum.23 We also found 3 small regions with decreased fractional anisotropy located in the left corona radiata and the left and right superior and inferior longitudinal fasciculus. However, these differences disappeared after correcting for age and, to lesser extent, IQ. This is in line with previous DTI studies reporting on regional differences in fractional anisotropy, as these have not employed whole-brain analyses that were corrected for multiple comparisons and for age and IQ. The only fractional anisotropy difference we did find was a higher kurtosis of the fractional anisotropy distribution in white matter for the autism group, indicating that fractional anisotropy values are slightly more extreme (outlier-prone) in people with autism. Contrary to our mean diffusivity findings, the significance of this result was much lower after adjustment for age and IQ. We therefore take note of this result but suspect that it may become insignificant with better matching or nonlinear adjustment for these covariates.
Combined with prior DTI studies on autism, our results indicate that reduced white matter integrity is among the most consistent findings of the neuroanatomy of autism.20,21,23 In the following paragraphs we discuss the implications of our fractional anisotropy and mean diffusivity findings.
Cerebral diffusivity
We found a mean diffusivity increase throughout the white matter of the cerebrum and cerebellum that extended into the grey matter. The mean diffusivity histogram (Fig. 1) shows that both the grey and white matter distributions were shifted in the autism group, but the difference in grey matter was only significant after adjustment for age and IQ (Table 3). The effect was greatest in the white matter, possibly with partial voluming effects driving part of the observed shift in grey matter mean diffusivity distribution. The mean diffusivity increase we found in our study is indicative of increased interstitial space, for example due to reduced neural or glial cell packing or cell size, or decreased water exchange rate between the intra- and extracellular compartments.28,40,41 We think it is less likely that membrane permeability is affected in people with autism and that mean diffusivity increases are more likely due to differences in cell size or number, but evidence as to which cells could be affected in people with autism is scarce. Among the white and grey matter brain cells are neurons that process and transmit information and glial cells that far outnumber neurons (about 9 to 1).42 The lack of pervasive concomitant fractional anisotropy abnormalities in white matter suggests that the mean diffusivity increases must be due to abnormalities in isotropic cells such as glial astrocytes. To date, empirical data have mainly been focused on neural abnormalities, but activation of neuroglia has been reported as well.43 Several postmortem studies have shown that neurons are abnormally large44 in certain structures of the brain in young autistic children (nucleus of the diagonal band of Broca in the limbic system). Neurons that are reduced in number and size have been found in the cerebellar nuclei and inferior olive in the brains of adults with autism.44
Cerebral volumetry
In our VBM analysis we found no region-specific differences in white or grey matter volume. Ke and colleagues22 reported deviations in white matter density and dotted spatial correspondence of these abnormalities with differences in fractional anisotropy. Their findings, however, did not match voxel-by-voxel and were based on a smaller sample (n = 12) that is more likely to suffer from lack of statistical power and adjustment for age and IQ. Moreover, a potentially important difference with our study is that their participants were on average 6 years younger. In 2 papers, Waiter and colleagues45,46 reported on abnormal regional grey and white matter densities in a well-matched and comparably large (n = 16) sample of patients with autism. The authors used separate segmentation and normalization algorithms rather than the more recent unified segmentation algorithm.39 Another difference is that they constructed custom templates from their sample of participants. We presume that the supposedly greater accuracy of the unified segmentation or perhaps a lack of power to construct custom templates from such a sample may well explain the different findings.
Cerebellum
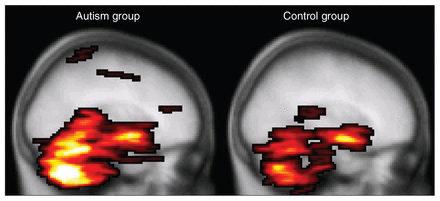
To date, only 1 DTI study involving people with autism has included the whole cerebellum.26 Although the authors found white matter integrity reductions as we did, they found fractional anisotropy decreases in combination with normal mean diffusivity in the superior cerebellar peduncles, whereas we found mean diffusivity increases in combination with normal fractional anisotropy in the midcerebellar peduncles. A possibly important difference between the studies is that in Catani and colleague’s study,26 adults with Asperger syndrome participated, whereas in our study adolescents with autism participated. Furthermore, findings in the cerebellum should be interpreted with caution, since the cerebellum is susceptible for scan artifacts owing to cardiac pulsation. We controlled for this by using a robust tensor estimation technique in which artifacts in the DTI images were robustly discarded from the analysis.37 Catani and colleagues26 have controlled for this by gating the data acquisition to the cardiac cycle. Both methods increase confidence that the measured differences indeed reflect differences in white matter integrity and not stress level and heart rate during data acquisition, for example. It should be noted, however, that cardiac gating is certainly not a perfect remedy and that in our study, cerebellar fractional anisotropy values were also found to be regionally decreased in the autism group when this preprocessing correction was not applied (data not shown). Moreover, our preprocessing method detected a larger number of DTI artifacts in the autism group (Fig. 4), suggesting that a difference in cardiac activity is likely and might well be implicated and providing false-positive results in the cerebellar area. We speculate that such greater cardiac activity may be due to greater stress susceptibility in the autism group for the experimental procedures and MRI acquisition.
Mean number of diffusion tensor imaging (DTI) artifacts. The highlighted areas denote the maximum intensity projection of the group averaged voxel-wise number of cardiac artifacts per participant (i.e., 34 DTI volumes) in the controls (right) versus participants with autism (left) overlaid on the Montreal Neurological Institute coordinate 152 T1 brain. It is apparent from this figure, as well as from the average total number of affected voxels per participant (mean 562, standard deviation [SD] 468 v. mean 1265, SD 1249, respectively; p = 0.010, 2-sided Student t test) that the DTI images from the autism group contain more cardiac pulsation artifacts than the control group.
Limitations
The nonlinear transformations from the unified segmentation algorithm were used in our VBM analysis as well as to transform our DTI data to a common space. Furthermore, we used rigid body transformations to coregister the (susceptibility distorted) DTI images to the corresponding T1 image. It is hence a risk that inaccuracies in these transformations may result in spurious differences in fractional anisotropy or mean diffusivity values. However, as we found no differences in overall brain volume (white matter and grey matter), or regionally specific differences between the groups, we believe this is not a concern in the current study. This does not imply that our nonlinear transformations were free of inaccuracies. On the contrary, we believe that the presence of a maximum in our white matter fractional anisotropy distributions around the position of the grey matter maximum can be (at least partly) explained by and is indicative for the reality of such inaccuracies. The presence of this maximum is most likely also due to partial voluming, failure of the tensor model, as well as geometric mismatch with the white matter mask due to magnetic susceptibility–induced distortions of the DTI images.
Developmental stage was an important factor to take into account when studying fractional anisotropy and mean diffusivity differences between our autism and control groups. We corrected for age and IQ using a linear model, but it may well be that a more advanced (nonlinear) account is more powerful. Also, our study would have benefited from using a larger sample, greater age range and a longitudinal design.
Conclusion
Diffusion tensor imaging studies in patients with autism published so far have consistently found decreased microstructural integrity in white matter in adolescents and adults with autism. Importantly, our data indicate that, first, the microstructural integrity is not region-specific but reduced throughout grey and white matter and, second, this microstructural deficit is isotropic and has no volumetric complement, suggesting that either the number or the size of neuroglial cells is reduced in people with autism. It may thus be especially worthwhile for future diffusion imaging studies to turn to more advanced diffusion MRI methods, such as q-space imaging, to elucidate the nature of the microstructural abnormalities and hopefully shed new light on the neurobiological causes of autism.
Footnotes
Competing interests: None declared by Drs. Groen, van der Gaag and Zwiers. Dr. Buitelaar reported being a board member and paid consultant of Janssen Cilag BV and Eli Lilly, receiving a grant from Eli Lilly and being paid for the development of educational presentations by Eli Lilly, Janssen Cilag and Medice.
Contributors: All authors helped design the article, analyzed the data, wrote and reviewed the article and approved its publication. Dr. Groen acquired the data.
- Received August 20, 2009.
- Revision received April 23, 2010.
- Revision received June 15, 2010.
- Accepted June 16, 2010.