Abstract
Background: Patients have traditionally been considered candidates for sentinel node biopsy (SNBx) only at the time of wide local excision (WLE). We hypothesized that patients with prior WLE may also be staged accurately with SNBx.
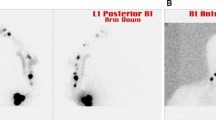
Methods: Seventy-six patients, including 18 patients from the University of Virginia and 58 from a multicenter study of SNBx led by investigators at the University of Vermont, who had previous WLE for clinically localized melanoma underwent lymphoscintigraphy with SNBx. Median follow-up time was 38 months.
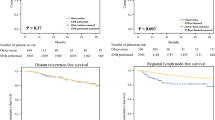
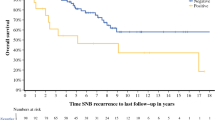
Results: Intraoperative identification of at least 1 sentinel node was accomplished in 75 patients (98.6%). The mean number of sentinel nodes removed per patient was 2.0. Eleven patients (15%) had positive sentinel nodes. Among the 64 patients with negative SNBx, 3 (4%) developed nodal recurrences in a sentinel node–negative basin simultaneous with systemic metastasis, and 1 (1%) developed an isolated first recurrence in a lymph node.
Conclusions:This multicenter study more than doubles the published experience with SNBx after WLE and provides much-needed outcome data on recurrence after SNBx in these patients. These outcomes compare favorably with the reported literature for patients with SNBx at the time of WLE, suggesting that accurate staging of the regional lymph node bed is possible in patients after WLE.
Similar content being viewed by others
REFERENCES
Morton DL, Thompson JF, Essner R, et al. Validation of the accuracy of intraoperative lymphatic mapping and sentinel lymph-adenectomy for early-stage melanoma: a multicenter trial. Multi-center Selective Lymphadenectomy Trial Group. Ann Surg 1999;230:453–63; discussion 463–5.
Morton DL. Sentinel lymphadenectomy for patients with clinical stage I melanoma. J Surg Oncol 1997;66:267–9.
Sim FH, Taylor WF, Pritchard DJ, Soule EH. Lymphadenectomy in the management of stage I malignant melanoma: a prospective randomized study. Mayo Clin Proc 1986;61:697–705.
Veronesi U, Adamus J, Bandiera DC, et al. Delayed regional lymph node dissection in stage I melanoma of the skin of the lower extremities. Cancer 1982;49:2420–30.
Balch CM, Soong SJ, Bartolucci AA, et al. Efficacy of an elective regional lymph node dissection of 1 to 4 mm thick melanomas for patients 60 years of age and younger. Ann Surg 1996;224:255–63;discussion 263–6.
Slingluff CL Jr, Stidham KR, Ricci WM, et al. Surgical management of regional lymph nodes in patients with melanoma. Experience with 4682 patients. Ann Surg 1994;219:120–30.
Krag DN, Meijer SJ, Weaver DL, et al. Minimal-access surgery for staging of malignant melanoma. Arch Surg 1995;130:654–8; discussion 659–60.
Glass LF, Messina JL, Cruse W, et al. The use of intraoperative radiolymphoscintigraphy for sentinel node biopsy in patients with malignant melanoma. Dermatol Surg 1996;22:715–20.
Essner R. The role of lymphoscintigraphy and sentinel node mapping in assessing patient risk in melanoma. Semin Oncol 1997;24(1Suppl 4):S8–10.
Kelley MC, Ollila DW, Morton DL. Lymphatic mapping and sentinel lymphadenectomy for melanoma. Semin Surg Oncol 1998;14:283–90.
Balch CM, Lange JR. Lymphatic mapping and sentinel node lymphadenectomy for cancer: an overview. Ann Surg Oncol 2001;8(9 Suppl):1S–4S.
Kelemen PR, Essner R, Foshag LJ, Morton DL. Lymphatic mapping and sentinel lymphadenectomy after wide local excision of primary melanoma. J Am Coll Surg 1999;189:247–52.
Harlow SP, Krag DN, Ashikaga T, et al. Gamma probe guided biopsy of the sentinel node in malignant melanoma: a multicentre study. Melanoma Res 2001;11:45–55.
Reintgen DS, Brobeil A. Lymphatic mapping and selective lymphadenectomy as an alternative to elective lymph node dissection in patients with malignant melanoma. Hematol Oncol Clin North Am 1998;12:807–21, vii.
Wanebo HJ, Harpole D, Teates CD. Radionuclide lymphoscintigraphy with technetium 99m antimony sulfide colloid to identify lymphatic drainage of cutaneous melanoma at ambiguous sites in the head and neck and trunk. Cancer 1985;55:1403–13.
Krag DN. Minimal access surgery for staging regional lymph nodes: the sentinel-node concept. Curr Probl Surg 1998;35:951–1016.
Friedman HD, Tatum AH. HMB-45-positive malignant lymphoma. A case report with literature review of aberrant HMB-45 reactivity. Arch Pathol Lab Med 1991;115:826–30.
Karakousis CP, Grigoropoulos P. Sentinel node biopsy before and after wide excision of the primary melanoma. Ann Surg Oncol 1999;6:785–9.
Morton DL, Wen DR, Wong JH, et al. Technical details of intraoperative lymphatic mapping for early stage melanoma. Arch Surg 1992;127:392–9.
Gershenwald JE, Colome MI, Lee JE, et al. Patterns of recurrence following a negative sentinel lymph node biopsy in 243 patients with stage I or II melanoma. J Clin Oncol 1998;16:2253–60.
Jansen L, Nieweg OE, Peterse JL, et al. Reliability of sentinel lymph node biopsy for staging melanoma. Br J Surg 2000;87:484–9.
Leong SP, Steinmetz I, Habib FA, et al. Optimal selective sentinel lymph node dissection in primary malignant melanoma. Arch Surg 1997;132:666–72; discussion 673.
Gadd MA, Cosimi AB, Yu J, et al. Outcome of patients with melanoma and histologically negative sentinel lymph nodes. Arch Surg 1999;134:381–7.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Evans, H.L., Krag, D.N., Teates, C.D. et al. Lymphoscintigraphy and Sentinel Node Biopsy Accurately Stage Melanoma in Patients Presenting After Wide Local Excision. Ann Surg Oncol 10, 416–425 (2003). https://doi.org/10.1245/ASO.2003.05.009
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1245/ASO.2003.05.009