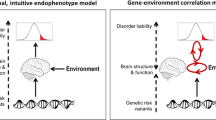
Abstract
We interrogate the joint genetic architecture of 11 major psychiatric disorders at biobehavioral, functional genomic and molecular genetic levels of analysis. We identify four broad factors (neurodevelopmental, compulsive, psychotic and internalizing) that underlie genetic correlations among the disorders and test whether these factors adequately explain their genetic correlations with biobehavioral traits. We introduce stratified genomic structural equation modeling, which we use to identify gene sets that disproportionately contribute to genetic risk sharing. This includes protein-truncating variant-intolerant genes expressed in excitatory and GABAergic brain cells that are enriched for genetic overlap across disorders with psychotic features. Multivariate association analyses detect 152 (20 new) independent loci that act on the individual factors and identify nine loci that act heterogeneously across disorders within a factor. Despite moderate-to-high genetic correlations across all 11 disorders, we find little utility of a single dimension of genetic risk across psychiatric disorders either at the level of biobehavioral correlates or at the level of individual variants.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are all publicly available or can be requested for access. Specific download links for various datasets are directly below.
Summary statistics for data from the PGC can be downloaded or requested here:
https://www.med.unc.edu/pgc/download-results/.
Summary statistics for the anxiety phenotype in UKB (TotANX_OR) can be downloaded here:
https://drive.google.com/drive/folders/1fguHvz7l2G45sbMI9h_veQun4aXNTy1v.
23andMe summary statistics are made available through 23andMe to qualified researchers under an agreement with 23andMe that protects the privacy of 23andMe participants. Please visit research.23andme.com/collaborate/#publication for more information.
Summary statistics for the volume-based neuroimaging phenotypes were downloaded from https://github.com/BIG-S2/GWAS.
Summary statistics for the health and well-being complex trait correlations can be downloaded from https://atlas.ctglab.nl/.
Summary statistics for the circadian rhythm correlations across 24 hours can be downloaded from https://cnsgenomics.com/software/gcta/#DataResource.
Data from gnomAD used to identify PI genes for creation of annotations can be downloaded here: https://storage.googleapis.com/gnomad-public/release/2.1.1/constraint/gnomad.v2.1.1.lof_metrics.by_gene.txt.bgz.
Gene count data per cell for creation of annotations were obtained from https://storage.googleapis.com/gtex_additional_datasets/single_cell_data/GTEx_droncseq_hip_pcf.tar.
Data that map individual cells to cell types (e.g., neuron, astrocyte) were obtained from https://static-content.springer.com/esm/art%3A10.1038%2Fnmeth.4407/MediaObjects/41592_2017_BFnmeth4407_MOESM10_ESM.xlsx.
Links to the LD scores, reference panel data and the code used to produce the current results can all be found at https://github.com/MichelNivard/GenomicSEM/wiki.
Links to the BaselineLD v2.2 annotations can be found here: https://data.broadinstitute.org/alkesgroup/LDSCORE/.
Code availability
Genomic SEM software (which now includes the stratified genomic SEM extension) is an R package that is available from GitHub at the following URL: https://github.com/GenomicSEM/GenomicSEM.
Directions for installing the genomic SEM R package can be found at https://github.com/GenomicSEM/GenomicSEM/wiki.
References
Martel, M. M. et al. A general psychopathology factor (P factor) in children: structural model analysis and external validation through familial risk and child global executive function. J. Abnorm. Psychol. 126, 137–148 (2017).
Dean, K. et al. The impact of parental mental illness across the full diagnostic spectrum on externalising and internalising vulnerabilities in young offspring. Psychol. Med. 48, 2257–2263 (2018).
McLaughlin, K. A. et al. Parent psychopathology and offspring mental disorders: results from the WHO World Mental Health Surveys. Br. J. Psychiatry 200, 290–299 (2012).
Kessler, R. C., Chiu, W. T., Demler, O. & Walters, E. E. Prevalence, severity, and comorbidity of 12-month DSM-IV disorders in the National Comorbidity Survey Replication. Arch. Gen. Psychiatry 62, 617–627 (2005).
Caspi, A. et al. The p factor: one general psychopathology factor in the structure of psychiatric disorders? Clin. Psychol. Sci. 2, 119–137 (2014).
Lahey, B. B. et al. Is there a general factor of prevalent psychopathology during adulthood? J. Abnorm. Psychol. 121, 971–977 (2012).
Pettersson, E., Larsson, H. & Lichtenstein, P. Common psychiatric disorders share the same genetic origin: a multivariate sibling study of the Swedish population. Mol. Psychiatry 21, 717–721 (2016).
Bulik-Sullivan, B. K. et al. LD Score regression distinguishes confounding from polygenicity in genome-wide association studies. Nat. Genet. 47, 291–295 (2015).
Selzam, S., Coleman, J. R., Caspi, A., Moffitt, T. E. & Plomin, R. A polygenic p factor for major psychiatric disorders. Transl. Psychiatry 8, 205 (2018).
Lee, S. H. et al. Genetic relationship between five psychiatric disorders estimated from genome-wide SNPs. Nat. Genet. 45, 984–994 (2013).
Anttila, V. et al. Analysis of shared heritability in common disorders of the brain. Science 360, eaap8757 (2018).
Lee, P. H. et al. Genomic relationships, novel loci, and pleiotropic mechanisms across eight psychiatric disorders. Cell 179, 1469–1482.e11 (2019).
Grotzinger, A. D. et al. Genomic structural equation modelling provides insights into the multivariate genetic architecture of complex traits. Nat. Hum. Behav. 3, 513–525 (2019).
Demontis, D. et al. Discovery of the first genome-wide significant risk loci for attention deficit/hyperactivity disorder. Nat. Genet. 51, 63–75 (2019).
Walters, R. K. et al. Transancestral GWAS of alcohol dependence reveals common genetic underpinnings with psychiatric disorders. Nat. Neurosci. 21, 1656–1669 (2018).
Watson, H. J. et al. Genome-wide association study identifies eight risk loci and implicates metabo-psychiatric origins for anorexia nervosa. Nat. Genet. 51, 1207–1214 (2019).
Grove, J. et al. Identification of common genetic risk variants for autism spectrum disorder. Nat. Genet. 51, 431–444 (2019).
Otowa, T. et al. Meta-analysis of genome-wide association studies of anxiety disorders. Mol. Psychiatry 21, 1391–1399 (2016).
Purves, K. L. et al. A major role for common genetic variation in anxiety disorders. Mol. Psychiatry 25, 3292–3303 (2020).
Stahl, E. A. et al. Genome-wide association study identifies 30 loci associated with bipolar disorder. Nat. Genet. 51, 793–803 (2019).
Wray, N. R. et al. Genome-wide association analyses identify 44 risk variants and refine the genetic architecture of major depression. Nat. Genet. 50, 668–681 (2018).
Howard, D. M. et al. Genome-wide association study of depression phenotypes in UK Biobank identifies variants in excitatory synaptic pathways. Nat. Commun. 9, 1470 (2018).
International Obsessive Compulsive Disorder Foundation Genetic Collaborative (IOCDF-GC) and OCD Collaborative Genetics Association Studies (OCGAS). Revealing the complex genetic architecture of obsessive–compulsive disorder using meta-analysis. Mol. Psychiatry 23, 1181–1188 (2018).
Meier, S. M. et al. Genetic variants associated with anxiety and stress-related disorders: a genome-wide association study and mouse-model study. JAMA Psychiatry 76, 924–932 (2019).
Duncan, L. E. et al. Largest GWAS of PTSD (N = 20 070) yields genetic overlap with schizophrenia and sex differences in heritability. Mol. Psychiatry 23, 666–673 (2018).
Trubetskoy, V. et al. Mapping genomic loci implicates genes and synaptic biology in schizophrenia. Nature 604, 502–508 (2022).
Yu, D. et al. Interrogating the genetic determinants of Tourette’s syndrome and other tic disorders through genome-wide association studies. Am. J. Psychiatry 176, 217–227 (2019).
Watanabe, K. et al. A global overview of pleiotropy and genetic architecture in complex traits. Nat. Genet. 51, 1339–1348 (2019).
Zhao, B. et al. Genome-wide association analysis of 19,629 individuals identifies novel genetic variants for regional brain volumes and refines their genetic co-architecture with cognitive and mental health traits. Nat. Genet. 51, 1637–1644 (2019).
Jiang, L. et al. A resource-efficient tool for mixed model association analysis of large-scale data. Nat. Genet. 51, 1749–1755 (2019).
Karatsoreos, I. N. Links between circadian rhythms and psychiatric disease. Front. Behav. Neurosci. 8, 162 (2014).
Finucane, H. K. et al. Partitioning heritability by functional annotation using genome-wide association summary statistics. Nat. Genet. 47, 1228–1235 (2015).
Karczewski, K. J. et al. The mutational constraint spectrum quantified from variation in 141,456 humans. Nature 581, 434–443 (2019).
Habib, N. et al. Massively parallel single-nucleus RNA-seq with DroNc-seq. Nat. Methods 14, 955–958 (2017).
Finucane, H. K. et al. Heritability enrichment of specifically expressed genes identifies disease-relevant tissues and cell types. Nat. Genet. 50, 621–629 (2018).
Turley, P. et al. Multi-trait analysis of genome-wide association summary statistics using MTAG. Nat. Genet. 50, 229–237 (2018).
Baselmans, B. M. L. et al. Multivariate genome-wide analyses of the well-being spectrum. Nat. Genet. 51, 445–451 (2019).
Pe’er, I., Yelensky, R., Altshuler, D. & Daly, M. J. Estimation of the multiple testing burden for genomewide association studies of nearly all common variants. Genet. Epidemiol. 32, 381–385 (2008).
Kranzler, H. R. et al. Genome-wide association study of alcohol consumption and use disorder in 274,424 individuals from multiple populations. Nat. Commun. 10, 1499 (2019).
Epskamp, S., Rhemtulla, M. & Borsboom, D. Generalized network psychometrics: combining network and latent variable models. Psychometrika 82, 904–927 (2017).
Borsboom, D. A network theory of mental disorders. World Psychiatry 16, 5–13 (2017).
Liu, L., Schulz, S. C., Lee, S., Reutiman, T. J. & Fatemi, S. H. Hippocampal CA1 pyramidal cell size is reduced in bipolar disorder. Cell. Mol. Neurobiol. 27, 351–358 (2007).
Ho, N. F. et al. Progressive decline in hippocampal CA1 volume in individuals at ultra-high-risk for psychosis who do not remit: findings from the Longitudinal Youth at Risk Study. Neuropsychopharmacology 42, 1361–1370 (2017).
Konradi, C. et al. Hippocampal interneurons in bipolar disorder. Arch. Gen. Psychiatry 68, 340–350 (2011).
Li, W. et al. Synaptic proteins in the hippocampus indicative of increased neuronal activity in CA3 in schizophrenia. Am. J. Psychiatry 172, 373–382 (2015).
Volk, D. W., Sampson, A. R., Zhang, Y., Edelson, J. R. & Lewis, D. A. Cortical GABA markers identify a molecular subtype of psychotic and bipolar disorders. Psychol. Med. 46, 2501–2512 (2016).
de Jonge, J. C., Vinkers, C. H., Hulshoff Pol, H. E. & Marsman, A. GABAergic mechanisms in schizophrenia: linking postmortem and in vivo studies. Front. Psychiatry 8, 118 (2017).
Yun, S. et al. Stimulation of entorhinal cortex–dentate gyrus circuitry is antidepressive. Nat. Med. 24, 658–666 (2018).
Boldrini, M. et al. Antidepressants increase neural progenitor cells in the human hippocampus. Neuropsychopharmacology 34, 2376–2389 (2009).
Shi, H. et al. Population-specific causal disease effect sizes in functionally important regions impacted by selection. Nat. Commun. 12, 1098 (2021).
Cai, N. et al. Minimal phenotyping yields genome-wide association signals of low specificity for major depression. Nat. Genet. 52, 437–447 (2020).
Newson, J. J., Hunter, D. & Thiagarajan, T. C. The heterogeneity of mental health assessment. Front. Psychiatry 11, 76 (2020).
Yengo, L., Yang, J. & Visscher, P. M. Expectation of the intercept from bivariate LD score regression in the presence of population stratification. Preprint at bioRxiv https://www.biorxiv.org/content/10.1101/310565v1 (2018).
Yang, J., Lee, S. H., Goddard, M. E. & Visscher, P. M. GCTA: a tool for genome-wide complex trait analysis. Am. J. Hum. Genet. 88, 76–82 (2011).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Meredith, W. Measurement invariance, factor analysis and factorial invariance. Psychometrika 58, 525–543 (1993).
Hujoel, M. L., Gazal, S., Hormozdiari, F., van de Geijn, B. & Price, A. L. Disease heritability enrichment of regulatory elements is concentrated in elements with ancient sequence age and conserved function across species. Am. J. Hum. Genet. 104, 611–624 (2019).
Kundaje, A. et al. Integrative analysis of 111 reference human epigenomes. Nature 518, 317–330 (2015).
GTEx Consortium. The Genotype-Tissue Expression (GTEx) pilot analysis: multitissue gene regulation in humans. Science 348, 648–660 (2015).
Pers, T. H. et al. Biological interpretation of genome-wide association studies using predicted gene functions. Nat. Commun. 6, 5890 (2015).
Acknowledgements
This work presented here would not have been possible without the enormous efforts put forth by the investigators and participants from Psychiatric Genetics Consortium, iPSYCH, UK Biobank and 23andMe. The work from these contributing groups was supported by numerous grants from governmental and charitable bodies as well as philanthropic donation. Research reported in this publication was supported by the National Institute of Mental Health of the National Institutes of Health (NIH) under Award Number R01MH120219. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. A.D.G. was additionally supported by NIH grant R01HD083613. E.M.T.-D. was additionally supported by NIH grants R01AG054628 and R01HD083613 and the Jacobs Foundation. E.M.T.-D. is a faculty associate of the Population Research Center at the University of Texas, which is supported by NIH grant P2CHD042849, and the Center on Aging and Population Sciences, which is supported by NIH grant P30AG066614. M.G.N. is additionally supported by ZonMW grants 849200011 and 531003014 from The Netherlands Organisation for Health Research and Development and a VENI grant awarded by NWO (VI.Veni.191 G.030) and is a Jacobs Foundation Fellow. W.A.A. is supported by the European Union’s Horizon 2020 Research and Innovation Programme, Marie Sklodowska Curie Actions – MSCA-ITN-2016 – Innovative Training Networks under grant agreement 721567. H.F.I. is supported by the Aggression in Children: unraveling gene-environment interplay to inform Treatment and InterventiON strategies (ACTION) project. ACTION receives funding from the European Union Seventh Framework Program (FP7/2007-2013) under grant agreement 602768. C.M.L. is supported by the National Institute for Health Research Biomedical Research Centre at South London and Maudsley NHS Foundation Trust and King’s College London. A.M.M. is supported by the Wellcome Trust (104036/Z/14/Z, 216767/Z/19/Z), UKRI MRC (MC_PC_17209, MR/S035818/1). K.-P.L. is supported by the Deutsche Forschungsgemeinschaft (DFG: CRU 125, CRC TRR 58 A1/A5, No. 44541416), the European Union’s Seventh Framework Programme under grant 602805 (Aggressotype), the Horizon 2020 Research and Innovation Programme under grant 728018 (Eat2beNICE) and 643051 (MiND), Fritz Thyssen Foundation (10.13.1185), ERA-Net NEURON/RESPOND 01EW1602B, ERA-Net NEURON/DECODE FKZ01EW1902 and 5-100 Russian Academic Excellence Project. G.B. is supported by the National Institute for Health Research Biomedical Research Centre at South London and Maudsley NHS Foundation Trust and King’s College London. P.H.L. is supported by NIH grants R01MH119243 and R00MH101367. The iPSYCH team was supported by grants from the Lundbeck Foundation (R102-A9118, R155-2014-1724 and R248-2017-2003), the EU FP7 Program (grant 602805, ‘Aggressotype’) and H2020 Program (grant 667302, ‘CoCA’), NIMH (1U01MH109514-01 to ADB) and the universities and university hospitals of Aarhus and Copenhagen. The Danish National Biobank resource was supported by the Novo Nordisk Foundation. High-performance computer capacity for handling and statistical analysis of iPSYCH data on the GenomeDK HPC facility was provided by the Center for Genomics and Personalized Medicine and the Centre for Integrative Sequencing (Aarhus University, Denmark) (grant to A.D.B.).
Author information
Authors and Affiliations
Consortia
Contributions
A.D.G., M.G.N. and E.M.T.-D. designed the study. A.D.G., M.G.N. and E.M.T.-D. developed the methods used in this study. A.D.G., H.F.I., M.G.N. and E.M.T.-D. developed the software used in this study. A.D.G., M.G.N. and E,M.T.-D. performed the simulation studies. W.A.A., A.D.G. and M.G.N. created gene sets and annotations. A.D.G., T.T.M., M.G.N. and E.M.T.-D. contributed to genetic factor modeling, multivariate GWASs, complex trait correlations and multivariate enrichment analyses. A.D.G., M.G.N. and E.M.T.-D. wrote the manuscript, and A.D.G., T.T.M., W.A.A., H.F.I., M.J.A., C.M.L., A.M.M., J.G., S.D., K.-P.L., N.S., S.M.M., M.M., A.D.B., O.M., G.B., P.H.L., K.S.K., J.W.S., E.M.T.-D. and M.G.N. provided manuscript feedback and editing.
Corresponding author
Ethics declarations
Competing interests
J.W.S. is an unpaid member of the Bipolar/Depression Research Community Advisory Panel of 23andMe. C.M.L. is on the SAB for Myriad Neuroscience. G.B. is a scientific advisor for COMPASS Pathways. The other authors declare no competing interests.
Peer review
Peer review information
Nature Genetics thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Note and Figures 1–45.
Supplementary Table 1
Supplementary Tables 1-55
Rights and permissions
About this article
Cite this article
Grotzinger, A.D., Mallard, T.T., Akingbuwa, W.A. et al. Genetic architecture of 11 major psychiatric disorders at biobehavioral, functional genomic and molecular genetic levels of analysis. Nat Genet 54, 548–559 (2022). https://doi.org/10.1038/s41588-022-01057-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41588-022-01057-4
This article is cited by
-
Interpregnancy interval and early infant neurodevelopment: the role of maternal–fetal glucose metabolism
BMC Medicine (2024)
-
The structure of psychiatric comorbidity without selection and assortative mating
Translational Psychiatry (2024)
-
Connecting clinical and genetic heterogeneity in ADHD
Nature Genetics (2024)
-
Psychosis superspectrum I: Nosology, etiology, and lifespan development
Molecular Psychiatry (2024)
-
Beyond the factor indeterminacy problem using genome-wide association data
Nature Human Behaviour (2024)