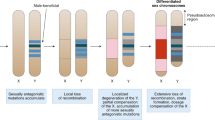
Key Points
-
Sexual dimorphism in gene expression is widespread across organisms and genomes.
-
Genes with sex-biased expression, especially those with male-biased expression, tend to evolve rapidly in both protein sequence and expression level.
-
Sex-biased genes are non-randomly distributed in the genome, with examples of both under-representation and over-representation on the X chromosome.
-
There is mounting evidence that positive selection is the driving force behind the rapid evolution of sex-biased genes. This is probably caused by sexual selection and antagonistic coevolution between the sexes.
-
Sex-linked genes that escape dosage compensation constitute a special case of sex-biased gene expression.
-
There are several scenarios for the origin of sex-biased genes, including single-locus antagonism, sexual antagonism plus gene duplication and duplication of sex-biased genes.
Abstract
Differences between males and females in the optimal phenotype that is favoured by selection can be resolved by the evolution of differential gene expression in the two sexes. Microarray experiments have shown that such sex-biased gene expression is widespread across organisms and genomes. Sex-biased genes show unusually rapid sequence evolution, are often labile in their pattern of expression, and are non-randomly distributed in the genome. Here we discuss the characteristics and expression of sex-biased genes, and the selective forces that shape this previously unappreciated source of phenotypic diversity. Sex-biased gene expression has implications beyond just evolutionary biology, including for medical genetics.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Connallon, T. & Knowles, L. L. Intergenomic conflict revealed by patterns of sex-biased gene expression. Trends Genet. 21, 495–499 (2005).
Rinn, J. L. & Snyder, M. Sexual dimorphism in mammalian gene expression. Trends Genet. 21, 298–305 (2005).
Arnqvist, G. & Rowe, L. Sexual Conflict (Princeton Univ. Press, Princeton, 2005).
Rice, W. R. Sexually antagonistic male adaptation triggered by experimental arrest of female evolution. Nature 381, 232–234 (1996).
Rice, W. R. Male fitness increases when females are eliminated from gene pool: implications for the Y chromosome. Proc. Natl Acad. Sci. USA 95, 6217–6221 (1998).
Chippindale, A. K., Gibson, J. R. & Rice, W. R. Negative genetic correlation for adult fitness between sexes reveals ontogenetic conflict in Drosophila. Proc. Natl Acad. Sci. USA 98, 1671–1675 (2001).
Ranz, J. M., Castillo-Davis, C. I., Meiklejohn, C. D. & Hartl, D. L. Sex-dependent gene expression and evolution of the D. melanogaster transcriptome. Science 300, 1742–1744 (2003). The authors compare male and female gene expression between two closely related Drosophila species, and find that the degree of sex bias often differs between species.
Parisi, M. et al. Paucity of genes on the D. melanogaster X chromosome showing male-biased expression. Science 299, 697–700 (2003). Microarray experiments reveal that male-biased genes with both somatic and germline expression are under-represented on the D. melanogaster X chromosome.
Yang, X. et al. Tissue-specific expression and regulation of sexually dimorphic genes in mice. Genome Res. 16, 995–1004 (2006). This reference represents an unparalleled example of microarray-based analysis of sex-biased gene expression, using a large number of replicates.
Thoemle, K. et al. Genome-wide analysis of sex-enriched gene expression during C. elegans larval development. Dev. Biol. 284, 500–508 (2005).
Zhang, Z., Hambuch, T. M. & Parsch, J. Molecular evolution of sex-biased genes in Drosophila. Mol. Biol. Evol. 21, 2130–2139 (2004).
Richards, S. et al. Comparative genome sequencing of Drosophila pseudoobscura: chromosomal, gene, and cis-element evolution. Genome Res. 15, 1–18 (2005).
Metta, M., Gudavalli, R., Gibert, J. M. & Schlötterer, C. No accelerated rate of protein evolution in male-biased Drosophila pseudoobscura genes. Genetics 174, 411–420 (2006).
Reinke, V., Gil, I. S., Ward, S. & Kazmer, K. Genome-wide germline-enriched and sex-biased expression profiles in Caenorhabditis elegans. Development 131, 311–323 (2004).
Cutter, A. D. & Ward, S. Sexual and temporal dynamics of molecular evolution in C. elegans development. Mol. Biol. Evol. 22, 178–188 (2005).
Altschul, S. F. et al. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res. 25, 3389–3402 (1997).
Torgerson, D. G., Kulathinal, R. J. & Singh, R. S. Mammalian sperm proteins are rapidly evolving: evidence of positive selection in functionally diverse genes. Mol. Biol. Evol. 19, 1973–1980 (2002).
Schultz, N., Hamra, F. K. & Garbers, D. L. A multitude of genes expressed solely in meiotic or postmeiotic spermatogenic cells offers a myriad of contraceptive targets. Proc. Natl Acad. Sci. USA 100, 12201–12206 (2003).
Good, J. M. & Nachman, M. W. Rates of protein evolution are positively correlated with developmental timing of expression during mouse spermatogenesis. Mol. Biol. Evol. 22, 1044–1052 (2005).
Khaitovich, P. et al. Parallel patterns of evolution in the genomes and transcriptomes of humans and chimpanzees. Science 309, 1850–1854 (2005). In a comparative genomic study of humans and chimpanzees, the authors find that testis-expressed genes have diverged rapidly in both protein sequence and expression level.
Zhang, Z. & Parsch, J. Positive correlation between evolutionary rate and recombination rate in Drosophila genes with male-biased expression. Mol. Biol. Evol. 22, 1945–1947 (2005).
Pröschel, M., Zhang, Z. & Parsch, J. Widespread adaptive evolution of Drosophila genes with sex-biased expression. Genetics 174, 893–900 (2006). DNA polymorphism and divergence data reveal that sex-biased genes undergo frequent adaptive evolution in Drosophila species.
Sawyer, S. A., Parsch, J., Zhang, Z. & Hartl, D. L. Prevalence of positive selection among nearly neutral amino acid replacements in Drosophila. Proc. Natl Acad. Sci. USA 104, 6504–6510 (2007).
Nielsen, R. et al. A scan for positively selected genes in the genomes of humans and chimpanzees. PLoS Biol. 3, e170 (2005).
Dorus, S. et al. Genomic and functional evolution of the Drosophila melanogaster sperm proteome. Nature Genet. 38, 1440–1445 (2006).
Swanson, W. J., Wong, A., Wolfner, M. F. & Aquadro, C. F. Evolutionary expressed sequence tag analysis of Drosophila female reproductive tracts identifies genes subjected to positive selection. Genetics 168, 1457–1465 (2004).
Panhuis, T. M. & Swanson, W. J. Molecular evolution and population genetic analysis of candidate female reproductive genes in Drosophila. Genetics 173, 2039–2047 (2006).
Swanson, W. J. & Vacquier, V. D. The rapid evolution of reproductive proteins. Nature Rev. Genet. 3, 137–144 (2002).
Rice, W. R. & Holland, B. The enemies within: intergenomic conflict, interlocus contest evolution (ICE), and the intraspecific Red Queen. Behav. Ecol. Sociobiol. 41, 1–10 (1997).
Gavrilets, S. Rapid evolution of reproductive barriers driven by sexual conflict. Nature 403, 886–889 (2000).
Stepinska, U. & Bakst, M. R. in Reproductive Biology and Phylogeny of Birds (ed. Jamieson, B. G. M.) 553–587 (Science, Enfield, 2007).
Stewart, S. G. et al. Species specificity in avian sperm:perivitelline interaction. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 137, 657–663 (2004).
Berlin, S. & Smith, N. G. Testing for adaptive evolution of the female reproductive protein ZPC in mammals, birds and fishes reveals problems with the M7-M8 likelihood ratio test. BMC Evol. Biol. 5, 65 (2005).
Voolstra, C., Tautz, D., Farbrother, P., Eichinger, L. & Harr, B. Contrasting evolution of expression differences in the testis between species and subspecies of the house mouse. Genome Res. 17, 42–49 (2007).
Meiklejohn, C. D., Parsch, J., Ranz, J. M. & Hartl, D. L. Rapid evolution of male-biased gene expression in Drosophila. Proc. Natl Acad. Sci. USA 100, 9894–9899 (2003).
Grantham, R., Gautier, C., Gouy, M., Jacobzone, M. & Mercier, R. Codon catalog usage is a genome strategy modulated for gene expressivity. Nucleic Acids Res. 9, R43–R74 (1981).
Bennetzen, J. L. & Hall, B. D. Codon selection in yeast. J. Biol. Chem. 257, 3026–3031 (1982).
Duret, L. & Mouchiroud, D. Expression pattern and, surprisingly, gene length shape codon usage in Caenorhabditis, Drosophila, and Arabidopsis. Proc. Natl Acad. Sci. USA 96, 4482–4487 (1999).
Coghlan, A. & Wolfe, K. H. Relationship of codon bias to mRNA concentration and protein length in Saccharomyces cerevisiae. Yeast 16, 1131–1145 (2000).
Ikemura, T. Correlation between the abundance of Escherichia coli transfer RNAs and the occurrence of the respective codons in its protein genes: a proposal for a synonymous codon choice that is optimal for the E. coli translational system. J. Mol. Biol. 151, 389–409 (1981).
Ikemura, T. Correlation between the abundance of yeast transfer RNAs and the occurrence of the respective codons in protein genes. Differences in synonymous codon choice patterns of yeast and Escherichia coli with reference to the abundance of isoaccepting transfer RNAs. J. Mol. Biol. 158, 573–597 (1982).
Moriyama, E. N. & Powell, J. R. Codon usage bias and tRNA abundance in Drosophila. J. Mol. Evol. 45, 514–523 (1997).
Duret, L. tRNA gene number and codon usage in the C. elegans genome are co-adapted for optimal translation of highly expressed genes. Trends Genet. 16, 287–289 (2000).
Hambuch, T. M. & Parsch, J. Patterns of synonymous codon usage in Drosophila melanogaster genes with sex-biased expression. Genetics 170, 1691–1700 (2005).
Whittle, C. A., Malik, M. R. & Krochko, J. E. Gender-specific selection on codon usage in plant genomes. BMC Genomics 8, 169 (2007).
Rice, W. R. Sex chromosomes and the evolution of sexual dimorphism. Evolution 38, 735–742 (1984).
Charlesworth, B., Coyne, J. A. & Barton, N. H. The relative rates of evolution of sex chromosomes and autosomes. Am. Nat. 130, 113–146 (1987). References 46 and 47 represent seminal work on the theoretical expectations for the probability of fixation of sexually antagonistic mutations.
Oliver, B. & Parisi, M. Battle of the Xs. Bioessays 26, 543–548 (2004).
Reinke, V. et al. A global profile of germ line gene expression in C. elegans. Mol. Cell 6, 605–616 (2000). One of the first large-scale studies of sex-biased gene expression.
Wang, P. J., McCarrey, J. R., Yang, F. & Page, D. C. An abundance of X-linked genes expressed in spermatogonia. Nature Genet. 27, 422–426 (2001).
Khil, P. P., Smirnova, N. A., Romanienko, P. J. & Camerini-Otero, R. D. The mouse X chromosome is enriched for sex-biased genes not subject to selection by meiotic sex chromosome inactivation. Nature Genet. 36, 642–646 (2004).
Wu, C. I. & Xu, E. Y. Sexual antagonism and X inactivation — the SAXI hypothesis. Trends Genet. 19, 243–247 (2003).
Saifi, G. M. & Chandra, H. S. An apparent excess of sex- and reproduction-related genes on the human X chromosome. Proc. Biol. Sci. 266, 203–209 (1999).
Birchler, J. A., Riddle, N. C., Auger, D. L. & Veitia, R. A. Dosage balance in gene regulation: biological implications. Trends Genet. 21, 219–226 (2005).
Marin, I., Siegal, M. L. & Baker, B. S. The evolution of dosage-compensation mechanisms. Bioessays 22, 1106–1114 (2000).
Heard, E. & Disteche, C. M. Dosage compensation in mammals: fine-tuning the expression of the X chromosome. Genes Dev. 20, 1848–1867 (2006).
Lucchesi, J. C., Kelly, W. G. & Panning, B. Chromatin remodeling in dosage compensation. Annu. Rev. Genet. 39, 615–631 (2005).
Disteche, C. M., Filippova, G. N. & Tsuchiya, K. D. Escape from X inactivation. Cytogenet. Genome Res. 99, 36–43 (2001).
Brown, C. J. & Greally, J. M. A stain upon the silence: genes escaping X inactivation. Trends Genet. 19, 432–438 (2003).
Carrel, L. & Willard, H. F. X-inactivation profile reveals extensive variability in X-linked gene expression in females. Nature 434, 400–404 (2005). This reference provides a detailed insight into the complexity of X-chromosome inactivation.
Jegalian, K. & Page, D. C. A proposed path by which genes common to mammalian X and Y chromosomes evolve to become X inactivated. Nature 394, 776–780 (1998).
Itoh, Y. et al. Dosage compensation is less effective in birds than in mammals. J. Biol. 6, 2 (2007).
Gupta, V. et al. Global analysis of X-chromosome dosage compensation. J. Biol. 5, 3 (2006).
Birchler, J. A., Bhadra, U., Bhadra, M. P. & Auger, D. L. Dosage-dependent gene regulation in multicellular eukaryotes: implications for dosage compensation, aneuploid syndromes, and quantitative traits. Dev. Biol. 234, 275–288 (2001).
Handley, L. J., Ceplitis, H. & Ellegren, H. Evolutionary strata on the chicken Z chromosome: implications for sex chromosome evolution. Genetics 167, 367–376 (2004).
Scholz, B. et al. Sex-dependent gene expression in early brain development of chicken embryos. BMC Neurosci. 7, 12 (2006).
Kaiser, V. B. & Ellegren, H. Nonrandom distribution of genes with sex-biased expression in the chicken genome. Evolution 60, 1945–1951 (2006).
Storchova, R. & Divina, P. Nonrandom representation of sex-biased genes on chicken Z chromosome. J. Mol. Evol. 63, 676–681 (2006).
Gnad, F. & Parsch, J. Sebida: a database for the functional and evolutionary analysis of genes with sex-biased expression. Bioinformatics 22, 2577–2579 (2006).
Waxman, D. & Peck, J. R. Pleiotropy and the preservation of perfection. Science 279, 1210–1213 (1998).
Mank, J. E., Hultin-Rosenberg, L., Zwahlén, M. & Ellegren, H. Pleiotropic constraint hampers the resolution of sexual antagonism in vertebrate gene expression. Am. Nat., (in the press).
Otto, S. P. & Lenormand, T. Resolving the paradox of sex and recombination. Nature Rev. Genet. 3, 252–261 (2002). A comprehensive review of the classical dilemma of why sexual reproduction is so much more common than asexual modes of reproduction.
Oshlack, A., Chabot, A. E., Smyth, G. K. & Gilad, Y. Using DNA microarrays to study gene expression in closely related species. Bioinformatics 23, 1235–1242 (2007).
Velculescu, V. E., Zhang, L., Vogelstein, B. & Kinzler, K. W. Serial analysis of gene expression. Science 270, 484–487 (1995).
Margulies, M. et al. Genome sequencing in microfabricated high-density picolitre reactors. Nature 437, 376–378 (2005).
Bentley, D. R. Whole-genome re-sequencing. Curr. Opin. Genet. Dev. 16, 545–552 (2006).
Stolc, V. et al. A gene expression map for the euchromatic genome of Drosophila melanogaster. Science 306, 655–660 (2004).
McIntyre, L. M. et al. Sex-specific expression of alternative transcripts in Drosophila. Genome Biol. 7, R79 (2006).
Parsch, J., Meiklejohn, C. D. & Hartl, D. L. in Selective Sweep (ed. Nurminsky, D.) 1–12 (Landes Bioscience, Georgetown, 2005).
Parsch, J., Meiklejohn, C. D., Hauschteck-Jungen, E., Hunziker, P. & Hartl, D. L. Molecular evolution of the ocnus and janus genes in the Drosophila melanogaster species subgroup. Mol. Biol. Evol. 18, 801–811 (2001).
Betrán, E., Thornton, K. & Long, M. Retroposed new genes out of the X in Drosophila. Genome Res. 12, 1854–1859 (2002).
Loppin, B., Lepetit, D., Dorus, S., Couble, P. & Karr, T. L. Origin and neofunctionalization of a Drosophila paternal effect gene essential for zygote viability. Curr. Biol. 15, 87–93 (2005).
Kalamegham, R., Sturgill, D., Siegfried, E. & Oliver, B. Drosophila mojoless, a retroposed GSK-3, has functionally diverged to acquire an essential role in male fertility. Mol. Biol. Evol. 24, 732–742 (2007).
Betrán, E. & Long, M. Dntf-2r, a young Drosophila retroposed gene with specific male expression under positive Darwinian selection. Genetics 164, 977–988 (2003).
Begun, D. J. & Lindfors, H. A. Rapid evolution of genomic Acp complement in the melanogaster subgroup of Drosophila. Mol. Biol. Evol. 22, 2010–2021 (2005).
Begun, D. J., Lindfors, H. A., Thompson, M. E. & Holloway, A. K. Recently evolved genes identified from Drosophila yakuba and D. erecta accessory gland expressed sequence tags. Genetics 172, 1675–1681 (2006).
Levine, M. T., Jones, C. D., Kern, A. D., Lindfors, H. A. & Begun, D. J. Novel genes derived from noncoding DNA in Drosophila melanogaster are frequently X-linked and exhibit testis-biased expression. Proc. Natl Acad. Sci. USA 103, 9935–9939 (2006).
Begun, D. J., Lindfors, H. A., Kern, A. D. & Jones C. D. Evidence for de novo evolution of testis-expressed genes in the Drosophila yakuba/Drosophila erecta clade. Genetics 176, 1131–1137 (2007).
Bustamante, C. D. et al. The cost of inbreeding in Arabidopsis. Nature 416, 531–534 (2002).
Acknowledgements
Financial support was obtained from the Swedish Research Council to H. E. and Deutsche Forschungsgemeinschaft to J. P.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
FURTHER INFORMATION
Glossary
- Sexual dimorphism
-
Phenotypic differences between males and females of the same species.
- Sexual selection
-
The process of natural selection acting on traits related directly to mating or reproductive success.
- Sexual antagonism
-
Conflict arising from traits that are beneficial to one sex but harmful to the other.
- Sex-biased genes
-
A gene that is expressed predominantly or exclusively in one sex.
- Codon bias
-
The non-random use of synonymous codons to encode a protein.
- dN/dS
-
The ratio of the non-synonymous (amino-acid altering) and synonymous substitution rates, used as a standard measure of the rate of evolution of a protein-encoding sequence.
- EST sequencing
-
Large-scale sequencing of clones from a cDNA library obtained from mRNA.
- Transcriptome profiling
-
A characterization of the mRNA molecules that are expressed in a certain tissue.
- Androdioecious
-
A population consisting of hermaphrodites, which contain both male and female reproductive tissues, and males, which contain only male reproductive tissues.
- Positive selection
-
Positive selection is key to adaptive evolution and implies that an advantageous allele gives its carrier a higher fitness and is therefore favoured by natural selection.
- Fixation
-
When a new mutation is eventually spread to all individuals in the population.
- Accessory glands
-
In insects, such as Drosophila, these are male reproductive tissues that produce and secrete seminal fluid proteins that are transferred to the female during copulation.
- Purifying selection
-
Negative selection against a deleterious or slightly deleterious mutation.
- Heterogametic sex
-
The sex that produces two different types of gametes with respect to sex chromosome content (for example, XY).
- Homogametic sex
-
The sex that produces one type of gamete with respect to sex chromosome content (for example, XX).
- Retrogene
-
A gene duplicate that arose through reverse transcription of a cellular mRNA.
- Subfunctionalization
-
The partitioning of multiple ancestral gene functions between gene copies following duplication.
- Neofunctionalization
-
A gain of a new function to a duplicated gene through mutation and selection.
- Paralogues
-
Genes that arose from duplication of a common, ancestral gene. A gene that was duplicated before the divergence of two species is present as two paralogues, each of which has an orthologue in the other species.
- Pleiotropy
-
The influence that a single gene has on multiple traits.
- Epistatic selection
-
Selection for a certain combination of alleles at two or more loci.
Rights and permissions
About this article
Cite this article
Ellegren, H., Parsch, J. The evolution of sex-biased genes and sex-biased gene expression. Nat Rev Genet 8, 689–698 (2007). https://doi.org/10.1038/nrg2167
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrg2167
This article is cited by
-
Evolution of chemosensory tissues and cells across ecologically diverse Drosophilids
Nature Communications (2024)
-
Sex differences in energy metabolism: natural selection, mechanisms and consequences
Nature Reviews Nephrology (2024)
-
Estimating transcriptome complexities across eukaryotes
BMC Genomics (2023)
-
Tests for associations between sexual dimorphism and patterns of quantitative genetic variation in the water strider, Aquarius remigis
Heredity (2023)
-
The roles of sexual selection and sexual conflict in shaping patterns of genome and transcriptome variation
Nature Ecology & Evolution (2023)