Abstract
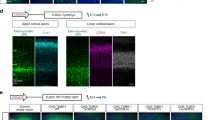
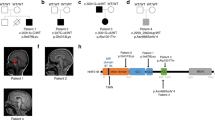
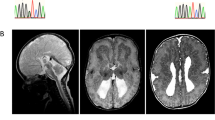
The genetic causes of malformations of cortical development (MCD) remain largely unknown. Here we report the discovery of multiple pathogenic missense mutations in TUBG1, DYNC1H1 and KIF2A, as well as a single germline mosaic mutation in KIF5C, in subjects with MCD. We found a frequent recurrence of mutations in DYNC1H1, implying that this gene is a major locus for unexplained MCD. We further show that the mutations in KIF5C, KIF2A and DYNC1H1 affect ATP hydrolysis, productive protein folding and microtubule binding, respectively. In addition, we show that suppression of mouse Tubg1 expression in vivo interferes with proper neuronal migration, whereas expression of altered γ-tubulin proteins in Saccharomyces cerevisiae disrupts normal microtubule behavior. Our data reinforce the importance of centrosomal and microtubule-related proteins in cortical development and strongly suggest that microtubule-dependent mitotic and postmitotic processes are major contributors to the pathogenesis of MCD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
28 June 2013
In the version of this article initially published, the label "– Tub" in Figure 3d was placed incorrectly. The label should appear adjacent to the lower bracketed panels rather than adjacent to the bottom portion of the upper bracketed panels. The error has been corrected in the HTML and PDF versions of the article.
References
Caviness, V.S. Jr., Takahashi, T. & Nowakowski, R.S. Numbers, time and neocortical neuronogenesis: a general developmental and evolutionary model. Trends Neurosci. 18, 379–383 (1995).
Rakic, P. & Caviness, V.S. Jr. Cortical development: view from neurological mutants two decades later. Neuron 14, 1101–1104 (1995).
Francis, F. et al. Human disorders of cortical development: from past to present. Eur. J. Neurosci. 23, 877–893 (2006).
Guerrini, R., Dobyns, W.B. & Barkovich, A.J. Abnormal development of the human cerebral cortex: genetics, functional consequences and treatment options. Trends Neurosci. 31, 154–162 (2008).
Barkovich, A.J., Guerrini, R., Kuzniecky, R.I., Jackson, G.D. & Dobyns, W.B. A developmental and genetic classification for malformations of cortical development: update 2012. Brain 135, 1348–1369 (2012).
Ostergaard, P. et al. Mutations in KIF11 cause autosomal-dominant microcephaly variably associated with congenital lymphedema and chorioretinopathy. Am. J. Hum. Genet. 90, 356–362 (2012).
Kousar, R. et al. Mutations in WDR62 gene in Pakistani families with autosomal recessive primary microcephaly. BMC Neurol. 11, 119 (2011).
Jaglin, X.H. & Chelly, J. Tubulin-related cortical dysgeneses: microtubule dysfunction underlying neuronal migration defects. Trends Genet. 25, 555–566 (2009).
Tischfield, M.A., Cederquist, G.Y., Gupta, M.L. Jr. & Engle, E.C. Phenotypic spectrum of the tubulin-related disorders and functional implications of disease-causing mutations. Curr. Opin. Genet. Dev. 21, 286–294 (2011).
Breuss, M. et al. Mutations in the β-tubulin gene TUBB5 cause microcephaly with structural brain abnormalities. Cell Rep. 2, 1554–1562 (2012).
Bilgüvar, K. et al. Whole-exome sequencing identifies recessive WDR62 mutations in severe brain malformations. Nature 467, 207–210 (2010).
Manzini, M.C. & Walsh, C.A. What disorders of cortical development tell us about the cortex: one plus one does not always make two. Curr. Opin. Genet. Dev. 21, 333–339 (2011).
Veltman, J.A. & Brunner, H.G. De novo mutations in human genetic disease. Nat. Rev. Genet. 13, 565–575 (2012).
Vissers, L.E. et al. A de novo paradigm for mental retardation. Nat. Genet. 42, 1109–1112 (2010).
Li, Y. et al. Resequencing of 200 human exomes identifies an excess of low-frequency non-synonymous coding variants. Nat. Genet. 42, 969–972 (2010).
Sindelar, C.V. & Downing, K.H. An atomic-level mechanism for activation of the kinesin molecular motors. Proc. Natl. Acad. Sci. USA 107, 4111–4116 (2010).
Dunn, S. et al. Differential trafficking of Kif5c on tyrosinated and detyrosinated microtubules in live cells. J. Cell Sci. 121, 1085–1095 (2008).
Nakata, T. & Hirokawa, N. Point mutation of adenosine triphosphate-binding motif generated rigor kinesin that selectively blocks anterograde lysosome membrane transport. J. Cell Biol. 131, 1039–1053 (1995).
Homma, N. et al. Kinesin superfamily protein 2A (KIF2A) functions in suppression of collateral branch extension. Cell 114, 229–239 (2003).
Kumar, P., Henikoff, S. & Ng, P.C. Predicting the effects of coding non-synonymous variants on protein function using the SIFT algorithm. Nat. Protoc. 4, 1073–1081 (2009).
Ramensky, V., Bork, P. & Sunyaev, S. Human non-synonymous SNPs: server and survey. Nucleic Acids Res. 30, 3894–3900 (2002).
Hirokawa, N., Noda, Y., Tanaka, Y. & Niwa, S. Kinesin superfamily motor proteins and intracellular transport. Nat. Rev. Mol. Cell Biol. 10, 682–696 (2009).
Poirier, K. et al. Expanding the spectrum of TUBA1A-related cortical dysgenesis to polymicrogyria. Eur. J. Hum. Genet. 21, 381–385 (2013).
Harms, M.B. et al. Mutations in the tail domain of DYNC1H1 cause dominant spinal muscular atrophy. Neurology 78, 1714–1720 (2012).
Weedon, M.N. et al. Exome sequencing identifies a DYNC1H1 mutation in a large pedigree with dominant axonal Charcot-Marie-Tooth disease. Am. J. Hum. Genet. 89, 308–312 (2011).
Carter, A.P. et al. Structure and functional role of dynein's microtubule-binding domain. Science 322, 1691–1695 (2008).
Gibbons, I.R. et al. The affinity of the dynein microtubule-binding domain is modulated by the conformation of its coiled-coil stalk. J. Biol. Chem. 280, 23960–23965 (2005).
Wise, D.O., Krahe, R. & Oakley, B.R. The γ-tubulin gene family in humans. Genomics 67, 164–170 (2000).
Kollman, J.M., Merdes, A., Mourey, L. & Agard, D.A. Microtubule nucleation by γ-tubulin complexes. Nat. Rev. Mol. Cell Biol. 12, 709–721 (2011).
Melki, R., Vainberg, I.E., Chow, R.L. & Cowan, N.J. Chaperonin-mediated folding of vertebrate actin-related protein and γ-tubulin. J. Cell Biol. 122, 1301–1310 (1993).
Erickson, R.P. Somatic gene mutation and human disease other than cancer: an update. Mutat. Res. 705, 96–106 (2010).
de Ligt, J. et al. Diagnostic exome sequencing in persons with severe intellectual disability. N. Engl. J. Med. 367, 1921–1929 (2012).
Dobyns, W.B. et al. Differences in the gyral pattern distinguish chromosome 17–linked and X–linked lissencephaly. Neurology 53, 270–277 (1999).
Tsurusaki, Y. et al. A DYNC1H1 mutation causes a dominant spinal muscular atrophy with lower extremity predominance. Neurogenetics 13, 327–332 (2012).
Willemsen, M.H. et al. Mutations in DYNC1H1 cause severe intellectual disability with neuronal migration defects. J. Med. Genet. 49, 179–183 (2012).
Higginbotham, H.R. & Gleeson, J.G. The centrosome in neuronal development. Trends Neurosci. 30, 276–283 (2007).
Wynshaw-Boris, A., Pramparo, T., Youn, Y.H. & Hirotsune, S. Lissencephaly: mechanistic insights from animal models and potential therapeutic strategies. Semin. Cell Dev. Biol. 21, 823–830 (2010).
Kanai, Y. et al. KIF5C, a novel neuronal kinesin enriched in motor neurons. J. Neurosci. 20, 6374–6384 (2000).
Harada, A. et al. Golgi vesiculation and lysosome dispersion in cells lacking cytoplasmic dynein. J. Cell Biol. 141, 51–59 (1998).
Hafezparast, M. et al. Mutations in dynein link motor neuron degeneration to defects in retrograde transport. Science 300, 808–812 (2003).
Ori-McKenney, K.M. & Vallee, R.B. Neuronal migration defects in the Loa dynein mutant mouse. Neural Dev. 6, 26 (2011).
Shu, T. et al. Ndel1 operates in a common pathway with LIS1 and cytoplasmic dynein to regulate cortical neuronal positioning. Neuron 44, 263–277 (2004).
Keays, D.A. et al. Mutations in α-tubulin cause abnormal neuronal migration in mice and lissencephaly in humans. Cell 128, 45–57 (2007).
Poirier, K. et al. Large spectrum of lissencephaly and pachygyria phenotypes resulting from de novo missense mutations in tubulin α 1A (TUBA1A). Hum. Mutat. 28, 1055–1064 (2007).
Jaglin, X.H. et al. Mutations in the β-tubulin gene TUBB2B result in asymmetrical polymicrogyria. Nat. Genet. 41, 746–752 (2009).
Poirier, K. et al. Mutations in the neuronal ss-tubulin subunit TUBB3 result in malformation of cortical development and neuronal migration defects. Hum. Mol. Genet. 19, 4462–4473 (2010).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
DePristo, M.A. et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat. Genet. 43, 491–498 (2011).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Flicek, P. et al. Ensembl 2012. Nucleic Acids Res. 40, D84–D90 (2012).
Studier, F.W., Rosenberg, A.H., Dunn, J.J. & Dubendorff, J.W. Use of T7 RNA polymerase to direct expression of cloned genes. Methods Enzymol. 185, 60–89 (1990).
Gao, Y., Thomas, J.O., Chow, R.L., Lee, G.H. & Cowan, N.J. A cytoplasmic chaperonin that catalyzes β-actin folding. Cell 69, 1043–1050 (1992).
Tian, G., Vainberg, I.E., Tap, W.D., Lewis, S.A. & Cowan, N.J. Specificity in chaperonin-mediated protein folding. Nature 375, 250–253 (1995).
Bernal, C., Palacin, C., Boronat, A. & Imperial, S. A colorimetric assay for the determination of 4-diphosphocytidyl-2-C-methyl-D-erythritol 4-phosphate synthase activity. Anal. Biochem. 337, 55–61 (2005).
Shelanski, M.L., Gaskin, F. & Cantor, C.R. Microtubule assembly in the absence of added nucleotides. Proc. Natl. Acad. Sci. USA 70, 765–768 (1973).
Acknowledgements
We are grateful to the patients and their families for their participation. We thank F.J. Fourniol, N. Levy and members of the Chelly lab for their thoughtful comments and help. We thank I. Gibbons and A. Carter (Medical Research Council Laboratory of Molecular Biology) for providing the seryl-tRNA synthetase dynein heavy chain fusion constructs used in the experiments described in this paper. A full-length cDNA encoding human KIF5C was generously provided by F.A. Stephenson (School of Pharmacy, University College London). A full-length cDNA encoding human γ-tubulin was generously provided by B.R. Oakley (Department of Molecular Biosciences, University of Kansas). We also thank members of the Cochin Hospital Cell Bank, the Centre National de Genotypage, Genoscope, the Paris Descartes Bioinformatics platforms and the Cochin Institute genomic platform for their technical and bioinformatic assistance. This work was supported by funding from INSERM, the Fondation pour la Recherche Médicale (FRM funding within the frame of the programme Equipe FRM; J.C.), the Fondation JED, the Fondation Maladies Rares, the Agence National de Recherche (ANR Blanc 1103 01, project R11039KK; ANR E-Rare-012-01, project E10107KP) and the EU-FP7 project GENECODYS, grant number 241995. This work was also supported by a grant (GM097376 to N.J.C.) from the US National Institutes of Health.
Author information
Authors and Affiliations
Contributions
J.C. coordinated and instigated the study with N.J.C. K.P. and N.L. analyzed WES data and performed genetic and molecular studies. L.B. and Y.S. performed and analyzed in utero RNAi experiments. F.F. provided technical assistance for in utero RNAi experiments. N.B.-B. and R.G. coordinated collection of clinical and imaging data. D.G., E.F., F.D., C.C., M.B., D.B., B.D.B., S.N., C.G., P.P., V.d.P., J.M.P., D.L. and V.L. helped in selecting patients. S.V., B.S.P. and M.O. analyzed variations in candidate genes and screened subject DNA. G.T. and N.J.C. performed the biochemical studies. C.B. and A.A. performed the yeast studies. L.C.-P. and E.P. performed all DNA extractions from patient samples. P.N., T.H. and C.M. performed bioinformatics analysis of exome sequencing data. D.Z. coordinated the WES procedure (library generation, exome enrichment and WES). J.C. and N.J.C. drafted and finalized the manuscript with the help of K.P., N.L., L.B. and Y.S.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Tables 1–8 (PDF 2769 kb)
Rights and permissions
About this article
Cite this article
Poirier, K., Lebrun, N., Broix, L. et al. Mutations in TUBG1, DYNC1H1, KIF5C and KIF2A cause malformations of cortical development and microcephaly. Nat Genet 45, 639–647 (2013). https://doi.org/10.1038/ng.2613
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2613
This article is cited by
-
Cargo specificity, regulation, and therapeutic potential of cytoplasmic dynein
Experimental & Molecular Medicine (2024)
-
Regulation of ciliary homeostasis by intraflagellar transport-independent kinesins
Cell Death & Disease (2024)
-
Spinal Muscular Atrophy with Predominant Lower Extremity (SMA-LED) with Intellectual Disability and Dysmorphism
Indian Journal of Pediatrics (2023)
-
Insights on the Role of α- and β-Tubulin Isotypes in Early Brain Development
Molecular Neurobiology (2023)
-
KIF5C deficiency causes abnormal cortical neuronal migration, dendritic branching, and spine morphology in mice
Pediatric Research (2022)