Abstract
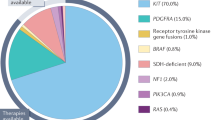
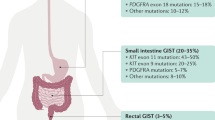
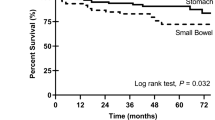
We are living in an exciting era in the treatment of cancer, using drugs that target specific proteins rather than agents that cause more general cytotoxic effects. The identification of proteins and signal transduction pathways that play crucial roles in the pathogenesis of cancer has allowed treatments to be designed that target these tumor-driven events. Gastrointestinal stromal tumors (GIST) are rare mesenchymal tumors and were among the first solid tumor types for which such a novel treatment (in this case imatinib) became available. The tyrosine kinase inhibitor imatinib targets the human KIT receptor and the platelet-derived growth factor receptor-α. This drug exhibits impressive antitumor effects against GIST and has become the first-line therapy for advanced disease. Major insights into the mechanism of action of this drug, drug resistance, and patient management issues have been gleaned. Additionally, new drugs developed for the treatment of GIST have been identified. As a consequence, lessons learned from GIST are widely applicable to other tumor entities, thereby rendering GIST the paradigm of solid tumors treated with tyrosine kinase inhibitors. This Review discusses the pathogenesis of GIST, treatment strategies, mechanisms accounting for drug resistance, and potential future perspectives.
Key Points
-
Improved insights into the molecular mechanisms that cause malignancy have been the foundation of the development of “cancer-(cell-)specific therapy” with tyrosine kinase inhibitors for the treatment of gastrointestinal stromal tumors (GIST)
-
Understanding the mechanisms that confer resistance against tyrosine kinase inhibitors has yielded additional novel treatment strategies for GIST
-
Elucidation of prognostic and predictive factors in GIST offers opportunities for individualizing patient treatment
-
GIST has become the paradigm for the treatment of solid tumors with tyrosine kinase inhibitors
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Corless CL et al. (2004) Biology of gastrointestinal stromal tumors. J Clin Oncol 22: 3813–3825
Tornillo L et al. (2005) An update on molecular genetics of gastrointestinal stromal tumours. J Clin Pathol 59: 557–563
Hirota S et al. (1998) Gain-of-function mutations of c-KIT in human gastro-intestinal tumors. Science 279: 577–580
Heinrich MC et al. (2003) Kinase mutations and imatinib responses in patients with metastatic gastrointestinal stromal tumors. J Clin Oncol 21: 4342–4349
Debiec-Rychter M et al. (2004) Use of c-KIT/PDGFRA mutational analysis to predict the clinical response to imatinib in patients with advanced gastrointestinal stromal tumors entered on phase I and II studies of the EORTC Soft Tissue and Bone Sarcoma Group. Eur J Cancer 40: 689–695
Debiec-Rychter M et al. (2005) Mechanisms of resistance to imatinib mesylate in gastrointestinal stromal tumors and activity of PKC412 against imatinib-resistant mutants. Gastroenterology 128: 270–279
Antonescu CR et al. (2004) Gene expression in gastrointestinal stromal tumors is distinguished by KIT genotype and anatomic site. Clin Cancer Res 10: 3282–3290
Heinrich MC et al. (2003) PDGFRA activating mutations in gastrointestinal stromal tumors. Science 299: 708–710
Rubin BP et al. (2001) KIT activation is a ubiquitous feature of gastrointestinal stromal tumor. Cancer Res 61: 8118–8121
Fletcher CDM et al. (2002) Diagnosis of gastrointestinal stromal tumors: a consensus approach. Hum Pathol 33: 459–465
Nilsson B et al. (2005) Gastrointestinal stromal tumors: the incidence, prevalence, clinical course, and prognostication in the preimatinib mesylate area. Cancer 103: 821–829
DeMatteo RP et al. (2000) Two hundred gastrointestinal stromal tumors. Recurrence patterns and prognostic factors for survival. Ann Surg 231: 51–58
Imamura M et al. (2007) Prognostic significance of angiogenesis in gastrointestinal stromal tumors. Mod Pathol 20: 529–537
Miettinen M et al. (2005) Gastrointestinal stromal tumors of the stomach: a clinicopathologic, immunohistochemical, and molecular genetic study of 1765 cases with long-term follow-up. Am J Surg Pathol 29: 52–68
Miettinen M et al. (2006) Gastrointestinal stromal tumors of the jejunum and ileum: a clinicopathologic, immunohistochemical, and molecular genetic study of 906 cases before imatinib with long-term follow-up. Am J Surg Pathol 30: 477–489
El-Rifai W et al. (2000) DNA sequence copy number changes in gastrointestinal stromal tumors: tumor progression and prognostic significance. Cancer Res 60: 3899–3903
Schneider-Stock R et al. (2005) Loss of p16 protein defines high-risk patients with gastrointestinal stromal tumors: a tissue microarray study. Clin Cancer Res 11: 638–645
Huang H-Y et al. (2006) Immunohistochemical expression of p16INK4A, Ki-67, and Mcm2 proteins in gastrointestinal stromal tumors: prognostic implications and correlations with risk stratification of NIH consensus. Ann Surg Oncol 13: 1633–1644
House MG et al. (2003) Tumor suppressor gene hypermethylation as a predictor of gastric stromal tumor behavior. J Gastrointest Surg 7: 1004–1014
Sleijfer S et al. (2006) Gastrointestinal stromal tumors. In: UICC: Prognostic Factors in Cancer, edn 3, 187–191 (Eds Gospodarowicz MK et al.) New Jersey: John Wiley & Sons
Singer S et al. (2002) Prognostic value of KIT mutation type, mitotic activity, and histologic subtype in gastrointestinal stromal tumors. J Clin Oncol 20: 3898–3905
Wardelmann E et al. (2003) Deletion of Trp-557 and Lys-558 in the juxtamembrane domain of the c-kit protooncogene is associated with metastatic behavior of gastrointestinal stromal tumors. Int J Cancer 106: 887–895
DeMatteo R et al. (2007) Adjuvant imatinib mesylate increases recurrence free survival (RFS) in patients with completely resected localized primary gastrointestinal stromal tumor (GIST): North American Intergroup Phase III trial ACOSOG Z9001 [abstract 10079]. Proc Am Soc Clin Oncol 25
Van Glabbeke M et al. (2002) Progression-free rate as the principal end point for phase II trials in soft tissue sarcomas. Eur J Cancer 38: 543–549
Verweij J et al. (2004) Progression-free survival in gastrointestinal stromal tumours with high-dose imatinib: a randomised trial. Lancet 364: 1127–1134
Fabian MA et al. (2005) A small molecule-kinase interaction map for clinical kinase inhibitors. Nat Biotechnol 23: 329–336
Van Oosterom AT et al. (2001) Safety and efficacy of imatinib (STI571) in metastatic gastrointestinal stromal tumours: a phase I study. Lancet 358: 1421–1423
Demetri GD et al. (2002) Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N Engl J Med 347: 472–480
Verweij J et al. (2003) Imatinib mesylate (STI-571 Glivec, Gleevec) is an active agent for gastrointestinal stromal tumors, but does not yield responses in other soft tissue sarcomas that are unselected for a molecular target. Results from an EORTC Soft Tissue and Bone Sarcoma phase II study. Eur J Cancer 39: 2006–2011
Van Glabbeke M et al. (2007) Comparison of two doses of imatinib for the treatment of gastrointestinal stromal tumors (GIST): a meta-analysis based on 1640 patients [abstract 10004]. Proc Am Soc Clin Oncol 25
Blanke CD et al. (2006) Long-term follow-up of a phase II randomized trial in advanced gastrointestinal stromal tumor patients treated with imatinib mesylate [abstract 9528]. Proc Am Soc Clin Oncol 24
Blay J-Y et al. (2007) Prospective multicentric randomized phase III study of imatinib in patients with advanced gastrointestinal stromal tumors comparing interruption versus continuation of treatment beyond 1 year: The French Sarcoma Group. J Clin Oncol 25: 1107–1113
Kerkela R et al. (2006) Cardiotoxicity of the cancer therapeutic agent imatinib mesylate. Nat Med 12: 908–916
Verweij J et al. (2007) Imatinib does not induce cardiac left ventricular failure in gastrointestinal stromal tumours patients: analysis of EORTC-ISG-AGITG study 62005. Eur J Cancer 43: 974–978
Van Glabbeke M et al. (2006) Predicting toxicities for patients with advanced gastrointestinal stromal tumours treated with imatinib: a study of the European Organisation for Research and Treatment of Cancer, the Italian Sarcoma Group, and the Australasian Gastro-Intestinal Trials Group (EORTC-ISG-AGITG). Eur J Cancer 42: 2277–2285
Van Glabbeke et al. (2007) Interactive risk calculator for predicting toxicities in patients with advanced gastro-intestinal stromal tumors (GIST) treated with imatinib mesylate. www.eortc.be/tools/imatinibtoxicity (accessed 10 May 2007)
Bechtold RE et al (2003) Cystic changes in hepatic and peritoneal metastases from gastrointestinal stromal tumors treated with Gleevec. Abdom Imaging 28: 808–814
Linton KM et al. (2006) Response evaluation in gastrointestinal stromal tumours treated with imatinib: misdiagnosis of disease progression on CT due to cystic change in liver metastases. Br J Radiol 79: e40–e44
Loughrey MB et al. (2005) Gastrointestinal stromal tumour treated with neoadjuvant imatinib. J Clin Pathol 58: 779–781
Choi H et al. (2007) Correlation of computed tomography and positron emission tomography in patients with metastatic gastrointestinal stromal tumor treated at a single institution with imatinib mesylate: proposal of new computed tomography response criteria. J Clin Oncol 25: 1753–1759
Benjamin RS et al. (2007) We should desist using RECIST, at least in GIST. J Clin Oncol 25: 1760–1764
Van Glabbeke M et al. (2005) Initial and late resistance to imatinib in advanced gastrointestinal stromal tumors are predicted by different prognostic factors: a European Organisation of Treatment and Research of Cancer—Italian Sarcoma Group—Australasian Gastrointestinal Trials Group Study. J Clin Oncol 23: 5795–5804
Heinrich MC et al. (2006) Molecular correlates of imatinib resistance in gastrointestinal stromal tumors. J Clin Oncol 24: 4764–4774
Debiec-Rychter M et al. (2006) KIT mutations and dose selection for imatinib in patients with advanced gastrointestinal stromal tumours. Eur J Cancer 42: 1093–1103
Wardelmann E et al. (2006) Polyclonal evolution of multiple secondary KIT mutations in gastrointestinal stromal tumors under treatment with imatinib mesylate. Clin Cancer Res 12: 1743–1749
Antonescu CR et al. (2005) Acquired resistance to imatinib in gastrointestinal stromal tumor occurs through secondary gene mutation. Clin Cancer Res 11: 4182–4190
Tamborini E et al. (2006) Functional analyses and molecular modelling of two c-Kit mutations responsible for imatinib secondary resistance in GIST patients. Oncogene 25: 6140–6146
Fletcher JA et al. (2003) Mechanisms of resistance to imatinib (IM) in advanced gastrointestinal stromal tumors (GIST) [abstract 3275]. Proc Am Soc Clin Oncol 22
Theou N et al. (2005) Multidrug resistance proteins in gastrointestinal stromal tumors: site-dependent expression and initial response to imatinib. Clin Cancer Res 11: 7593–7598
Mahon FX et al. (2003) MDR1 gene overexpression confers resistance to imatinib mesylate in leukemia cell line models. Blood 101: 2368–2373
Burger H et al. (2004) Imatinib mesylate (STI571) is a substrate for the breast cancer resistance protein (BCRP)/ABCG2 drug pump. Blood 104: 2940–2942
Gambacorti-Passerini C et al. (2003) Alpha1 acid glycoprotein binds to imatinib (STI571) and substantially alters its pharmacokinetics in chronic myeloid leukemia patients. Clin Cancer Res 9: 625–632
Delbado C et al. (2006) Pharmacokinetic-pharmadynamic relationships of imatinib and its main metabolite in patients with advanced gastrointestinal stromal tumors. Clin Cancer Res 12: 6073–6078
Judson IR et al. (2005) Imatinib pharmacokinetics in patients with gastrointestinal stromal tumour: a retrospective population pharmacokinetic study over time. Cancer Chemother Pharmacol 55: 379–386
Zalcberg JR et al. (2005) Outcome of patients with advanced gastro-intestinal stromal tumours crossing over to a daily imatinib dose of 800 mg after progression on 400 mg. Eur J Cancer 41: 1751–1757
Prenen H et al. (2006) Efficacy of the kinase inhibitor SU11248 against gastrointestinal stromal tumor mutants refractory to imatinib mesylate. Clin Cancer Res 12: 2622–2627
Bui BN et al. (2007) Preliminary efficacy and safety results of masitinib administered, front line in patients with advanced GIST. A phase II study [abstract 10025]. Proc Am Soc Clin Oncol 25
Joensuu H et al. (2006) A phase II, open-label study of PTK787/ZK222584 in the treatment of metastatic gastrointestinal stromal tumors resistant to imatinib mesylate [abstract 9531]. Proc Am Soc Clin Oncol 24
Reichardt P et al. (2005) A phase I/II trial of the oral PKC-inhibitor PKC412 in combination with imatinib mesylate (IM) in patients with gastrointestinal stromal tumors refractory to IM [abstract 3016]. Proc Am Soc Clin Oncol 23
Von Mehren M et al. (2007) A phase I study of nilotinib alone and in combination with imatinib (IM) in patients (pts) with imatinib-resistant gastrointestinal stromal tumors (GIST)—study update [abstract 10023]. Proc Am Soc Clin Oncol 25
Demetri GD et al. (2006) Efficacy and safety of sunitinib in patients with advanced gastrointestinal stromal tumours after failure of imatinib: a randomised controlled trial. Lancet 368: 1329–1338
Heinrich M et al. (2006) Sunitinib response in imatinib-resistant GIST correlates with KIT and PDGFRA mutations [abstract 9502]. Proc Am Soc Clin Oncol 24
Van Oosterom A et al. (2005) A phase I/II trial of the oral mTOR-inhibitor everolimus and imatinib mesylate (IM) in patients with gastrointestinal stromal tumor refractory to IM [abstract 9033]. Proc Am Soc Clin Oncol 23
Steinert DM et al. (2006) Expression of Bcl-2 in gastrointestinal stromal tumors. Cancer 106: 1617–1623
Takahashi R et al. (2003) Expression of vascular endothelial growth factor and angiogenesis in gastrointestinal stromal tumor of the stomach. Oncology 64: 266–274
Salto-Tellez M et al. (2007) Tissue microarrays characterize the clinical significance of a VEGF-A protein expression signature in gastrointestinal stromal tumours. Br J Cancer 96: 776–782
Bauer S et al. (2006) Heat shock protein 90 inhibition in imatinib-resistant gastrointestinal stromal tumor. Cancer Res 66: 9153–9161
Demetri GD et al. (2007) Inhibition of the heat shock protein 90 (Hsp90) chaperone with the novel agent IPI-504 to overcome resistance to tyrosine kinase inhibitors (TKIs) in metastatic GIST: updated results of a phase I trial [abstract 10024]. Proc Am Soc Clin Oncol 25
Acknowledgements
This research was supported by the EC FP6 CONTICANET project (LSHC-CT-2005-018806) from the European Commission.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Sleijfer, S., Wiemer, E. & Verweij, J. Drug Insight: gastrointestinal stromal tumors (GIST)—the solid tumor model for cancer-specific treatment. Nat Rev Clin Oncol 5, 102–111 (2008). https://doi.org/10.1038/ncponc1037
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncponc1037
This article is cited by
-
Combination therapy with c-met inhibitor and TRAIL enhances apoptosis in dedifferentiated liposarcoma patient-derived cells
BMC Cancer (2019)
-
MRI in predicting the response of gastrointestinal stromal tumor to targeted therapy: a patient-based multi-parameter study
BMC Cancer (2018)
-
Adjuvant imatinib for patients with high-risk gastrointestinal stromal tumors: a retrospective cohort study
Scientific Reports (2017)
-
Pathologie von neuroendokrinen Neoplasien (NEN) und gastrointestinalen Stromatumoren (GIST)
Der Gastroenterologe (2015)
-
Determining the optimal dose in the development of anticancer agents
Nature Reviews Clinical Oncology (2014)