Abstract
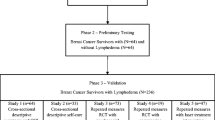
This paper documents the validation of a quality of life scale (QOL) designed to assess the impact of arm morbidity on patients following breast cancer surgery. A four item arm subscale was developed to supplement a multi-dimensional, validated breast cancer QOL tool, the functional assessment of cancer therapy (FACT-B.) The new questionnaire, the FACT-B+4, was validated on 279 women participating in a trial of sentinel node guided axillary therapy and 29 women attending a lymphoedema clinic. The subscale demonstrated good internal consistency (alpha co-efficient=0.62 to 0.88) and stability (test–retest reliability=0.97). Lymphoedema patients reported significantly greater arm problems than a matched sample of pre-operative trial participants. The lymphoedema group also scored lower than trial patients on the FACT-B+4 indicating a poorer quality of life (p<0.05). A subset of 66 trial patients who had completed three consecutive assessments was used to evaluate the sensitivity of the questionnaire to change over time. Scores on the FACT-B+4 were found to decline significantly between the pre-operative assessment and post-operative assessment at 1 month. Arm problems significantly increased during this period. FACT-B+4 score increased again from 1 month to 12 weeks post-surgery and symptoms reduced, as the extent of arm morbidity resolved. The FACT-B+4 appears to be psychometrically robust and sensitive to patient rehabilitation, making it suitable for use in longitudinal surgical trials. Given the dearth of existing scales available to measure arm morbidity, we hope this new tool will prove useful to researchers
Similar content being viewed by others
References
Harries S: A survey of the management of breast cancer in England and Wales. Ann Surg 220: 197-202, 1994
Hladiuk M, Huchcroft S, Temple W, Schnurr B: Arm function after axillary dissection for breast cancer: a pilot study to provide parameter estimates. J Surg Oncol 50: 47-52, 1992
Tasmuth T, von Smitten K, Kalso E: Pain and other symptoms during the first year after radical and conservative surgery for breast cancer. Br J Cancer 74: 2024-2031, 1996
Ivens D, Hoe A, Podd T, Hamilton C, Taylor I, Royle G: Assessment of morbidity from complete axillary dissection. Br J Cancer 66: 135-138, 1992
Kissin M, Querci G, Easton D, Westbury G: Risk of lymphoedema following the treatment of breast cancer. Br J Surg 73: 580-584, 1986
Maunsell E, Brisson J, Deshenes L: Arm problems and psychological distress after surgery for breast cancer. Can J Surg 36: 315-320, 1993
Logan V: Incidence and prevalence of lymphoedema: a literature review. J Clin Nurs 4: 213-219, 1995
Fellowes D, Fallowfield L, Saunders C, Houghton J: Tolerability of hormone therapies for breast cancer: a comparison of documented and patient-reported symptoms. Breast Cancer Res Treat 66: 73-81, 2001
Warmuth M, Bowen G, Prosnitz L, Chu L, Broadwater G, Peterson B: Complications of axillary lymph node dissection for carcinoma of the breast: a report based on a patient survey. Cancer 83: 1362-1368, 1998
Liljgren G, Holmberg L, Uppsala-Orebro Breast Cancer Study Group: Arm morbidity after sector resection and axillary dissection with or without postoperative radiotherapy in breast cancer stage 1. Results from a randomised trial. Eur J Cancer 33: 193-199, 1997
Kiel K, Rademacker A: Early stage breast cancer: arm edema after wide excision and breast irradiation. Radiology 198: 279-283, 1996
Keramopoulos A, Tsionou C, Minaretzis D, Michalas S, Aravantinos D: Arm morbidity following treatment for breast cancer with total axillary dissection: a multivariated approach. Oncol 50: 445-449, 1993
Denton S (on behalf of EONS and the Working Group for Living with Advanced Breast Cancer Hormone Treatment (WGLABCHT)): Living with hormone therapy for advanced breast cancer: a European survey of oncologists’ and nurses’ perceptions. Eur J Cancer 31A: 5: 1280, 1995
Shimozuma K, Ganz P, Petersen L, Hirji K: Quality of life in the first year after breast cancer surgery: rehabilitation needs and patterns of recovery. Breast Cancer Res Treat 56: 45-47, 1999
Hack T, Cohen L, Katz J, Robson L, Goss P: Physical and psychological morbidity following axillary lymph node dissection for breast cancer. J Clin Oncol 17: 143-149, 1999
Woods M, Tobin M, Mortimer P: The psychological morbidity of breast cancer patients with lymphoedema. Cancer Nurs 18: 467-471, 1995
Groenvold M: Quality of life in breast cancer adjuvant therapy: validation and pilot testing of a combination of questionnaires. The Breast 6: 97-107, 1997
Tob in M, Lacey H, Meyer L, Mortimer P: The psychological morbidity of breast cancer-related swelling. Cancer 72: 3248-3252, 1993
Hall A, A'Hern R, Fallowfield L: Are we using appropriate self report questionnaires for detecting anxiety and depression in women with early breast cancer. Eur J Cancer 35(1): 79-85, 1999
Sitzia J, Sobrido L: Measurement of health-related quality of life of patients receiving conservative treatment for limb lymphoedema using the Nottingham health profile. Qual Life Res 6: 373-384, 1997
Velanovich V, Szymanski W: Quality of life of breast cancer patients with lymphoedema. Am J Surg 177: 184-188, 1999
Woods M: Patients’ perceptions of breast cancer related lymphoedema. Eur J Cancer Care 2: 125-128, 1993
Mirolo B, Bunce I, Chapman M, Eliadis P, Henessy J: Psychosocial benefits of post mastectomy lymphoedema therapy. Cancer Nurs 18: 197-205, 1995
Brady M, Cella D, Mo F: Reliability and validity of the functional assessment of cancer therapy-breast [FACT-B] quality of life instrument. J Clin Oncol 15: 974-986, 1997
Fallowfield L, Leaity S, Howell A, Benson S, Cella D: Assessment of quality of life in women undergoing hormonal therapy for breast cancer: validation of an endocrine symptom subscale for the FACT-B. Breast Cancer Res Treat 55: 189-199, 1999
Aitken R: Arm morbidity within a trial of mastectomy and either nodal sample with selective radiotherapy or axillary clearance. Br J Surg 76: 568-571, 1989
Chetty U, Jack W, Prescott R, Tyler C, Rodger A: Management of the axilla in operable breast cancer treated by breast conservation: a randomised clinical trial. Edinburgh Breast Unit. Br J Surgery 87(2): 163-169, 2000
ALMANAC: A randomised trial of sentinel node guided axillary therapy compared with standard axillary therapy in early breast cancer. Protocol MRC London 1999
Nunnally J: Psychometric Theory. 2nd edn, McGraw-Hill, New York, 1978
Bonomi A, Cella D, Hahn E: Multilingual translation of the functional assessment of cancer therapy [FACT] quality of life measurement system. Qual Life Res 5: 309-329, 1996
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Coster, S., Poole, K. & Fallowfield, L.J. The validation of a quality of life scale to assess the impact of arm morbidity in breast cancer patients post-operatively. Breast Cancer Res Treat 68, 273–282 (2001). https://doi.org/10.1023/A:1012278023233
Issue Date:
DOI: https://doi.org/10.1023/A:1012278023233