Abstract
Background
Regulatory agencies as well as private organizations pursue programs that advocate patient centricity and emphasize the importance of dialog with patients. Various methods are applied to elicit the preferences of patients regarding the aspects of treatment they lend more importance to. Decisions on treatment choices are critical to patients with lung cancer because of their poor prognosis and the serious trade-off between safety and efficacy in traditional cytotoxic chemotherapy.
Methods
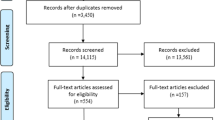
We conducted a systematic literature review of quantitative patient preference studies of patients with lung cancer. Our exhaustive search of MEDLINE, CINAHL, EMBASE, PLOS, and SpringerLink identified 15 relevant studies published from January 2000 to April 2020 that enabled us to assess the relative importance of treatment attributes according to lung cancer patients’ perspective.
Results
The literature review revealed that patients with lung cancer tend to place a higher weight on efficacy and quality of life (QoL) attributes than on other attributes. Overall survival was found to be the most important among the efficacy attributes. The consequences of adverse events seemed less important than the possible efficacy from therapies. The clinical utility of treatment, such as the route of administration, was generally not considered important. It remains inconclusive whether sociodemographic factors and/or medical history affect the relative importance of a patient’s preference.
Conclusion
Our systematic review clarified that patients generally prefer a better efficacy profile to a better safety profile, which underscores the importance of improved benefits in anti-lung cancer drug development.


Similar content being viewed by others
Availability of Data and Material
All data generated or analyzed during this systematic review study are included in this published article.
References
FDA, Patient-Focused Drug Development. https://www.fda.gov/drugs/developmentapprovalprocess/ucm579400.htm. Accessed 2 Jan 2020.
EMA, Patients and Consumers. https://www.ema.europa.eu/en/partners-networks/patients-consumers. Accessed 2 Jan 2020.
DIA, Patient Engagement. https://www.diaglobal.org/en/resources/areas-of-interest/patient-engagement. Accessed 2 Jan 2020.
Zwitter M. Toxicity and quality of life in published clinical trials for advanced lung cancer. Support Care Cancer. 2018;26:3453–9.
ICH, Revision of M4E Guideline on Enhancing the Format and Structure of Benefit-Risk Information in ICH. June 2016. https://www.ich.org/products/ctd/ctdsingle/article/m4er1-efficacy.html. Accessed 2 Jan 2020.
FDA, Benefit-risk assessment in drug regulatory decision-making, Draft PDUFA VI Implementation Plan (FY2018-2022). 30 March 2018. https://www.fda.gov/downloads/ForIndustry/UserFees/PrescriptionDrugUserFee/UCM602885.pdf. Accessed 2 Jan 2020.
Schnipper LE, Davidson NE, Wollins DS, Tyne C, Blayney DW, Blum D, et al. American Society of Clinical Oncology Statement: a conceptual framework to assess the value of cancer treatment options. J Clin Oncol. 2015;33:2563–77.
Coplan PM, Noel RA, Levitan BS, Ferguson J, Mussen F. Development of a framework for enhancing the transparency, reproducibility and communication of the benefit-risk balance of medicines. Clin Pharmacol Ther. 2011;89:312–5.
Postmus D, Richard S, Bere N, van Valkenhoef G, Galinsky J, Low E, et al. Individual trade-offs between possible benefits and risks of cancer treatments: results from a stated preferences study with patients with multiple myeloma. Oncologist. 2018;23:44–51.
MDIC, Medical Device Innovation Consortium (MDIC) Patient Centered Benefit-Risk Project Report: A framework for incorporating information on patient preferences regarding benefit and risk into regulatory assessments of new medical technology. 2015. http://mdic.org/wp-content/uploads/2015/05/MDIC_PCBR_Framework_Web.pdf. Accessed 2 Jan 2020.
Gonzalez JM, Johnson FR, Levitan B, Noel R, Peay H. Symposium title: preference evidence for regulatory decisions. Patient. 2018;11:467–73.
Janssens R, Russo S, van Overbeeke E, Whichello C, Harding S, Kubler J, et al. Patient preferences in the medical product life cycle: what do stakeholders think? Semi-structured qualitative interviews in Europe and the USA. Patient. 2019;12:513–26.
Benz HL, Lee TJ, Tsai JH, Bridges JFP, Eggers S, Moncur M, et al. Advancing the use of patient preference information as scientific evidence in medical product evaluation: a summary report of the patient preference workshop. Patient. 2019;12:553–7.
Marshall D, Bridges JF, Hauber B, Cameron R, Donnalley L, Fyie K, Johnson FR. Conjoint analysis applications in health—how are studies being designed and reported? An update on current practice in the published literature between 2005 and 2008. Patient. 2010;3:249–56.
Soekhai V, de Bekker-Grob EW, Ellis AR, Vass CM. Discrete choice experiments in health economics: past, present and future. Pharmacoeconomics. 2019;37:201–26.
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359–86.
Devesa SS, Bray F, Vizcaino AP, Parkin DM. International lung cancer trends by histologic type: male:female differences diminishing and adenocarcinoma rates rising. Int J Cancer. 2005;117:294–9.
Hanna N, Johnson D, Temin S, Baker S Jr, Brahmer J, Ellis PM, et al. Systemic therapy for stage IV non-small-cell lung cancer: American Society of Clinical Oncology Clinical Practice Guideline update. J Clin Oncol. 2017;35:3484–515.
Planchard D, Popat S, Kerr K, Novello S, Smit EF, Faivre-Finn C, et al. Metastatic non-small cell lung cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018;29:iv192–237.
Blinman P, Alam M, Duric V, McLachlan SA, Stockler MR. Patients’ preferences for chemotherapy in non-small-cell lung cancer: a systematic review. Lung Cancer. 2010;69:141–7.
Blinman P, McLachlan SA, Nowak AK, Duric VM, Brown C, Wright G, et al. Lung cancer clinicians’ preferences for adjuvant chemotherapy in non-small-cell lung cancer: what makes it worthwhile? Lung Cancer. 2011;72:213–8.
Wilson SR, Strub P, Buist AS, Knowles SB, Lavori PW, Lapidus J, et al. Shared treatment decision making improves adherence and outcomes in poorly controlled asthema. Am J Respir Crit Care Med. 2010;181:566–77.
Von Korff M, Katon W, Rutter C, Ludman E, Simon G, Lin E, Bush T. Effect on disability outcomes of a depression relapse prevention program. Psychosom Med. 2003;65:938–43.
Mokhles S, Nuyttens JJME, de Mol M, Aerts JGJV, Maat APWM, Birim O, et al. Treatment selection of early stage non-small cell lung cancer: the role of the patient in clinical decision making. BMC Cancer. 2018;18:79.
Schmidt K, Damm K, Prenzier A, Golpon H, Welte T. Preferences of lung cancer patients for treatment and decision-making: a systematic literature review. Eur J Cancer Care. 2016;25:580–91.
Bridges JF, Hauber AB, Marshall D, Lloyd A, Prosser LA, Regier DA, et al. Conjoint analysis applications in health—a checklist: a report of the ISPOR Good Research Practices for Conjoint Analysis Task Force. Value Health. 2011;14:403–13.
Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA Statement. J Clin Epidemiol. 2009;62:1006–12.
Kind P, Macran S. Eliciting social preference weights for functional assessment of cancer therapy-lung health states. Pharmacoeconomics. 2005;23:1143–53.
Johnson FR, Hauber AB, Osoba D, Hsu MA, Coombs J, Copley-Merriman C. Are chemotherapy patients’ HRQoL importance weights consistent with linear scoring rules? A stated-choice approach. Qual Life Res. 2006;15:285–98.
Nafees B, Stafford M, Gavriel S, Bhalla S, Watkins J. Health state utilities for non small cell lung cancer. Health Qual Life Outcomes. 2008;6:84.
Girones R, Torregrosa D, Gomez-Codina J, Maestu I, Tenias JM, Rosell R. Lung cancer chemotherapy decisions in older patients: the role of patient preference and interactions with physicians. Clin Transl Oncol. 2012;14:183–9.
Bridges JF, Mohamed AF, Finnern HW, Woehl A, Hauber AB. Patients’ preferences for treatment outcomes for advanced non-small cell lung cancer: a conjoint analysis. Lung Cancer. 2012;77:224–31.
Miller PJ, Balu S, Buchner D, Walker MS, Stepanski EJ, Schwartzberg LS. Willingness to pay to prevent chemotherapy induced nausea and vomiting among patients with breast, lung, or colorectal cancer. J Med Econ. 2013;16:1179–80.
Muhlbacher AC, Bethge S. Patients’ preferences: a discrete-choice experiment for treatment of non-small-cell lung cancer. Eur J Health Econ. 2015;16:657–70.
Lehman M, Gorayski P, Watson S, Edeling D, Jackson J, Whitty J. Patient preferences regarding prophylactic cranial irradiation: a discrete choice experiment. Radiother Oncol. 2016;121:225–31.
Tong BC, Wallace S, Hartwig MG, D’Amico TA, Huber JC. Patient preferences in treatment choices for early-stage lung cancer. Ann Thorac Surg. 2016;102:1837–44.
Fallowfield LJ, Catt SL, May SF, Matthews L, Shilling VM, Simcock R, et al. Therapeutic aims of drugs offering only progression-free survival are misunderstood by patients, and oncologists may be overly optimistic about likely benefits. Support Care Cancer. 2017;25:237–44.
Schmidt K, Damm K, Vogel A, Golpon H, Manns MP, Welte T, von der Schulenburg GJM. Therapy preferences of patients with lung and colon cancer: a discrete choice experiment. Patient Prefer Adherence. 2017;11:1647–56.
Bridges JF, Cruz M, Pavilack M, Flood E, Janssen EM, Chehab N, et al. Patient preferences for attributes of tyrosine kinase inhibitor treatments for EGFR mutation-positive non-small-cell lung cancer. Future Oncol. 2019;15:3895–907.
Sullivan DR, Eden KB, Dieckmann NF, Golden SE, Vranas KC, Nugent SM, et al. Understanding patients’ values and preferences regarding early stage lung cancer treatment decision making. Lung Cancer. 2019;131:47–57.
Sun H, Wang H, Xu N, Li J, Shi J, Zhou N, et al. Patient preferences for chemotherapy in the treatment of non-small cell lung cancer: a multicenter discrete choice experiment (DCE) study in China. Patient Prefer Adherence. 2019;13:1701–9.
Valenti V, Ramos J, Perez C, Capdevila L, Ruiz I, Tikhomirova L, et al. Increased survival time or better quality of life? Trade-off between benefits and adverse events in the systemic treatment of cancer. Clin Transl Oncol. 2020;22:935–42.
Camps C, del Pozo N, Blasco A, Blasco P, Siera R. Importance of quality of life in patients with non-small-cell lung cancer. Clin Lung Cancer. 2009;10:83–90.
Hopper AN, Jamison MH, Lewis WG. Learning curves in surgical practice. Postgrad Med J. 2007;83:777–9.
Von Arx LB, Kjeer T. The patient perspective of diabetes care: a systematic review of stated preference research. Patient. 2014;7:283–300.
McCarthy MC, De Abreu Lourenco R, McMillan LJ, Meshcheriakova E, Cao A, Gillam L. Finding out what matters in decision-making related to genomics and personalized medicine in pediatric oncology: developing attributes to include in a discrete choice experiment. Patient. 2020;13:347–61.
Louis E, Ramos-Goni JM, Cuervo J, Kopylov U, Barreiro-de Acosta M, McCartney S, et al. A qualitative research for defining meaningful attributes for the treatment of inflammatory bowel disease from the patient perspective. Patient. 2020;13:317–25.
Hollin IL, Craig BM, Coast J, Beusterien K, Vass C, DiSantostefano R, et al. Reporting formative qualitative research to support the development of quantitative preference study protocols and corresponding survey instruments: guidelines for authors and reviewers. Patient. 2020;13:121–36.
Author information
Authors and Affiliations
Contributions
YS and SO contributed to the study conception and design. Material preparation, data collection, and analysis were performed by YS. The independent systematic review was performed by NS. The first draft of the manuscript was written by YS, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
YS and NS are full-time employees at Chugai Pharmaceutical Co., Ltd. All authors have no other relevant conflicts of interest to disclose.
Funding
This work was conducted as part of the authors'employment with their organisations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix
Appendix
The literature search strategies for the databases are detailed below. All searches were conducted on June 3, 2020.
-
a.
MEDLINE
(“conjoint analysis”[All Fields] OR “conjoint analyses”[All Fields] OR “choice behavior”[All Fields] OR “stated preference”[All Fields] OR “discrete choice”[All Fields] OR “latent class analysis”[All Fields] OR “latent class analyses”[All Fields]) AND “Lung Neoplasms”[MeSH] AND (“2000/01/01”[PDAT]: “2020/04/30”[PDAT])
-
b.
CINAHL
Published date ranged from 20000101 to 20200430 and (“conjoint analysis” OR “conjoint analyses” OR “choice behavior” OR “stated preference” OR “discrete choice” OR “latent class analysis” OR “latent class analyses”) AND (“lung neoplasms” OR “lung tumor” OR “lung cancer” OR “lung tumour”)
-
c.
EMBASE
(“conjoint analysis” OR “conjoint analyses” OR “choice behavior” OR “stated preference” OR “discrete choice” OR “latent class analysis” OR “latent class analyses”) AND (“lung tumor”/exp OR “lung tumor”) AND [2000-2020]/py
-
d.
PLOS
Published date ranged from 2000-01-01 to 2020-04-30 and (everything:”conjoint analysis” OR everything:”conjoint analyses” OR everything:”choice behavior” OR everything:”stated preference” OR everything:”discrete choice” OR everything:”latent class analysis” OR everything:”latent class analyses”) AND (everything:”lung neoplasms” OR everything:”lung tumor” OR everything:”lung cancer” OR everything:”lung tumour”)
-
e.
SpringerLink
Published date ranged from 2000 to 2020 and (“conjoint analysis” OR “choice behavior” OR “stated preference” OR “discrete choice” OR “latent class analysis”) AND (“lung neoplasms” OR “lung tumor” OR “lung cancer” OR “lung tumour”).
Rights and permissions
About this article
Cite this article
Sugitani, Y., Sugitani, N. & Ono, S. Quantitative Preferences for Lung Cancer Treatment from the Patients’ Perspective: A Systematic Review. Patient 13, 521–536 (2020). https://doi.org/10.1007/s40271-020-00434-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40271-020-00434-7