Abstract
Purpose of Review
With the growing obesity epidemic among children and adolescents, the evaluation of disease origin to slow disease progression is necessary. Racial disparities which are evident amid prevalence and treatment must be studied to counteract disease propagation.
Recent Findings
Disparities are pronounced among Black and Hispanic pediatric patients prior to conception and birth due to genetic composition and fetal environment. Postnatal environment and psychosocial influences can further increase a child/adolescent’s propensity to increased weight. Current treatment options including nutrition, physical activity, behavior modification, pharmacotherapy, and surgery are underutilized in communities of color due to limited access to care and cost.
Summary
Data is limited to demonstrate disparities among treatment of obesity in children and adolescents. The reviewed studies show the role of race on disease treatment. Increased research efforts, especially in pharmacotherapy and metabolic and bariatric surgery (MBS), will help combat obesity in pediatric communities of color.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obesity in children and adolescents is the most prevalent chronic disease which affects approximately 18.5% of US children age 2-17 years old [1]. Accompanying the increased rate of obesity is the rising presence of multiple medical conditions including hypertension, type 2 diabetes, and non-alcoholic liver disease. Because a child or adolescent with obesity is 70% more likely to struggle with their weight as an adult [2], it is imperative to diagnose and treat obesity in pediatric patients to prevent progression into adulthood [3, 4].
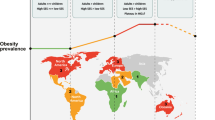
Obesity disproportionately affects racial minority groups, especially Hispanic and Black children and adolescents [2, 5]. Asian American children and adolescents have the lowest rate of obesity [5]. The most recent National Health and Nutrition Examination (NHANES) survey from 2015-2016 demonstrated the prevalence of class I obesity, defined as body mass index (BMI) greater than 95th percentile but less than the 99th percentile or greater than 30 kg/m2 (whichever is lower), was highest among Black females at 25.1% and Hispanic males at 20% in comparison to compared to 13.6% and 14.7% in White females and males, respectively [5]. In addition, the rate of severe obesity, characterized as BMI greater than 99th percentile, is higher among Black and Hispanic children and adolescents [5]. Obesity is a multifactorial disease with many contributing etiologies. This literature review is comprised of the most recent studies found on PubMed regarding the origin and management of obesity in children and adolescents. Because racial disparities contribute to the obesity epidemic at all ages, early identification of these inequalities within children and adolescents is imperative to effectively treat obesity.
Risk Factors for Obesity in Children and Adolescents in Racially Diverse Populations
Genetics
While obesity is often found to “run in families,” the specific genes that make a child more at risk for obesity is difficult to ascertain as environment clearly plays a role [6]. Racial disparities in obesity have prompted exploration for the genetic basis of these differences. Genome-wide association studies (GWAS) have found 551 genetic loci associated with obesity, including the most studied loci, FTO (fat mass and obesity related gene) [7]. However, most of the studies are conducted in participants of European decent [8]. Whenever FTO variants were studied in patients of African background or Black, the association with obesity was often not correlated [8]. There are no current studies to evaluate FTO variants in other racial minorities.
The African Ancestry Anthropometric Genetics Consortium (AAAGC) is a GWAS created to identify specific alleles for obesity related traits in those of African descent and to map loci originally associated in those of European ancestry [9]. After three stages of evaluation, less than 20 variants were discovered that are closely related to obesity, including a BMI variant single nucleotide polymorphism (SNP), rs16951275 on chromosome 5 and a cis-eQTL variant which is important in regulation of gene expression in subcutaneous and visceral adipose tissue [9]. Other GWAS like the Population Architecture using Genomics and Epidemiology (PAGE) and HiSpanic/Latino Anthropometry (HISLA) Consortium include Hispanic and Latino Americans to determine genetic etiologies of obesity [9]. There are 14 genotyped BMI loci including ATP2A1, COBLL1, MAP2K5, POC5, SLC22A3, and TCF7L2 specifically present in Blacks [9]. The SNP, rs76493495 is another BMI variant found in Blacks and Hispanic/Latino Americans [9].
There are currently minimal studies evaluating racial disparities involved with specific monogenetic causes including Prader Willi Syndrome, leptin deficiency, and melanocortin 4 receptor (MC4R) deficiency [6]. However, one study evaluated a cohort of 312 children with severe early-onset obesity of Black or Latino ancestry and showed the prevalence of MC4R variants was 2.6 %, comparable to that of studies among children of European ancestry [10]. More investigations are needed to fully evaluate the genetic causes of obesity in children and adolescents.
Prenatal Factors
Maternal smoking during pregnancy has been associated with increased rates of childhood obesity [11,12,13,14]. One Centers for Disease Control and Prevention (CDC) report showed the percentage of self-reported habitual smokers was highest among American Indian/Native Alaskan populations (22.6%) compared to Non-Hispanic White Americans (15.0%) and Non-Hispanic Black Americans (14.6%)[15]. These results are comparable to another study conducted on across racial groups mentioned, as American Indian/Native Alaskan 4 years old were 10% more likely to have obesity [16]. Despite documented differences, women with less than a high school education level have an increased tendency to smoke throughout pregnancy [17].
Multiple studies have shown higher pre-pregnancy BMI and significant gestational weight gain correlates with an increased risk of obesity in children [18,19,20]. One particular study analyzed data from the Supplemental Nutrition Assistance Program (SNAP) in Wisconsin and found that for every 1 kg/m2 increase in pre-pregnancy BMI, there was a 4.5% increase risk of obesity when compared to mothers with normal BMI [21]. Another study showed mothers who underwent Roux-en-Y gastric bypass (RYGB) surgery prior to pregnancy were less likely to deliver a large-for-gestational-age (LGA) child in comparison to mother’s with obesity[22]. LGA neonates are more likely to develop childhood obesity later in development [23,24,25,26]. There is minimal evidence to illustrate variation among racial minorities. However, Mehta and colleagues specifically evaluated LGA and the development of childhood obesity among Blacks living in the inner-city; LGA newborns were 2.5 times more likely to have childhood obesity than average size newborns [24].
Perinatal Factors
Research shows that duration of breastfeeding is inversely related to risk of childhood obesity [27]. One study found that infants who were breastfed for more than seven months were significantly less likely to develop overweight or obesity in their adolescence than those who were breastfed for less than 3 months [27]. In fact, breastfed infants were 22% less likely to struggle with obesity compared to those who were not breastfed at all [27]. There is mixed reasoning to explain this causation. While some evidence suggest the greater nutritional density in breast milk, prompts infants to feel more satiated with breast milk than formula, others have shown that the ability and time to breastfeed is indicative of a mother who is in a higher income bracket [27, 28]. The United States Bureau of Labor Statistics reported Black and Hispanic women made the least amount of money in the USA [29]. Multiple reports have noted non-Hispanic Black children are 20% less likely than White children to be breastfed [30, 31]. Black women are also more likely to introduce formula earlier than other racial groups [32]. However, there is no consistent finding to support early introduction of solid foods is associated with increase prevalence of obesity [33]. More research must be done to conclude any correlation or causation between childhood obesity and introduction to solid foods.
Infancy and Childhood Factors
Nutrition
Intake of sugar-sweetened beverages among school aged children is associated with increased BMI [34, 35]. Multiple studies show an increased consumption of sugar-sweetened beverages among Black and Hispanic children in comparison to White children [35,36,37,38]. In addition, several studies demonstrate increased fast food intake among Black and Hispanic children compared to White children [30, 36, 39, 40]. Because higher consumption of fast food is associated with increased portion sizes [41], poor nutritional quality [42], and higher energy density, there is a definite relationship with obesity [43].
Consumption of increased processed foods among Hispanic and Black families from lower income is likely a result of the increased cost of healthier foods [44]. Studies demonstrate that healthy foods with high levels nutritional quality are far more expensive and less affordable than foods that contain added sugars, trans fats, or saturated fats [45]. Food insecurity defined as “having inadequate access to sufficient, safe, and nutritious food to meet dietary needs and food preferences for an active and healthy lifestyle” is also a contributor of increased obesity among children from low income families as there is often limited access to fresh produce and whole grains [46].
Having family meals has been shown to be protective against development of obesity due to higher quality diet in addition to other psychosocial benefits [47]. An investigation by Skala and colleagues examined racial differences in the home food environment among low-income, Black and Hispanic families of preschoolers [48]. Hispanic parents had more family-oriented meals and increased access to fresh fruits and vegetables compared to Black parents who were more likely to restrict and reward with dessert during meals, which is known to contribute to increased obesity prevalence [48].
Behavior
Parenting practices on children’s bedtime have been shown to affect BMI in all racial groups [49]. Nevarez and colleagues showed that at two years of age, Black, Hispanic, and Asian infants slept fewer hours per day compared to White children [50]. Insufficient sleep in children can be related to several factors including sleeping with a television in the bedroom [36], sleeping near a small screen and having more screen time [51] and a later bedtime routine. Past studies have shown later bedtimes higher levels of television viewing and more television watching in the bedrooms among Black and Hispanic children as compared to White children [52, 53].
Several studies have shown lower levels of physical activity among Black and Hispanic youth compared to White youth [54, 55]. The 5-2-1-0 counseling tool is a nationally recognized pediatric obesity prevention program to promote healthy behaviors around nutrition, physical activity, and screen time in children from birth to 18 years of age [56]. In a cross-sectional national study using the 2011-2012 NHANES data, Haughton and colleagues showed that non-Hispanic Black children were less likely to meet the 5-2-1-0 nutrition and activity targets compared to their non-Hispanic counterparts [56]. Also, Hispanic adolescents were less likely to meet the targets compared to their non-Hispanic White counterparts [56].
Environment
Psychosocial stress including perceived discrimination, parental stress and stigma plays a significant role in obesity prevalence [57]. One study conducted by Heerman and colleagues reviewed survey results from the CDC National Survey of Children’s Health (NSCH) to assess if there was a relationship between adverse family experiences (AFEs) during childhood and the development of adolescent obesity [58]. Children with a history of AFEs were more likely to have overweight or obesity [58]. More specifically, children from age 10-17 years old who experienced greater than 2 AFE have 80% higher odds of obesity [58]. Unfortunately, Heerman and colleagues did not specifically evaluate this finding among children and adolescents of different racial groups.
Childcare arrangements in infancy may also increase risk of obesity in childhood and beyond, though the results are inconclusive [59, 60]. Black and White families are more likely to use a center-based childcare care compared to Hispanic families, who tend to rely on their family for child care responsibilities, particularly in infancy and toddlerhood [61, 62]. Many preschool aged children are not meeting their dietary and physical activity recommendations in a childcare center which could be contributing to obesity [63,64,65,66].
Treatment
Nutrition
Nutrition-based interventions include increasing access to fruits and vegetables, limiting intake of sugar-sweetened beverages, minimizing fat content in meals, adjusting nutrition standards in schools, and educating families about healthy nutrition practices [67,68,69]. Thus far, the efficacy of such treatments has been limited, showing only modest success in reducing BMI and other obesity related factors [67, 68, 70]. Multi-component interventions demonstrate similar results, although research suggests programs with environmental components have the potential to produce larger, long-term changes [68, 71,72,73]. This evidence emphasizes the need for additional research to determine if nutrition-related treatment options could provide longer-lasting positive health outcomes in children and adolescents with obesity.
Although some nutrition-based interventions have targeted childhood obesity, few have focused on addressing racial disparities. One study, designed for low-income students, implemented a multi-component program in which researchers modified school meals and developed a nutrition education course to evaluate overall health and academic achievement [74]. During the two-year trial, children in the intervention schools were more likely to have higher math score on the Florida Comprehensive Achievement Test and displayed significant weight decreases [74]. These findings emphasize the efficacy of such an approach to prevent obesity among low-income children, many of whom were Black and Hispanic [74].
SNAP, a federal nutrition assistance program, aims to improve dietary quality among its participants, many of whom are racial minorities [75]. Grummon and Taillie used information from the US Homescan Consumer Panel dataset to describe racial disparities in purchase outcomes in SNAP participants and non-participants. Their analysis determined Black individuals, both participants and nonparticipants, which purchased greater amounts of meats, sweeteners, sugar-sweetened beverages, sugar, and sodium. Black SNAP participants purchased more junk food, and SNAP participation in Black households was associated with increased calories purchased [75]. Overall, these data suggest White individuals had better dietary quality than other racial minorities, particularly Black participants. Therefore, SNAP may not alleviate existing racial disparities in dietary behaviors [75]. While this analysis did not directly evaluate dietary patterns in children and adolescents, the SNAP demographic characteristics accounted for household size, suggesting children are indirectly analyzed. Furthermore, studies evaluating food consumption in children and adolescents demonstrate similar trends regarding unhealthy eating habits [76, 77].
The lack of adequate research addressing nutrition paired with the increased risk of developing obesity in racial minorities emphasizes the urgency of identifying effective treatments. Future interventions must consider the myriad of factors that may contribute to unhealthy eating habits seen in racial minorities. For instance, inaccessibility of supermarkets, lack of fruits and vegetables, availability of fast food restaurants, proximity to convenient stores, and neighborhood socioeconomic status are known to contribute to unhealthy eating habits and the onset of childhood obesity [78,79,80,81]. By understanding the relationship between these factors, researchers can establish interventions tailored to the specific needs of children and adolescents from varying demographic backgrounds.
Physical Activity
Regular physical activity is known to improve weight status in children, making it a common lifestyle intervention for treating childhood obesity [82, 83]. Programs targeting physical activity aim to decrease sedentary behavior, increase time spent engaging in moderate-to-vigorous physical activity, and educate families about the benefits of staying active [82, 84, 85]. Such interventions are effective, to some extent, in improving body mass index (BMI) and preventing obesity [69, 82, 86, 87]. Increased physical activity is also associated with improved bone health, cardiometabolic health, cognition, and reduced risk of depression in children and adolescents [83]. Thus, treatments addressing physical activity have additional health benefits beyond reducing weight status, further elucidating their usefulness. Although programs evaluating physical activity have demonstrated some efficacy, research suggests a multi-level approach is most beneficial [69, 88, 89]. This method often involves behavior modification alongside education curriculums in which both physical activity and nutrition are targeted.
Considering certain racial minorities have an increased risk of developing childhood obesity [5, 76, 90, 91], it is critical for researchers to develop treatments which can minimize such disparities. Some research has successfully targeted physical activity to treat childhood obesity. However, the number of studies is limited and fail to show consistent or long-term changes in positive health outcomes. One study developed a school-based aerobic exercise program for Black and Hispanic adolescents [92]. After the 12-week intervention, females and males exhibited a decrease in BMI and resting heart rate, although the changes seen in males were not statistically significant [92].
A similar program was designed to analyze the impact of moderate-to-vigorous physical activity on body composition and cardiovascular fitness in Black girls [93]. Results indicated a decrease in percent body fat and BMI in both normal weight and overweight girls [93]. Another study targeted preschool age Black and Hispanic children for obesity prevention by implementing a multi-level program focused on nutrition and physical activity [94]. Black preschoolers in the intervention group demonstrated significantly smaller relative changes in BMI while Latino preschoolers did not [94, 95]. Based on the results of this study, researchers adjusted their methodology to include a curriculum for parents. At the 1-year follow-up, Latino preschoolers, both in the experimental group and control group, had lower BMI and BMI Z-scores [96]. Despite this, the obesity rate in the study sample was still somewhat higher compared to a nationally representative sample [96]. Although these interventions produced varied results, they illustrate potential benefits of targeting physical activity and implementing health education courses to address obesity in children of racial minorities. Furthermore, they underscore the importance of developing programs tailored to specific demographics that consider cultural differences [94]. Overall, the lack of clinically significant, long-term effects suggests a need for the development of additional treatment options as a way to mitigate the effects of racial disparities in childhood and adolescent obesity.
Behavioral Modification
Behavioral therapy is another mainstay of weight management that includes collaboration between patients and their patient care team including physicians, nurses, and nutritionists. There are different types of behavioral therapy including cognitive behavior therapy (CBT) which encourages children and adolescents to self-regulate their nutrition and physical activity [97]. Within these sessions, patients can expect to modify their behaviors through goal setting, accountability, self-monitoring, stimulus control, problem solving, and meal planning [98].
The Behavioral Choice Treatment (BCT) program is a group-based intervention that helps patients view eating and exercise as a choice [98]. This program has shown significant long-term weight loss within primary care settings and has demonstrated success among Black patients [98].
Weight loss programs that are family-focused are found to have greater success than individualized programs. Studies have shown that family-based weight loss interventions lead to a significant decrease in percent overweight compared to those utilizing the standard of care [99,100,101,102,103,104] and that family participation and cohesion within Black patients leads to increased weight loss [105]. However, a recent review by Ash and colleagues showed that while Hispanic American families are widely represented in studies evaluating the efficacy of family-based interventions, Black/African-American families are historically underrepresented [106].
Several racial disparities exist in terms of access to behavioral therapy and success within family-based weight loss treatment programs. Behavioral therapy is most effective when conducted in person for at least fourteen sessions within a 6-month period [98]. However, due to issues with primary care provider (PCP) communication, differences in social class perception, and tendency for making same-day appointments, attending these necessary appointments is difficult for Black and Hispanic families [107]. Further, successful family-based interventions require access to parks and open spaces for maximized weight loss, but Black and Hispanic families have limited access to these resources given their tendency to live within more urban communities. Therefore, although studies have shown promising weight loss success when behavioral modification is utilized, racial lifestyle differences can significantly impact meaningful weight loss within Black and Hispanic communities compared to their White counterparts.
Pharmacotherapy
Pharmacotherapy is an underutilized option in treatment of obesity in children and adolescents with BMI ≥95th percentile with a comorbidity or BMI ≥120th percentile of 95th percentile [108]. The most commonly prescribed anti-obesity medications (AOMs) in children and adolescents include orlistat, FDA approved in children over 12 years old and phentermine, FDA approved in adolescents over the age of 16 [108] . As of December 2020, Setmelanotide and Liraglutide 3.0 mg are additional medications FDA approved for treatment of obesity in children and adolescents. Other medications, including metformin, phentermine-topiramate, and bupropion-naltrexone, are prescribed for treatment of obesity in children and adolescents off-label.
Pharmacotherapy is underutilized in pediatric populations and, if prescribed, is often only used if care is received within a multidisciplinary obesity care center. This is especially evident among Hispanic and Black children and adolescents who are mainly treated for obesity in a primary care center as referral to advanced level of care is limited. The cost of AOMs is a factor in considering use among patients from lower socioeconomic backgrounds, which disproportionately affects Latino and Black patients. Though a number of private insurance companies cover weight loss medications, only seven states provide coverage through Medicaid: Alabama, North Dakota, South Carolina, South Dakota, Virginia, Wisconsin, and Delaware [109]. In addition, some AOMs including orlistat have been shown to be less effective in Blacks in comparison to Whites [2]. The efficacy of other medications between racial groups has not been studied [2]. Nevertheless, the reason for these weight loss differences remains unclear but must be considered when it comes to optimizing weight loss pharmacotherapy.
Metabolic and Bariatric Surgery
Metabolic and bariatric surgery (MBS), including Roux-en-Y gastric bypass (RYGB) and vertical sleeve gastrectomy (VSG), is the most effective tool available to treat moderate to severe obesity in children and adolescents [110]. Leading medical organizations including the American Academy of Pediatrics (AAP) and the American Society of Metabolic and Bariatric Surgery (ASMBS) support early referral for MBS consideration in patients with a BMI ≥ 120% of the 95th percentile with an associated comorbidity or BMI ≥ 140% of the 95th percentile [110, 111]. MBS continues to be underused, especially in racial minorities [112].
Nunez Lopez and colleagues conducted a retrospective review of adolescent bariatric surgery using the Bariatric Outcome Longitudinal Database (BOLD). While Black patients are have a higher preoperative BMI and are more likely to have hypertension, obstructive sleep apnea, and asthma prior to surgery, there was no statistical difference in weight loss and resolution of comorbidities when comparing Black and White patient population [113]. However, the authors note that minority adolescents are less likely to undergo bariatric surgery [113].
There are many barriers regarding consideration of MBS among children and adolescents with severe obesity. While access to care is multifactorial, race and socioeconomic status is are important considerations in regard to insurance authorization for surgery [114]. Just under 50% of pediatric patients who qualify for surgery receive insurance coverage initially following request for authorization [114, 115]. While age less than 18 years old is the most common reason for denial, Perez and colleagues have shown authorization for bariatric surgery is most common among White females with private insurance [115]. Black and Hispanic adolescents with Medicaid are less likely to receive authorization in comparison to their White counterparts [115]. The authors hypothesize, and minorities are less likely to appeal following initial denial [115].
To ensure quality and safety, clinical guidelines recommend bariatric surgery should be performed at a pediatric center approved by through the Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program (MBSAQIP) [110]. Unfortunately, there is a lack of these centers throughout the country which contributes to the lack of pediatric providers referring children and adolescents with severe obesity for early surgical consideration. The disproportionate number of centers is even more evident within geographical areas serving minority populations.
Conclusion
Considering the increasing number of Black and Hispanic pediatric patients with obesity and obesity-related conditions, most recently including COVID-19, there is a growing need to treat obesity early and often to halt disease progression into adulthood. Obesity with its biological origins such as genetic makeup is in turn altered by various environmental conditions both prenatally and throughout one’s life. Similar to the adult population, there are clear disparities in the response and utilization of the most effective treatment modalities including pharmacotherapy and bariatric surgery. In order to adequately address the obesity epidemic, there is a need to focus more research efforts specifically on treatment within underserved populations with emphasis in children and adolescents.
References
Hales CM, Fryar CD, Carroll MD, Freedman DS, Ogden CL. Trends in obesity and severe obesity prevalence in US youth and adults by sex and age, 2007-2008 to 2015-2016. Jama. 2018;319(16):1723–5.
Byrd AS, Toth AT, Stanford FC. Racial disparities in obesity treatment. Curr Obes Rep. 2018;7(2):130–8.
Kohut T, Robbins J, Panganiban J. Update on childhood/adolescent obesity and its sequela. Curr Opin Pediatr. 2019;31(5):645–53.
Kelsey MM, Zaepfel A, Bjornstad P, Nadeau KJ. Age-related consequences of childhood obesity. Gerontology. 2014;60(3):222–8.
Skinner AC, Ravanbakht SN, Skelton JA, Perrin EM, Armstrong SC. Prevalence of obesity and severe obesity in US children, 1999-2016. Pediatrics. 2018;141(3).
Brown CL, Halvorson EE, Cohen GM, Lazorick S, Skelton JA. Addressing childhood obesity: opportunities for prevention. Pediatr Clin N Am. 2015;62(5):1241–61.
Loos RJ, Yeo GS. The bigger picture of FTO: the first GWAS-identified obesity gene. Nat Rev Endocrinol. 2014;10(1):51–61.
Adeyemo A, Chen G, Zhou J, Shriner D, Doumatey A, Huang H, et al. FTO genetic variation and association with obesity in West Africans and African Americans. Diabetes. 2010;59(6):1549–54.
Young KL, Graff M, Fernandez-Rhodes L, North KE. Genetics of obesity in diverse populations. Curr Diab Rep. 2018;18(12):145.
De Rosa MC, Chesi A, McCormack S, Zhou J, Weaver B, McDonald M, et al. Characterization of rare variants in MC4R in African American and Latino children with severe early-onset obesity. J Clin Endocrinol Metab. 2019;104(7):2961–70.
Albers L, Sobotzki C, Kuß O, Ajslev T, Batista RF, Bettiol H, et al. Maternal smoking during pregnancy and offspring overweight: is there a dose-response relationship? An individual patient data meta-analysis. Int J Obes. 2018;42(7):1249–64.
von Kries R, Toschke AM, Koletzko B, Slikker W Jr. Maternal smoking during pregnancy and childhood obesity. Am J Epidemiol. 2002;156(10):954–61.
Leary SD, Smith GD, Rogers IS, Reilly JJ, Wells JC, Ness AR. Smoking during pregnancy and offspring fat and lean mass in childhood. Obesity (Silver Spring). 2006;14(12):2284–93.
Magriplis E, Farajian P, Panagiotakos DB, Risvas G, Zampelas A. Maternal smoking and risk of obesity in school children: investigating early life theory from the GRECO study. Prev Med Rep. 2017;8:177–82.
National Survey on Drug Use and Health, 2016. Public-use data and file documentation: National Center for Health Statistics; [cited 2020 October 19, 2020]. Available from: https://www.samhsa.gov/data/sites/default/files/NSDUH-DetTabs-2016/NSDUH-DetTabs-2016.pdf
Anderson SE, Whitaker RC. Prevalence of obesity among US preschool children in different racial and ethnic groups. Arch Pediatr Adolesc Med. 2009;163(4):344–8.
Kondracki AJ. Prevalence and patterns of cigarette smoking before and during early and late pregnancy according to maternal characteristics: the first national data based on the 2003 birth certificate revision, United States, 2016. Reprod Health. 2019;16(1):142.
Voerman E, Santos S, Patro Golab B, Amiano P, Ballester F, Barros H, et al. Maternal body mass index, gestational weight gain, and the risk of overweight and obesity across childhood: an individual participant data meta-analysis. PLoS Med. 2019;16(2):e1002744.
Williams CB, Mackenzie KC, Gahagan S. The effect of maternal obesity on the offspring. Clin Obstet Gynecol. 2014;57(3):508–15.
Josey MJ, McCullough LE, Hoyo C, Williams-DeVane C. Overall gestational weight gain mediates the relationship between maternal and child obesity. BMC Public Health. 2019;19(1):1062.
Ohlendorf JM, Robinson K, Garnier-Villarreal M. The impact of maternal BMI, gestational weight gain, and breastfeeding on early childhood weight: analysis of a statewide WIC dataset. Prev Med. 2019;118:210–5.
Adams TD, Hammoud AO, Davidson LE, Laferrère B, Fraser A, Stanford JB, et al. Maternal and neonatal outcomes for pregnancies before and after gastric bypass surgery. Int J Obes. 2015;39(4):686–94.
Derraik JGB, Maessen SE, Gibbins JD, Cutfield WS, Lundgren M, Ahlsson F. Large-for-gestational-age phenotypes and obesity risk in adulthood: a study of 195,936 women. Sci Rep. 2020;10(1):2157.
Mehta SH, Kruger M, Sokol RJ. Being too large for gestational age precedes childhood obesity in African Americans. Am J Obstet Gynecol. 2011;204(3):265.e1-5.
Gillman MW, Rifas-Shiman S, Berkey CS, Field AE, Colditz GA. Maternal gestational diabetes, birth weight, and adolescent obesity. Pediatrics. 2003;111(3):e221–6.
Martin CL, Siega-Riz AM, Sotres-Alvarez D, Robinson WR, Daniels JL, Perrin EM, et al. Maternal dietary patterns during pregnancy are associated with child growth in the first 3 years of life. J Nutr. 2016;146(11):2281–8.
Azad MB, Vehling L, Chan D, Klopp A, Nickel NC, McGavock JM, et al. Infant feeding and weight gain: separating breast milk from breastfeeding and formula from food. Pediatrics. 2018;142(4):e20181092.
Institute of Medicine Committee on the Evaluation of the Addition of Ingredients New to Infant F. Infant Formula: Evaluating the Safety of New Ingredients. Washington (DC): National Academies Press (US) Copyright 2004 by the National Academy of Sciences. All rights reserved.; 2004.
Labor USDo. Earnings and employment by occupation, race, ethnicyt, and sex, 2010 2004 [Available from: https://www.bls.gov/opub/ted/2011/ted_20110914.htm.
Wang Y, Chen X. How much of racial/ethnic disparities in dietary intakes, exercise, and weight status can be explained by nutrition- and health-related psychosocial factors and socioeconomic status among US adults? J Am Diet Assoc. 2011;111(12):1904–11.
Satia JA. Diet-related disparities: understanding the problem and accelerating solutions. J Am Diet Assoc. 2009;109(4):610–5.
Papoutsou S, Savva SC, Hunsberger M, Jilani H, Michels N, Ahrens W, et al. Timing of solid food introduction and association with later childhood overweight and obesity: the IDEFICS study. Matern Child Nutr. 2018;14(1).
Barrera CM, Perrine CG, Li R, Scanlon KS. Age at introduction to solid foods and child obesity at 6 years. Child Obes. 2016;12(3):188–92.
Ludwig DS, Peterson KE, Gortmaker SL. Relation between consumption of sugar-sweetened drinks and childhood obesity: a prospective, observational analysis. Lancet. 2001;357(9255):505–8.
Wang YC, Bleich SN, Gortmaker SL. Increasing caloric contribution from sugar-sweetened beverages and 100% fruit juices among US children and adolescents, 1988-2004. Pediatrics. 2008;121(6):e1604–14.
Taveras EM, Gillman MW, Kleinman K, Rich-Edwards JW, Rifas-Shiman SL. Racial/ethnic differences in early-life risk factors for childhood obesity. Pediatrics. 2010;125(4):686–95.
Giammattei J, Blix G, Marshak HH, Wollitzer AO, Pettitt DJ. Television watching and soft drink consumption: associations with obesity in 11- to 13-year-old schoolchildren. Arch Pediatr Adolesc Med. 2003;157(9):882–6.
Welsh JA, Cogswell ME, Rogers S, Rockett H, Mei Z, Grummer-Strawn LM. Overweight among low-income preschool children associated with the consumption of sweet drinks: Missouri, 1999-2002. Pediatrics. 2005;115(2):e223–9.
Bowman SA, Gortmaker SL, Ebbeling CB, Pereira MA, Ludwig DS. Effects of fast-food consumption on energy intake and diet quality among children in a national household survey. Pediatrics. 2004;113(1 Pt 1):112–8.
Piernas C, Popkin BM. Increased portion sizes from energy-dense foods affect total energy intake at eating occasions in US children and adolescents: patterns and trends by age group and sociodemographic characteristics, 1977-2006. Am J Clin Nutr. 2011;94(5):1324–32.
Nielsen SJ, Popkin BM. Patterns and trends in food portion sizes, 1977-1998. Jama. 2003;289(4):450–3.
Taveras EM, Berkey CS, Rifas-Shiman SL, Ludwig DS, Rockett HR, Field AE, et al. Association of consumption of fried food away from home with body mass index and diet quality in older children and adolescents. Pediatrics. 2005;116(4):e518–24.
Ebbeling CB, Pawlak DB, Ludwig DS. Childhood obesity: public-health crisis, common sense cure. Lancet. 2002;360(9331):473–82.
Acheampong I, Haldeman L. Are nutrition knowledge, attitudes, and beliefs associated with obesity among low-income Hispanic and African American women caretakers? J Obes. 2013;2013:123901.
Denny S. Is there research to support the statement that healthy diets cost more? J Acad Nutr Diet. 2012;112(9):1504.
Tester JM, Rosas LG, Leung CW. Food insecurity and pediatric obesity: a double whammy in the era of COVID-19. Curr Obes Rep. 2020;9(4):442–50.
Gillman MW, Rifas-Shiman SL, Frazier AL, Rockett HR, Camargo CA Jr, Field AE, et al. Family dinner and diet quality among older children and adolescents. Arch Fam Med. 2000;9(3):235–40.
Skala K, Chuang RJ, Evans A, Hedberg AM, Dave J, Sharma S. Ethnic differences in the home food environment and parental food practices among families of low-income Hispanic and African-American preschoolers. J Immigr Minor Health. 2012;14(6):1014–22.
Min J, Wen X, Xue H, Wang Y. Ethnic disparities in childhood BMI trajectories and obesity and potential causes among 29,250 US children: findings from the Early Childhood Longitudinal Study-Birth and Kindergarten Cohorts. Int J Obes. 2018;42(9):1661–70.
Nevarez MD, Rifas-Shiman SL, Kleinman KP, Gillman MW, Taveras EM. Associations of early life risk factors with infant sleep duration. Acad Pediatr. 2010;10(3):187–93.
Falbe J, Davison KK, Franckle RL, Ganter C, Gortmaker SL, Smith L, et al. Sleep duration, restfulness, and screens in the sleep environment. Pediatrics. 2015;135(2):e367–75.
Dennison BA, Erb TA, Jenkins PL. Television viewing and television in bedroom associated with overweight risk among low-income preschool children. Pediatrics. 2002;109(6):1028–35.
McLaughlin Crabtree V, Beal Korhonen J, Montgomery-Downs HE, Faye Jones V, O'Brien LM, Gozal D. Cultural influences on the bedtime behaviors of young children. Sleep Med. 2005;6(4):319–24.
Andersen RE, Crespo CJ, Bartlett SJ, Cheskin LJ, Pratt M. Relationship of physical activity and television watching with body weight and level of fatness among children: results from the Third National Health and Nutrition Examination Survey. Jama. 1998;279(12):938–42.
Polacsek M, Orr J, Letourneau L, Rogers V, Holmberg R, O’Rourke K, et al. Impact of a primary care intervention on physician practice and patient and family behavior: keep ME Healthy---the Maine Youth Overweight Collaborative. Pediatrics. 2009;123(Suppl 5):S258–66.
Haughton CF, Wang ML, Lemon SC. Racial/ethnic disparities in meeting 5-2-1-0 recommendations among children and adolescents in the United States. J Pediatr. 2016;175:188-94.e1.
Baskind MJ, Taveras EM, Gerber MW, Fiechtner L, Horan C, Sharifi M. Parent-perceived stress and its association with children’s weight and obesity-related behaviors. Prev Chronic Dis. 2019;16:E39.
Heerman WJ, Krishnaswami S, Barkin SL, McPheeters M. Adverse family experiences during childhood and adolescent obesity. Obesity (Silver Spring). 2016;24(3):696–702.
Costa S, Adams J, Gonzalez-Nahm S, Benjamin Neelon SE. Childcare in infancy and later obesity: a narrative review of longitudinal studies. Curr Pediatr Rep. 2017;5(3):118–31.
Peden ME, Jones R, Costa S, Ellis Y, Okely AD. Relationship between children’s physical activity, sedentary behavior, and childcare environments: a cross sectional study. Prev Med Rep. 2017;6:171–6.
Phillips D, Adams G. Child care and our youngest children. The Future of children / Center for the Future of Children, the David and Lucile Packard Foundation. 2001;11:34–51.
Capizzano J, Adams G, Sonenstein FL. Child care arrangements for children under five: variation across states. 2000.
Dowda M, Pate RR, Trost SG, Almeida MJ, Sirard JR. Influences of preschool policies and practices on children’s physical activity. J Community Health. 2004;29(3):183–96.
Padget A, Briley ME. Dietary intakes at child-care centers in central Texas fail to meet Food Guide Pyramid recommendations. J Am Diet Assoc. 2005;105(5):790–3.
Briley ME, Roberts-Gray C, Rowe S. What can children learn from the menu at the child care center? J Community Health. 1993;18(6):363–77.
Bollella MC, Spark A, Boccia LA, Nicklas TA, Pittman BP, Williams CL. Nutrient intake of Head Start children: home vs. school. J Am Coll Nutr. 1999;18(2):108–14.
James J, Thomas P, Cavan D, Kerr D. Preventing childhood obesity by reducing consumption of carbonated drinks: cluster randomised controlled trial. Bmj. 2004;328(7450):1237.
Kerr JA, Loughman A, Knox A, Koplin JJ, Allen KJ, Wake M. Nutrition-related interventions targeting childhood overweight and obesity: a narrative review. Obes Rev. 2019;20(Suppl 1):45–60.
Ottley PG, Dawkins-Lyn N, Harris C, Dooyema C, Jernigan J, Kauh T, et al. Childhood obesity declines project: an exploratory study of strategies identified in communities reporting declines. Child Obes. 2018;14(S1):S12–s21.
Ojeda-Rodríguez A, Zazpe I, Morell-Azanza L, Chueca MJ, Azcona-Sanjulian MC, Marti A. Improved diet quality and nutrient adequacy in children and adolescents with abdominal obesity after a lifestyle intervention. Nutrients. 2018;10(10).
Al-Khudairy L, Loveman E, Colquitt JL, Mead E, Johnson RE, Fraser H, et al. Diet, physical activity and behavioural interventions for the treatment of overweight or obese adolescents aged 12 to 17 years. Cochrane Database Syst Rev. 2017;6(6):Cd012691.
Cauchi D, Glonti K, Petticrew M, Knai C. Environmental components of childhood obesity prevention interventions: an overview of systematic reviews. Obes Rev. 2016;17(11):1116–30.
Colquitt JL, Loveman E, O'Malley C, Azevedo LB, Mead E, Al-Khudairy L, et al. Diet, physical activity, and behavioural interventions for the treatment of overweight or obesity in preschool children up to the age of 6 years. Cochrane Database Syst Rev. 2016;3(3):Cd012105.
Hollar D, Messiah SE, Lopez-Mitnik G, Hollar TL, Almon M, Agatston AS. Effect of a two-year obesity prevention intervention on percentile changes in body mass index and academic performance in low-income elementary school children. Am J Public Health. 2010;100(4):646–53.
Grummon AH, Taillie LS. Supplemental nutrition assistance program participation and racial/ethnic disparities in food and beverage purchases. Public Health Nutr. 2018;21(18):3377–85.
Ford MC, Gordon NP, Howell A, Green CE, Greenspan LC, Chandra M, et al. Obesity severity, dietary behaviors, and lifestyle risks vary by race/ethnicity and age in a Northern California cohort of children with obesity. J Obes. 2016;2016:4287976.
Guerrero AD, Chung PJ. Racial and ethnic disparities in dietary intake among California children. J Acad Nutr Diet. 2016;116(3):439–48.
Block JP, Scribner RA, DeSalvo KB. Fast food, race/ethnicity, and income: a geographic analysis. Am J Prev Med. 2004;27(3):211–7.
Fiechtner L, Sharifi M, Sequist T, Block J, Duncan DT, Melly SJ, et al. Food environments and childhood weight status: effects of neighborhood median income. Child Obes. 2015;11(3):260–8.
Johnson KA, Showell NN, Flessa S, Janssen M, Reid N, Cheskin LJ, et al. Do neighborhoods matter? A systematic review of modifiable risk factors for obesity among low socio-economic status black and hispanic children. Child Obes. 2019;15(2):71–86.
Lovasi GS, Hutson MA, Guerra M, Neckerman KM. Built environments and obesity in disadvantaged populations. Epidemiol Rev. 2009;31:7–20.
Hong I, Coker-Bolt P, Anderson KR, Lee D, Velozo CA. Relationship between physical activity and overweight and obesity in children: findings from the 2012 National Health and Nutrition Examination Survey National Youth Fitness Survey. Am J Occup Ther. 2016;70(5):7005180060p1-8.
Piercy KL, Troiano RP, Ballard RM, Carlson SA, Fulton JE, Galuska DA, et al. The physical activity guidelines for Americans. Jama. 2018;320(19):2020–8.
Minges KE, Chao A, Nam S, Grey M, Whittemore R. Weight status, gender, and race/ethnicity: are there differences in meeting recommended health behavior guidelines for adolescents? J Sch Nurs. 2015;31(2):135–45.
Mitchell JA, Dowda M, Pate RR, Kordas K, Froberg K, Sardinha LB, et al. Physical activity and pediatric obesity: a quantile regression analysis. Med Sci Sports Exerc. 2017;49(3):466–73.
Wang Y, Cai L, Wu Y, Wilson RF, Weston C, Fawole O, et al. What childhood obesity prevention programmes work? A systematic review and meta-analysis. Obes Rev. 2015;16(7):547–65.
Whooten RC, Perkins ME, Gerber MW, Taveras EM. Effects of before-school physical activity on obesity prevention and wellness. Am J Prev Med. 2018;54(4):510–8.
Hoffman J, Frerichs L, Story M, Jones J, Gaskin K, Apple A, et al. An integrated clinic-community partnership for child obesity treatment: a randomized pilot trial. Pediatrics. 2018;141(1).
Smith JD, Fu E, Kobayashi MA. Prevention and management of childhood obesity and its psychological and health comorbidities. Annu Rev Clin Psychol. 2020;16:351–78.
Bauer KW, Marcus MD, Larson N, Neumark-Sztainer D. Socioenvironmental, personal, and behavioral correlates of severe obesity among an ethnically/racially diverse sample of US adolescents. Child Obes. 2017;13(6):470–8.
Wang Y. Disparities in pediatric obesity in the United States. Adv Nutr. 2011;2(1):23–31.
Flores R. Dance for health: improving fitness in African American and Hispanic adolescents. Public Health Rep. 1995;110(2):189–93.
Barbeau P, Johnson MH, Howe CA, Allison J, Davis CL, Gutin B, et al. Ten months of exercise improves general and visceral adiposity, bone, and fitness in black girls. Obesity (Silver Spring). 2007;15(8):2077–85.
Fitzgibbon ML, Stolley MR, Schiffer L, Van Horn L, KauferChristoffel K, Dyer A. Hip-Hop to Health Jr. for Latino preschool children. Obesity (Silver Spring). 2006;14(9):1616–25.
Fitzgibbon ML, Stolley MR, Schiffer L, Van Horn L, KauferChristoffel K, Dyer A. Two-year follow-up results for Hip-Hop to Health Jr.: a randomized controlled trial for overweight prevention in preschool minority children. J Pediatr. 2005;146(5):618–25.
Fitzgibbon ML, Stolley MR, Schiffer L, Kong A, Braunschweig CL, Gomez-Perez SL, et al. Family-based hip-hop to health: outcome results. Obesity (Silver Spring). 2013;21(2):274–83.
Salam RA, Padhani ZA, Das JK, Shaikh AY, Hoodbhoy Z, Jeelani SM, et al. Effects of lifestyle modification interventions to prevent and manage child and adolescent obesity: a systematic review and meta-analysis. Nutrients. 2020;12(8).
Kelley CP, Sbrocco G, Sbrocco T. Behavioral modification for the management of obesity. Prim Care. 2016;43(1):159–75 x.
Wilfley DE, Kass AE, Kolko RP. Counseling and behavior change in pediatric obesity. Pediatr Clin N Am. 2011;58(6):1403–24 x.
Epstein LH, Valoski A, Wing RR, McCurley J. Ten-year follow-up of behavioral, family-based treatment for obese children. Jama. 1990;264(19):2519–23.
Kalarchian MA, Levine MD, Arslanian SA, Ewing LJ, Houck PR, Cheng Y, et al. Family-based treatment of severe pediatric obesity: randomized, controlled trial. Pediatrics. 2009;124(4):1060–8.
Young KM, Northern JJ, Lister KM, Drummond JA, O'Brien WH. A meta-analysis of family-behavioral weight-loss treatments for children. Clin Psychol Rev. 2007;27(2):240–9.
Perdew M, Liu S, Naylor PJ. Family-based nutrition interventions for obesity prevention among school-aged children: a systematic review. Transl Behav Med. 2021;11(3):709–23.
Chai LK, Collins C, May C, Brain K, Wong See D, Burrows T. Effectiveness of family-based weight management interventions for children with overweight and obesity: an umbrella review. JBI Database System Rev Implement Rep. 2019;17(7):1341–427.
Samuel-Hodge CD, Gizlice Z, Cai J, Brantley PJ, Ard JD, Svetkey LP. Family functioning and weight loss in a sample of african americans and whites. Ann Behav Med. 2010;40(3):294–301.
Ash T, Agaronov A, Young T, Aftosmes-Tobio A, Davison KK. Family-based childhood obesity prevention interventions: a systematic review and quantitative content analysis. Int J Behav Nutr Phys Act. 2017;14(1):113.
Parker MM, Moffet HH, Schillinger D, Adler N, Fernandez A, Ciechanowski P, et al. Ethnic differences in appointment-keeping and implications for the patient-centered medical home--findings from the Diabetes Study of Northern California (DISTANCE). Health Serv Res. 2012;47(2):572–93.
Srivastava G, Fox CK, Kelly AS, Jastreboff AM, Browne AF, Browne NT, et al. Clinical considerations regarding the use of obesity pharmacotherapy in adolescents with obesity. Obesity (Silver Spring). 2019;27(2):190–204.
Gomez G, Stanford FC. US health policy and prescription drug coverage of FDA-approved medications for the treatment of obesity. Int J Obes. 2018;42(3):495–500.
Pratt JSA, Browne A, Browne NT, Bruzoni M, Cohen M, Desai A, et al. ASMBS pediatric metabolic and bariatric surgery guidelines, 2018. Surg Obes Relat Dis. 2018;14(7):882–901.
Armstrong SC, Bolling CF, Michalsky MP, Reichard KW. Pediatric metabolic and bariatric surgery: evidence, barriers, and best practices. Pediatrics. 2019;144(6):e20193223.
Griggs CL, Perez NP Jr, Goldstone RN, Kelleher CM, Chang DC, Stanford FC, et al. National trends in the use of metabolic and bariatric surgery among pediatric patients with severe obesity. JAMA Pediatr. 2018;172(12):1191–2.
Nunez Lopez O, Jupiter DC, Bohanon FJ, Radhakrishnan RS, Bowen-Jallow KA. Health disparities in adolescent bariatric surgery: nationwide outcomes and utilization. J Adolesc Health. 2017;61(5):649–56.
Bolling CF, Armstrong SC, Reichard KW, Michalsky MP. Metabolic and bariatric surgery for pediatric patients with severe obesity. Pediatrics. 2019;144(6):e20193224.
Perez NP, Westfal ML, Stapleton SM, Stanford FC, Griggs CL, Pratt JS, et al. Beyond insurance: race-based disparities in the use of metabolic and bariatric surgery for the management of severe pediatric obesity. Surg Obes Relat Dis. 2020;16(3):414–9.
Acknowledgments
This work is supported by the following sources of funding: Massachusetts General Hospital Executive Committee on Research (ECOR)(FCS) and National Institutes of Health NIDDK P30 DK040561 (FCS) and L30 DK118710 (FCS).
Author information
Authors and Affiliations
Contributions
VRJ, NOA, ACD, AK, EHW, and FCS all contributed to drafting and revising manuscript. All authors approved final version of manuscript to be published.
Corresponding author
Ethics declarations
Conflict of Interest
Veronica R. Johnson, Nonyerem O. Acholonu, Ana C. Dolan, Ashwin Krishnan, Emily Hsu-Chi Wang, and Fatima Cody Stanford declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Psychological Issues
Rights and permissions
About this article
Cite this article
Johnson, V.R., Acholonu, N.O., Dolan, A.C. et al. Racial Disparities in Obesity Treatment Among Children and Adolescents. Curr Obes Rep 10, 342–350 (2021). https://doi.org/10.1007/s13679-021-00442-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13679-021-00442-0