Abstract
Background
Fatigue is a frequent symptom in breast cancer patients and breast cancer survivors. The aims of this study were to test a newly developed fatigue questionnaire (EORTC QLQ-FA13) and to detect prognostic factors of fatigue.
Methods
A total of 308 breast cancer patients (response rate: 91 %) were tested with the fatigue questionnaire EORTC QLQ-FA13 and the fatigue scale of the EORTC QLQ-C30. In addition, several other questionnaires were also adopted.
Results
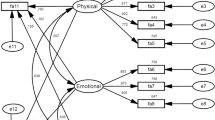
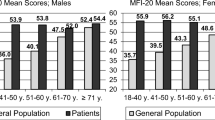
The youngest age group of breast cancer patients was most strongly affected by fatigue. The psychometric analysis of the EORTC QLQ-FA13 supported a three-dimensional structure (physical, emotional, and cognitive fatigue). The reliability (Cronbach’s alpha = 0.94) was very good. Clinical factors such as time since diagnosis and received therapy did not significantly predict the level of fatigue.
Conclusion
Fatigue remains a severe problem in breast cancer patients, especially in younger women. Researchers or clinicians who want to measure fatigue have the choice between the short 3-item fatigue scale of the EORTC QLQ-C30, stressing the physical aspect of fatigue, and the long 13-item EORTC QLQ-FA13 scale with slightly better psychometric properties, including cognitive and emotional aspects of fatigue.
Similar content being viewed by others
References
Ahlberg K, Ekman T, Gaston-Johansson F, Mock V. Assessment and management of cancer-related fatigue in adults. Lancet. 2003;362:640–50.
Bruera E, Yennurajalingam S. Challenge of managing cancer-related fatigue. J Clin Oncol. 2010;28:3671–2.
Weis J. Cancer-related fatigue: prevalence, assessment and treatment strategies. Expert Rev Pharmacoeconom Outcomes Res. 2011;11:441–6.
Allemani C, Minicozzi P, Berrino F, Bastiaannet E, Gavin A, Galceran J, et al. Predictions of survival up to 10 years after diagnosis for European women with breast cancer in 2000–2002. Int J Cancer. 2013;132:2404–12.
Allemani C, Sant M, Weir HK, Richardson LC, Baili P, Storm H, et al. Breast cancer survival in the US and Europe: a CONCORD high-resolution study. Int J Cancer. 2013;132:1170–81.
Sant M, Allemani C, Santaquilani M, Knijn A, Marchesi F, Capocaccia R. EUROCARE-4. Survival of cancer patients diagnosed in 1995–1999. Results and commentary. Eur J Cancer. 2009;45:931–91.
Arndt V, Merx H, Sturmer T, Stegmaier C, Ziegler H, Brenner H. Age-specific detriments to quality of life among breast cancer patients one year after diagnosis. Eur J Cancer. 2004;40:673–80.
Waldmann A, Pritzkuleit R, Raspe H, Katalinic A. The OVIS study: health related quality of life measured by the EORTC QLQ-C30 and-BR23 in German female patients with breast cancer from Schleswig-Holstein. Qual Life Res. 2007;16:767–76.
Kramer JA, Curran D, Piccart M, De Haes JCJM, Bruning P, Klijn J, et al. Identification and interpretation of clinical and quality of life prognostic factors for survival and response to treatment in first-line chemotherapy in advanced breast cancer. Eur J Cancer. 2000;36:1498–506.
Kuhnt S, Ernst J, Singer S, Rüffer JU, Kortmann R-D, Stolzenburg J-U, et al. Fatigue in cancer survivors—prevalence and predictors. Onkologie. 2009;32:312–7.
Schleife H, Sachtleben C, Finck BC, Singer S, Hinz A. Anxiety, depression, and quality of life in German ambulatory breast cancer patients. Breast Cancer. 2014;. doi:10.1007/s12282-012-0378-6.
Schou I, Ekeberg O, Sandvik L, Hjermstad MJ, Ruland CM. Multiple predictors of health-related quality of life in early stage breast cancer. Data from a year follow-up study compared with the general population. Qual Life Res. 2005;14:1813–23.
Vollrath M, Zenger M, Singer S, Einenkel J, Hinz A. The course of fatigue in patients with gynecologic and breast cancer. J Gynecol Oncol. 2013;24:280–6.
Singer S, Kuhnt S, Zwerenz R, Eckert K, Hofmeister D, Dietz A, et al. Age- and sex-standardised prevalence rates of fatigue in a large hospital-based sample of cancer patients. Br J Cancer. 2011;105:445–51.
Seyidova-Khoshknabi D, Davis MP, Walsh D. A systematic review of cancer-related fatigue measurement questionnaires. Am J Hospice Palliative Med. 2011;28:119–29.
Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ, et al. The European-Organization-For-Research-And-Treatment-Of-Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85:365–76.
Smets EMA, Garssen B, Bonke B, Dehaes JCJM. The Multidimensional Fatigue Inventory (MFI): psychometric qualities of an instrument to assess fatigue. J Psychosom Res. 1995;39:315–25.
Beutel ME, Hinz A, Albani C, Brahler E. Fatigue Assessment Questionnaire: standardization of a cancer-specific instrument based on the general population. Oncology. 2006;70:351–7.
Reeve BB, Stover AM, Alfano CM, Smith AW, Ballard-Barbash R, Bernstein L, et al. The Piper Fatigue Scale-12 (PFS-12): psychometric findings and item reduction in a cohort of breast cancer survivors. Breast Cancer Res Treat. 2012;136:9–20.
Weis J, Arraras JI, Conroy T, Efficace F, Fleissner C, Gorog A, et al. Development of an EORTC quality of life phase III module measuring cancer-related fatigue (EORTC QLQ-FA13). Psychooncology. 2013;22:1002–7.
Schwarz R, Hinz A. Reference data for the quality of life questionnaire EORTC QLQ-C30 in the general German population. Eur J Cancer. 2001;37:1345–51.
Lowe B, Wahl I, Rose M, Spitzer C, Glaesmer H, Wingenfeld K, et al. A 4-item measure of depression and anxiety: validation and standardization of the Patient Health Questionnaire-4 (PHQ-4) in the general population. J Affect Disord. 2010;122:86–95.
Henrich G, Herschbach P. Questions on Life Satisfaction (FLZ(M)): a short questionnaire for assessing subjective quality of life. Eur J Psychol Assess. 2000;16:150–9.
Brooks R. EuroQoL: the current state of play. Health Policy. 1996;37:53–72.
Scheier MF, Carver CS, Bridges MW. Distinguishing optimism from neuroticism (and trait anxiety, self-mastery, and self-esteem): a reevaluation of the Life Orientation Test. J Pers Soc Psychol. 1994;67:1063–78.
Hu LT, Bentler PM. Cutoff criteria for fit indexes in covariance structure analysis: conventional criteria versus new alternatives. Struct Equ Model. 1999;6:1–55.
Cohen J. Statistical power analysis for the behavioral sciences, 2nd ed. Erlbaum, Hillsdale, NJ; 1988.
Derogar M, van der Schaaf M, Lagergren P. Reference values for the EORTC QLQ-C30 quality of life questionnaire in a random sample of the Swedish population. Acta Oncol. 2012;51:10–6.
Michelson H, Bolund C, Nilsson B, Brandberg Y. Health-related quality of life measured by the EORTC QLQ-C30—reference values from a large sample of the Swedish population. Acta Oncol. 2000;39:477–84.
Romito F, Cormio C, Giotta F, Colucci G, Mattioli V. Quality of life, fatigue and depression in Italian long-term breast cancer survivors. Support Care Cancer. 2012;20:2941–8.
Prinsen H, Bleijenberg G, Heijmen L, Zwarts MJ, Leer JWH, Heerschap A, et al. The role of physical activity and physical fitness in postcancer fatigue: a randomized controlled trial. Support Care Cancer. 2013;21:2279–88.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Fuhrmann, K., Mehnert, A., Geue, K. et al. Fatigue in breast cancer patients: psychometric evaluation of the fatigue questionnaire EORTC QLQ-FA13. Breast Cancer 22, 608–614 (2015). https://doi.org/10.1007/s12282-014-0527-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12282-014-0527-1