Abstract
Background
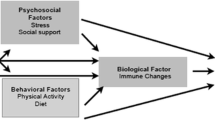
Cognitive changes following breast cancer treatment are likely multifactorial and have been linked to emotional factors, biophysiological factors, and fatigue, among others. Little is known about the contributions of modifiable factors such as stress, loneliness, and sleep quality. The purpose of this study was to explore the direct and indirect effects of perceived stress, loneliness, and sleep quality on perceived cognitive function (PCF) in breast cancer survivors (BCS) after chemotherapy completion.
Methods
In this observational study, BCS 6 months to 10 years post chemotherapy were recruited from the community. We measured perceived stress, loneliness, sleep quality, anxiety, depression, fatigue, and PCF. Data analyses included descriptive statistics, correlations, and mediation analyses utilizing ordinary least square regression.
Results
Ninety women who were on average 3 years post chemotherapy completion participated in the study. Moderate to largely negative correlations were found between PCF and the psychosocial and sleep variables (r values ranged from − 0.31 to − 0.70, p values < .0009). Mediation analyses revealed that stress and daytime sleepiness both directly and indirectly impact PCF and that loneliness and sleep quality only have indirect effects (through anxiety and fatigue).
Conclusion
Our findings suggest that perceived cognitive changes following breast cancer treatment are multifactorial and that higher stress levels, loneliness, daytime sleepiness, and poorer sleep quality are linked to worse perceived cognitive functioning. Also, stress, loneliness, and sleep quality may affect cognitive functioning through a shared psychobiological pathway.
Implications for cancer survivors
Interventions targeting stress, loneliness, and sleep quality may improve perceived cognitive functioning in breast cancer survivors.

Similar content being viewed by others
References
Ganz PA, Bower JE, Kwan L, et al. Does tumor necrosis factor-alpha (TNF-α) play a role in post-chemotherapy cerebral dysfunction? Brain Behav Immun. 2013;30(Suppl):S99–S108.
Janelsins MC, Kesler SR, Ahles TA, Morrow GR. Prevalence, mechanisms, and management of cancer-related cognitive impairment. Int Rev Psychiatry. 2014;26:102–13.
Koppelmans V, Breteler MMB, Boogerd W, Seynaeve C, Gundy C, Schagen SB. Neuropsychological performance in survivors of breast cancer more than 20 years after adjuvant chemotherapy. J Clin Oncol. 2012;30:1080–6.
Hutchinson AD, Hosking JR, Kichenadasse G, Mattiske JK, Wilson C. Objective and subjective cognitive impairment following chemotherapy for cancer: a systematic review. Cancer Treat Rev. 2012;38:926.
Janelsins MC, Heckler CE, Peppone LJ, et al. Cognitive complaints in survivors of breast cancer after chemotherapy compared with age-matched controls: an analysis from a nationwide, multicenter, prospective longitudinal study. J Clin Oncol. 2017;35:506–14.
Wefel JS, Kesler SR, Noll KR, Schagen SB. Clinical characteristics, pathophysiology, and management of noncentral nervous system cancer-related cognitive impairment in adults. CA Cancer J Clin. 2015;65:123–38.
Dietrich J, Prust M, Kaiser J. Chemotherapy, cognitive impairment and hippocampal toxicity. Neuroscience. 2015;309:224.
Ahles TA, Root JC, Ryan EL. Cancer- and cancer treatment-associated cognitive change: an update on the state of the science. J Clin Oncol. 2012;30:3675–86.
Jim HSL, Phillips KM, Chait S, et al. Meta-analysis of cognitive functioning in breast cancer survivors previously treated with standard-dose chemotherapy. J Clin Oncol. 2012;30:3578–87.
Pullens MJJ, de Vries J, Roukema JA. Subjective cognitive dysfunction in breast cancer patients: a systematic review. Psycho-Oncology. 2010;19:1127–38.
Bower JE, Lamkin DM. Inflammation and cancer-related fatigue: mechanisms, contributing factors, and treatment implications. Brain Behav Immun. 2013;30(Suppl):S48–57.
Cheung YT, Lim SR, Ho HK, Chan A. Cytokines as mediators of chemotherapy-associated cognitive changes: current evidence, limitations and directions for future research. PLoS One. 2013;8:e81234.
Hodgson KD, Hutchinson AD, Wilson CJ, Nettelbeck T. A meta-analysis of the effects of chemotherapy on cognition in patients with cancer. Cancer Treat Rev. 2013;39:297–304.
Reid-Arndt SA, Cox CR. Stress, coping and cognitive deficits in women after surgery for breast cancer. J Clin Psychol Med Settings. 2012;19:127–37.
Cheung YT, Shwe M, Tan YP, Fan G, Ng R, Chan A. Cognitive changes in multiethnic Asian breast cancer patients: a focus group study. Ann Oncol. 2012;23:2547–52.
Jaremka LM, Peng J, Bornstein R, et al. Cognitive problems among breast cancer survivors: loneliness enhances risk: loneliness and cognitive function. Psycho-Oncology. 2014;23:1356–64.
Sanford SD, Beaumont JL, Butt Z, Sweet JJ, Cella D, Wagner LI. Prospective longitudinal evaluation of a symptom cluster in breast cancer. J Pain Symptom Manag. 2014;47:721–30.
Von Ah D, Tallman EF. Perceived cognitive function in breast cancer survivors: evaluating relationships with objective cognitive performance and other symptoms using the functional assessment of cancer therapy-cognitive function instrument. J Pain Symptom Manage. 2015;49:697.
Xiao C, Miller AH, Felger J, Mister D, Liu T, Torres MA. Depressive symptoms and inflammation are independent risk factors of fatigue in breast cancer survivors. Psychol Med 2017:1–11.
Patient-Reported Outcomes Measurement Information System 2017; http://www.healthmeasures.net/explore-measurement-systems/promis/obtain-administer-measures. Accessed July 17, 2017.
Cohen S, Kamarck T, Mermelstein R. A global measure of perceived stress. J Health Soc Behav. 1983;24:385–96.
Russell DWUCLA. Loneliness scale (version 3): reliability, validity, and factor structure. J Pers Assess. 1996;66:20–40.
Buysse DJ, Reynolds CF, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh sleep quality index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28:193–213.
Wagner LI, Sweet JJ, Butt Z, Lai JS, Cella D. Measuring patient self-reported cognitive function: development of the functional assessment of cancer therapy–cognitive function instrument. J Support Oncol. 2009;7:7.
Hayes AF. Introduction to mediation, moderation, and conditional process analysis: a regression-based approach. New York: The Guilford Press; 2013.
Cessna JM, Jim HSL, Sutton SK, et al. Evaluation of the psychometric properties of the PROMIS cancer fatigue short form with cancer patients. J Psychosom Res. 2016;81:9–13.
Bray VJ, Dhillon HM, Bell ML, et al. Evaluation of a web-based cognitive rehabilitation program in cancer survivors reporting cognitive symptoms after chemotherapy. J Clin Oncol. 2017;35:217–25.
Cheung YT, Foo YL, Shwe M, et al. Minimal clinically important difference (MCID) for the functional assessment of cancer therapy: cognitive function (FACT-Cog) in breast cancer patients. J Clin Epidemiol. 2014;67:811–20.
Li J, Yu L, Long Z, Li Y, Cao F. Perceived cognitive impairment in Chinese patients with breast cancer and its relationship with post-traumatic stress disorder symptoms and fatigue: perceived cognitive impairment in Chinese patients with breast cancer. Psycho-Oncology. 2015;24:676–82.
Myers JS, Wick JA, Klemp J. Potential factors associated with perceived cognitive impairment in breast cancer survivors. Support Care Cancer. 2015;23:3219–28.
Parelkar P, Thompson NJ, Kaw CK, Miner KR, Stein KD. Stress coping and changes in health behavior among cancer survivors: a report from the American Cancer Society’s Study of Cancer Survivors-II (SCS-II). J Psychosoc Oncol. 2013;31:136.
Rosedale M. Survivor loneliness of women following breast cancer. Oncol Nurs Forum. 2009;36:175–83.
Palesh O, Peppone L, Innominato PF, Janelsins M, Jeong M, Sprod L, et al. Prevalence, putative mechanisms, and current management of sleep problems during chemotherapy for cancer. Nat Sci Sleep. 2012;4:151–62. https://doi.org/10.2147/NSS.S18895.
Andreotti C, Root JC, Ahles TA, McEwen BS, Compas BE. Cancer, coping, and cognition: a model for the role of stress reactivity in cancer-related cognitive decline: cancer, coping, and cognition. Psycho-Oncology. 2015;24:617–23.
Arndt J, Das HHJ, Schagen S, Reid-Arndt S, Cameron L, Ahles T. Broadening the cancer and cognition landscape: the role of self-regulatory challenges. Psycho-Oncology. 2014;23:1–8.
Bower JE, Ganz PA, Irwin MR, Kwan L, Breen EC, Cole SW. Inflammation and behavioral symptoms after breast cancer treatment: do fatigue, depression, and sleep disturbance share a common underlying mechanism? J Clin Oncol. 2011;29:3517–22. https://doi.org/10.1200/JCO.2011.36.1154.
Cacioppo JT, Hawkley LC. Perceived social isolation and cognition. Trends Cogn Sci. 2009;13:447–54.
Yaffe K, Falvey C, Hoang T. Connections between sleep and cognition in older adults. Lancet Neurol. 2014;13:1017–28.
Killgore WDS. Effects of sleep deprivation on cognition. In: Vol 185. AMSTERDAM: Elsevier Science & Technology; 2010:105–129.
Oh P. Predictors of cognitive decline in people with cancer undergoing chemotherapy. Eur J Oncol Nurs 2016.
Funding
Research reported in this publication was supported by the National Institute of Nursing Research of the National Institutes of Health under award number F31NR015707. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Ashley M. Henneghan was supported by the Doctoral Degree Scholarship in Cancer Nursing, DSCN-15-072-01 from the American Cancer Society.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Henneghan, A., Stuifbergen, A., Becker, H. et al. Modifiable correlates of perceived cognitive function in breast cancer survivors up to 10 years after chemotherapy completion. J Cancer Surviv 12, 224–233 (2018). https://doi.org/10.1007/s11764-017-0661-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11764-017-0661-9