Summary
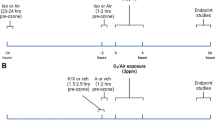
Female infertility after occupational exposure to inhaled anesthetic agents has attracted critical attention, but systematic studies focusing on the impact of inhaled anesthetics on the female reproductive system have not been well-established. We used a murine model to study the effect of isoflurane exposure on infertility in female adult mice and investigated the potential underlying mechanism. One hundred adult female C57 mice were randomly allocated into 5 groups exposed in air containing 0, 2500, 5000, 10 000 or 20 000 ppm isoflurane for 15 consecutive days. Estrous cycle length was measured based on vaginal smear examination, ovarian histopathologic enumeration of follicles, and serum estradiol (E2), anti-Mullerian hormone (AMH), follicle-stimulating hormone (FSH), and luteinizing hormone (LH) levels to assess the effect of isoflurane on ovarian reserve. Compared to the control group, significant prolongation of the estrous cycle of the adult female mice was observed in the 20 000 ppm isoflurane exposure group. Serum AMH was significantly decreased, and FSH and LH levels profoundly increased in the 5000, 10 000, and 20 000 ppm isoflurane exposure groups compared to the control group. The histopathologic examination revealed a reduced number of developing follicles and an increased number of atretic follicles after isoflurane exposure, but the difference was not statistically significant. Thus, exposure to a higher concentration of isoflurane might have an adverse effect on ovarian reserve in sexually-mature female mice.
Similar content being viewed by others
References
Byhahn C, Wilke HJ, Westphal K, et al. Occupational exposure to volatile anaesthetics: Epidemiology and approaches to reducing the problem. CNS Drugs, 2001,15(3): 197–215
Smith FD. Management of Exposure to Waste Anesthetic Gases. AORN, 2010, 94(4):482–494
Nagella AB, Ravishankar M, Hemanth Kumar VR. Anaesthesia practice and reproductive outcomes: Facts unveiled. Indian J Anaesth, 2015,59(11):706–714
Conlee KM, Stephens ML, Rowan AN, et al. Carbon dioxide for euthanasia: Concerns regarding pain and distress, with special reference to mice and rats. Lab Anim, 2005,39(2): 137–161
Johnstone KR, Lau C, Whitelaw JL. Evaluation of waste isoflurane gas exposure during rodent surgery in an Australian university. J Occup Environ Hyg, 2017,14(12):955–964
Topf N, Jenkins A, Baron N, et al. Effects of Isoflurane on y-Aminobutyric Acid Type A Receptors Activated by Full and Partial Agonists. Anesthesiology, 2003,98(2): 306–311
Neumahr S, Hapfelmeier G, Scheller M, et al. Dual action of isoflurane on the gamma-aminobutyric acid (GABA)-mediated currents through recombinant alpha(1)beta(2)gamma(2L)-GABA(A)-receptor channels. Anesth Analg, 2000,90(5): 1184–1190
Woll KA, Zhou X, Bhanu N, et al. Identification of binding sites contributing to volatile anesthetic effects on GABA type A receptors. FASEB J, 2018,32(8): 4172–4189
Li S, Zhang Y, Liu H, et al. Identification and expression of GABAC receptor in rat testis and spermatozoa. Acta Biochimica et Biophysica Sinica, 2008,40(8): 761–767
Christian CA, Moenter SM. The neurobiology of preovulatory and estradiol-induced gonadotropin-releasing hormone surges. Endocr Rev, 2010,31(4):544–577
Ni C, Li C, Dong Y, et al. Anesthetic Isoflurane Induces DNA Damage through Oxidative Stress and p53 Pathway. Mol. Neurobiol, 2017,54(4):3591–3605
Venkatesan T, Amar N. Genotoxic effects of anaesthetic agents. Acta Anaesthesiol Scand, 2006,50(5):636
Xu XL, Pan C, Hu JX, et al. Effects of isoflurane inhalation on the male reproductive system in rats. Environ Toxicol Pharmacol, 2012,34(3): 688–693
Cora MC, Kooistra L, Travlos G. Vaginal Cytology of the Laboratory Rat and Mouse:Review and Criteria for the Staging of the Estrous Cycle Using Stained Vaginal Smears. Toxicol Pathol, 2015,43(6):776–793
Westwood FR. The Female Rat Reproductive Cycle: A Practical Histological Guide to Staging. Toxicol Pathol, 2008,36(3):375–384
Ding Y, Yu J, Qu P, et al. The negative effects of chronic exposure to isoflurane on spermatogenesis via breaking the hypothalamus-pituitary-gonadal equilibrium. Inhal Toxicol, 2015,27(12): 621–628
Campion SN, Cappon GD, Chapin R, et al. Isoflurane reduces motile sperm counts in the Sprague-Dawley rat. Drug Chem Toxicol, 2012,35(1): 20–24
O’Connor JC, Frame SR, Ladics GS. Evaluation of a 15-day screening assay using intact male rats for identifying antiandrogens. Toxicol Sci, 2002,69(1):92–108
Steiner AZ, Pritchard D, Stanczyk FZ, et al. Association between biomarkers of ovarian reserve and infertility among older women of reproductive age. JAMA, 2017, 318(14):1367–1376
Victoria M, Labrosse J, Krief F, et al. Anti Müllerian Hormone: More than a biomarker of female reproductive function. J Gynecol Obstet Hum Reprod, 2018,48(1): 19–24
Kevenaar ME, Meerasahib MF, Kramer P, et al. Serum Anti-Müllerian Hormone Levels Reflect the Size of the Primordial Follicle Pool in Mice. Endocrinology, 2006,147(4): 3228–3234
Guo JQ, Gao X, Lin ZJ, et al. BMSCs reduce rat granulosa cell apoptosis induced by cisplatin and perimenopause. BMC Cell Biology, 2013,14:18
Hussein MR. Apoptosis in the ovary: Molecular mechanisms. Hum Reprod Update, 2005,11(2):161–177
Wang HW, Zhao WP, Liu J, et al. Fluoride-induced oxidative stress and apoptosis are involved in the reducing of oocytes development potential in mice. Chemosphere, 2017,186:911–918
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by the National Key Research and Development Program (No. 2018YFC1002103).
Conflict of Interest Statement
The authors have no conflicts of interest relevant to this article to disclose.
Rights and permissions
About this article
Cite this article
Tang, Xn., Yao, W., Yao, Hx. et al. Influence of Isoflurane Exposure for 15 Consecutive Days on Ovarian Function in Adult Female Mice. CURR MED SCI 40, 1177–1181 (2020). https://doi.org/10.1007/s11596-020-2300-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-020-2300-3