Abstract
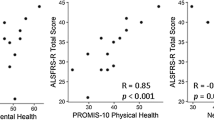
The patient reported outcomes measurement information system (PROMIS) provides clinicians and researchers access to reliable, validated measures of physical, mental, and social well-being. The use of PROMIS can facilitate comparisons among clinical subpopulations and with the U.S. general population. We report on the first study using PROMIS measures in patients with neurofibromatosis (NF). Eighty-six adult patients (mean age = 44; 55% female; 87% white; 50% NF1, 41% NF2 and 9% schwannomatosis) completed a battery of PROMIS computerized adaptive tests (CATs). Across all PROMIS instruments, mean scores for each CAT were between 48.97 and 52.60, which is within ±0.5 SD of the U.S. general population norms. However, scores were distributed across a broad range for each PROMIS measure (±3 SDs). Clinically meaningful scores (defined >1 SD impairment) were observed in 20% (pain interference), 17% (pain behavior), 16% (physical function), 16% (anxiety), 16% (depression), 15% (satisfaction with social roles), 13% (fatigue), 6% (anger), and 5% (satisfaction with discretionary social activities) of the sample. All PROMIS measures were highly interrelated in bivariate analysis (P ≤ .001). There were no differences in PROMIS scores by disease type (NF1, NF2 and schwannomatosis), or self reported learning disabilities, or compared with the US population. Scores suggest a broad continuum of symptoms and functioning in patients with NF that is not affected by NF type, as well as interrelation among the physical and psychosocial domains as measured by PROMIS. PROMIS measures may be useful in clinical practice to monitor changes in symptoms and functioning over time, as well as in clinical trials to determine patient reported changes during drug and psychosocial clinical trials.

Similar content being viewed by others
References
Wolters PL, Martin S, Merker VL et al (2016) Patient-reported outcomes of pain and physical functioning in neurofibromatosis clinical trials. Neurology 87(7 Suppl 1):S4–S12
Ferner RE, Shaw A, Evans DG et al (2014) Longitudinal evaluation of quality of life in 288 patients with neurofibromatosis 2. J Neurol 261(5):963–969
Cosetti MK, Golfinos JG, Roland JT Jr. (2015) Quality of life (QoL) assessment in patients with neurofibromatosis type 2 (NF2). Otolaryngol Head Neck Surg 153(4):599–605
Neary WJ, Hillier VF, Flute T, Stephens D, Ramsden RT, Evans DG (2010) Use of a closed set questionnaire to measure primary and secondary effects of neurofibromatosis type 2. J Laryngol Otol 124(7):720–728
Wolkenstein P, Zeller J, Revuz J, Ecosse E, Leplege A (2001) Quality-of-life impairment in neurofibromatosis type 1: a cross-sectional study of 128 cases. Arch Dermatol 137(11):1421–1425
Page PZ, Page GP, Ecosse E, Korf BR, Leplege A, Wolkenstein P (2006) Impact of neurofibromatosis 1 on quality of life: a cross-sectional study of 176 American cases. Am J Med Genet A 140A:1893–1898
Merker VL, Bredella MA, Cai W et al (2014) Relationship between whole-body tumor burden, clinical phenotype, and quality of life in patients with neurofibromatosis. Am J Med Genet A 164(6):1431–1437
Vranceanu AM, Merker VL, Park E, Plotkin SR (2013) Quality of life among adult patients with neurofibromatosis 1, neurofibromatosis 2 and schwannomatosis: a systematic review of the literature. J Neurooncol 114(3):257–262
Wang DL, Smith KB, Esparza S et al (2012) Emotional functioning of patients with neurofibromatosis tumor suppressor syndrome. Genet Med 14(12):977–982
Vranceanu AM, Merker VL, Plotkin SR, Park ER (2014) The relaxation response resiliency program (3RP) in patients with neurofibromatosis 1, neurofibromatosis 2, and schwannomatosis: results from a pilot study. J Neurooncol 120(1):103–109
Cella D, Chang CH (2000) A discussion of item response theory and its applications in health status assessment. Med Care 38(9 Suppl):II66–II72
Cella D, Yount S, Rothrock N et al (2007) The patient-reported outcomes measurement information system (PROMIS): progress of an NIH roadmap cooperative group during its first 2 years. Med Care 45(5 Suppl 1):S3–S11
Liu H, Cella D, Gershon R et al (2010) Representativeness of the patient-reported outcomes measurement information system internet panel. J Clin Epidemiol 63(11):1169–1178
Fries JF, Bruce B, Cella D (2005) The promise of PROMIS: using item response theory to improve assessment of patient-reported outcomes. Clin Exp Rheumatol 23(5 Suppl 39):S53–S57
Fries JF, Cella D, Rose M, Krishnan E, Bruce B (2009) Progress in assessing physical function in arthritis: PROMIS short forms and computerized adaptive testing. J Rheumatol 36(9):2061–2066
Schwartz CE, Bode R, Repucci N, Becker J, Sprangers MA, Fayers PM (2006) The clinical significance of adaptation to changing health: a meta-analysis of response shift. Qual Life Res 15(9):1533–1550
Mautner VF, Granstrom S, Leark RA (2012) Impact of ADHD in adults with neurofibromatosis type 1: associated psychological and social problems. J Atten Disord 19(1):35–43
Pride NA, Crawford H, Payne JM, North KN (2013) Social functioning in adults with neurofibromatosis type 1. Res Dev Disabil 34(10):3393–3399
Acknowledgements
The funding was provided by National Institutes of Health, U.S. Department of Defense and Children’s Tumor Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Mojtaba Talaei-Khoei, Eric Riklin, Monica R. Sheridan and Justin T. Jordan report no disclosures. Vanessa L. Merker receives research support from the Program in Cancer Outcomes Research Training (NCIR25CA92203) and a Young Investigator Award from the Children’s Tumor Foundation. Scott R. Plotkin receives research support from the Children’s Tumor Foundation, NIH, and Department of Defense Neurofibromatosis Clinical Trials Consortium. Ana-Maria Vranceanu receives research support from the Children’s Tumor Foundation.
Additional information
Scott R. Plotkin and Ana-Maria Vranceanu have contributed equally.
Rights and permissions
About this article
Cite this article
Talaei-Khoei, M., Riklin, E., Merker, V.L. et al. First use of patient reported outcomes measurement information system (PROMIS) measures in adults with neurofibromatosis. J Neurooncol 131, 413–419 (2017). https://doi.org/10.1007/s11060-016-2314-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-016-2314-7