Abstract
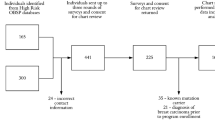
While participation in cancer screening can facilitate early detection and improved prognosis, all screening tests yield some proportion of abnormal test results which are later determined benign. These false positive (FP) results can negatively impact affective, cognitive, and behavioral outcomes. Women participating in an ovarian cancer (OC) screening program receiving an abnormal screening test result (n = 375) were matched with women receiving normal results (n = 375). Both groups completed a baseline and 1- and 4-month follow-up assessments. FP test results were clearly associated with increased cancer-specific distress and increased perceptions of OC risk with more limited evidence for increased perceived positive consequences of screening and increased intentions to participate in future OC screening. FP OC screening test results negatively impact both affective and cognitive outcomes which may serve to reduce motivation to participate in future routine screening. The development and testing of brief, timely interventions to minimize this negative impact is warranted.


Similar content being viewed by others
References
Ajzen, I. (1991). The theory of planned behavior. Organizational Behavior and Human Decision Processes, 50, 179–211.
American Cancer Society. (2015). Cancer facts and figures. Atlanta: American Cancer Society.
American Cancer Society. (2016). Cancer facts and figures. Atlanta: American Cancer Society.
Andersen, M. R., Drescher, C. W., Zheng, Y., Bowen, D. J., Wilson, S., Young, A., et al. (2007). Changes in cancer worry associated with participation in ovarian cancer screening. Psycho-Oncology, 16, 814–820.
Andrykowski, M. A., Boerner, L. M., Salsman, J. M., & Pavlik, E. (2004). Psychological response to test results in an ovarian cancer screening program: A prospective, longitudinal study. Health Psychology, 23, 622–630.
Andrykowski, M. A., Carpenter, J. S., Studts, J. L., Cordova, M. J., Cunningham, L. L., Beacham, A., et al. (2002). Psychological impact of benign breast biopsy: A longitudinal, comparative study. Health Psychology, 21, 485–494.
Andrykowski, M. A., Zhang, M., Pavlik, E. J., & Kryscio, R. J. (2007). Factors associated with return for routine annual screening in an ovarian cancer screening program. Gynecologic Oncology, 104, 695–701.
Antoni, M. H., Lehman, J. M., Kilbourn, K. M., Boyers, A. E., Culver, J. L., Alferi, S. M., et al. (2001). Cognitive-behavioral stress management intervention decreases the prevalence of depression and enhances benefit finding among women under treatment for early-stage breast cancer. Health Psychology, 20, 20–32.
Barrett, J., Jenkins, V., Farewell, V., Menon, U., Jacobs, I., Kilkerr, J., et al. (2014). Psychological morbidity associated with ovarian cancer screening: Results from more than 23,000 women in the randomised trial of ovarian cancer screening (UKCTOCS). British Journal of Obstetrics and Gynecology, 121, 1071–1079.
Brain, K. E., Lifford, K. J., Fraser, L., Rosenthal, A. N., Rogers, M. T., Lancastle, D., et al. (2012). Psychological outcomes of familial ovarian cancer screening: No evidence of long-term harm. Gynecologic Oncology, 127, 556–563.
Brewer, N. T., Salz, T., & Lillie, S. E. (2007). Systematic review: The long-term effects of false-positive mammograms. Annals of Internal Medicine, 146, 502–510.
Cockburn, J., De Luise, T., Hurley, S., & Clover, K. (1992). Development and validation of the PCQ: A questionnaire to measure the psychological consequences of screening mammography. Social Science and Medicine, 34, 1129–1134.
DePriest, P. D., & DeSimone, C. P. (2003). Ultrasound screening for early detection of ovarian cancer. Journal of Clinical Oncology, 21, 194s–199s.
Floyd, A., Steffens, R. F., Pavlik, E., & Andrykowski, M. A. (2011). Receipt of a false positive test result during routine screening for ovarian cancer: A teachable moment?. Journal of Clinical Psychology in Medical Settings, 18, 70–77.
Hays, R. D., & Wooley, J. M. (2000). The concept of clinically meaningful differences in health-related quality of life research: How meaningful is it? Pharmacoeconomics, 18, 419–423.
Helgeson, V. S., Reynolds, K. A., & Tomich, P. L. (2006). A meta-analytic review of benefit finding and growth. Journal of Consulting and Clinical Psychology, 74, 797–816.
Horowitz, M. (1982). Stress response symptoms and their treatment. In L. Goldberger & S. Breznitz (Eds.), Handbook of stress: Theoretical and clinical aspects (pp. 711–732). New York: Free Press.
Horowitz, M., Wilner, N., & Alvarez, W. (1979). Impact of event scale: A measure of subjective stress. Psychosomatic Medicine, 41, 209–218.
Jenkins, V., for and on behalf of UKCTOS Trialists, Fallowfield, L., Langridge, C., Barrett, J., Ryan, A., Jacobs, I., Kilkerr, J., Menon, U., & Farewell, V. (2015). Psychological factors associated with withdrawal from the United Kingdom Collaborative Trial of Ovarian Cancer Screening after 1 episode of repeat screening. International Journal of Gyncegological Cancer, 25, 1519–1525.
Kauff, N. D., Hurley, K. E., Hensley, M. L., Robson, M. E., Lev, G., Goldfrank, D., et al. (2005). Ovarian carcinoma screening in women at intermediate risk: Impact on quality of life and need for invasive follow-up. Cancer, 104, 314–320.
Lifford, K. J., Fraser, L., Rosenthal, A. N., Rogers, M. T., Lancastle, D., Phelps, C., et al. (2012). Withdrawal from familial ovarian screening for surgery: Findings from a psychological evaluation study (Psy FOCS). Gynecologic Oncology, 124, 158–163.
Lin, K., Lipsitz, R., Miller, T., & Janakiraman, S. (2008). Benefits and harms of prostate-specific antigen screening for prostate cancer: An evidence update for the U.S. Preventive services task force. Annals of Internal Medicine, 149, 192–199.
McGovern, P. M., Gross, C. R., Krueger, R. A., Engelhard, D. A., Cordes, J. E., & Church, T. R. (2004). False-positive cancer screens and health-related quality of life. Cancer Nursing, 27, 347–352.
Menon, U., Griffin, M., & Gentry-Maharaj, A. (2014). Ovarian cancer screening—current status, future directions. Gynecologic Oncology, 132, 490–495.
Moyer, V.A., & U.S. Preventive Services Task Force. (2012). Screening for ovarian cancer: U.S. preventive services task force reaffirmation recommendation statement. Annals of Internal Medicine, 157, 900–904.
Norman, G. R., Sloan, J. A., & Wyrwich, K. W. (2003). Interpretation of changes in health-related quality of life: The remarkable universality of half a standard deviation. Medical Care, 41, 582–592.
Pavlik, E. J., Ueland, F. R., Miller, R. V. V., Ubellacker, J. M., DeSomone, C. P., Hoff, J., et al. (2013). Frequency and disposition of ovarian abnormalities followed with serial transvaginal sonography. Obstetrics and Gynecology, 122, 210–217.
Radloff, L. S. (1977). The CES-D scale: A self-report depression scale for research in the general population. Applied Psychological Measurement, 1, 385–401.
Reade, C. J., Riva, J. J., Busse, J. W., Goldsmith, C. H., & Elit, L. (2013). Risks and benefits of screening asymptomatic women for ovarian cancer: A systematic review and meta-analysis. Gynecologic Oncology, 130, 674–681.
Scheier, M. F., Carver, C. S., & Bridges, M. W. (1994). Distinguishing optimism from neuroticism (and trait anxiety, self-mastery, and self-esteem): A reevaluation of the Life Orientation Test. Journal of Personality and Social Psychology, 67, 1063–1078.
Schorge, J. O., Modesitt, S. C., Coleman, R. L., Cohn, D. E., Kauff, N. D., Duska, L. R., et al. (2010). SGO white paper on ovarian cancer: Etiology, screening and surveillance. Gynecologic Oncology, 119, 7–17.
Schwartz, L. M., Woloshin, S., Fowler, F. J., Jr., & Welch, H. G. (2004). Enthusiasm for cancer screening in the United States. Journal of the American Medical Association, 291, 71–78.
Slatore, C. G., Sullivan, D. R., Pappas, M., & Humphrey, L. L. (2014). Patient-centered outcomes among lung cancer screening recipients with computed tomography: A systematic review. Journal of Thoracic Oncology, 9, 927–934.
Stanton, A. L. (2010). Positive consequences of the experience of cancer: Perception of growth and meaning. In J. C. Holland, W. S. Breitbart, P. B. Jacobsen, et al. (Eds.), Psycho-oncology (2nd ed., pp. 547–550). New York: Oxford University Press.
Stanton, A. L., Bower, J. E., & Low, C. A. (2006). Posttraumatic growth after cancer. In L. G. Calhoun & R. G. Tedeschi (Eds.), Handbook of posttraumatic growth: Research and practice (pp. 138–175). Mahwah, NJ: Erlbaum.
Steptoe, A. (1989). An abbreviated version of the Miller Behavioral Style Scale. British Journal of Clinical Psychology, 28(Pt 2), 183–184.
Taylor, K. L., Shleby, R., Gelmann, E., & McGuire, C. (2004). Quality of life and trial adherence among participants in the prostate, lung, colorectal, and ovarian cancer screening trial. Journal of the National Cancer Institute, 96, 1083–1094.
Tedeschi, R. G., & Calhoun, L. G. (1995). Trauma and transformation: Growing in the aftermath of significant suffering. Newbury Park, CA: Sage Publications.
Tedeschi, R. G., & Calhoun, L. G. (2004). Posttraumatic growth: Conceptual foundations and empirical evidence. Psychological Inquiry, 15, 1–18.
van Nagell, J. R., & Pavlik, E. J. (2012). Ovarian cancer screening. Clinics in Obstetrics and Gynecology, 55, 43–51.
Wardle, J., Collins, W., Pernet, A. L., Whitehead, M. I., Bourne, T. H., & Campbell, S. (1993). Psychological impact of screening for familial ovarian cancer. Journal of the National Cancer Institute, 85, 653–657.
Wardle, J., Pernet, A., Collins, W., & Bourne, T. (1994). False positive results in ovarian cancer screening: One year follow-up of psychological status. Psychology and Health, 10, 33–40.
Wardle, J., & Pope, R. (1992). The psychological costs of screening for cancer. Journal of Psychosomatic Research, 36, 609–624.
Wardle, J., Robb, K., Verson, S., & Waller, J. (2015). Screening for prevention and early diagnosis of cancer. American Psychologist, 70, 119–133.
Ware, J., Jr., Kosinski, M., & Keller, S. D. (1996). A 12-item short-form health survey: Construction of scales and preliminary tests of reliability and validity. Medical Care, 34, 220–233.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Amanda T. Wiggins, Edward J. Pavlik, and Michael A. Andrykowski declares that they do not have any conflict of interest.
Human and animal rights and informed consent
All procedures followed were in accordance with ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000. Informed consent was obtained from all patients for being included in the study.
Rights and permissions
About this article
Cite this article
Wiggins, A.T., Pavlik, E.J. & Andrykowski, M.A. Affective, cognitive and behavioral outcomes associated with a false positive ovarian cancer screening test result. J Behav Med 40, 803–813 (2017). https://doi.org/10.1007/s10865-017-9851-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10865-017-9851-1