Abstract
Background
Current instruments used to measure disease activity and health-related quality of life in patients with Crohn’s disease (CD) and ulcerative colitis (UC) are often cumbersome, time-consuming, and expensive; although used in clinical trials, they are not convenient for clinical practice. A numeric rating scale (NRS) is a quick, inexpensive, and convenient patient-reported outcome that can capture the patient’s overall perception of health.
Aims
The aim of this study was to assess the validity, reliability, and responsiveness of an NRS and evaluate its use in clinical practice in patients with CD and UC.
Methods
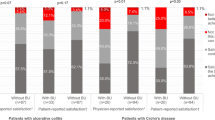
We prospectively evaluated patient-reported NRS scores and measured correlations between NRS and a range of severity measures, including physician-reported NRS, Crohn’s disease activity index (CDAI), Harvey-Bradshaw index (HBI), inflammatory bowel disease questionnaire (IBDQ), and C-reactive protein (CRP) in patients with CD. Subsequently, we evaluated the correlation between the NRS and standard measures of health status (HBI or simple colitis clinical activity index [SCCAI]) and laboratory tests (sedimentation rate [ESR], CRP, and fecal calprotectin) in patients with CD and UC.
Results
The patient-reported NRS showed excellent correlation with CDAI (R 2 = 0.59, p < 0.0001), IBDQ (R 2 = 0.66, p < 0.0001), and HBI (R 2 = 0.32, p < 0.0001) in patients with CD. The NRS showed poor, but statistically significant correlation with SCCAI (R 2 = 0.25, p < 0.0001) in patients with UC. The NRS did not correlate with CRP, ESR, or calprotectin. The NRS was reliable and responsive to change.
Conclusions
The NRS is a valid, reliable, and responsive measure that may be useful to evaluate patients with CD and possibly UC.




Similar content being viewed by others
References
Magasi S, Ryan G, Revicki D, et al. Content validity of patient-reported outcome measures: perspectives from a PROMIS meeting. Qual Life Res. 2011;21:739–746.
Fiscella K, Ransom S, Jean-Pierre P, et al. Patient-reported outcome measures suitable to assessment of patient navigation. Cancer. 2011;117:3601–3615.
Marshall S, Haywood K, Fitzpatrick R. Impact of patient-reported outcome measures on routine practice: a structured review. J Eval Clin Pract. 2006;12:559–568.
Velikova G, Booth L, Smith AB, et al. Measuring quality of life in routine oncology practice improves communication and patient well-being: a randomized controlled trial. J Clin Oncol. 2004;22:714–724.
Detmar SB, Muller MJ, Schornagel JH, et al. Health-related quality-of-life assessments and patient-physician communication: a randomized controlled trial. JAMA. 2002;288:3027–3034.
Sandborn WJ, Feagan BG, Hanauer SB, et al. A review of activity indices and efficacy endpoints for clinical trials of medical therapy in adults with Crohn’s disease. Gastroenterology. 2002;122:512–530.
Salaffi F, Stancati A, Silvestri CA, et al. Minimally clinically important changes in chronic musculoskeletal pain intensity measured on a numeric rating scale. Eur J Pain. 2004;8:283–291.
Samulak D, Michalska M, Gaca M, et al. Efficiency of postoperative pain management after gynecologic oncological surgeries with the use of morphine + acetaminophen + ketoprofen versus morphine + metamizol + ketoprofen. Eur J Gynaecol Oncol. 2001;32:168–170.
Van Seventer R, Serpell M, Bach FW, et al. Relationships between changes in pain severity and other patient-reported outcomes: an analysis in patients with posttraumatic peripheral neuropathic pain. Health Qual Life Outcomes. 2011;9:17.
Eckmann MS, Ramamurthy S, Griffin JG. Intravenous regional ketorolac and lidocaine in the treatment of complex regional pain syndrome of the lower extremity: a randomized, double-blinded, crossover study. Clin J Pain. 2011;27:203–206.
Simpson DM, Schifitto G, 1066 HIV Neuropathy Study Group, et al. Pregabalin for painful HIV neuropathy: a randomized, double-blind, placebo-controlled trial. Neurology. 2010;74:413–420.
Birnbaum J, Kip M, Spies CD, et al. The effect of stimulation versus nonstimulating catheters for continuous interscalene plexus blocks in short-term pain management. J Clin Anesth. 2007;19:434–439.
Funk W, Podmelle F, Guiol C, et al. Aesthetic satisfaction scoring- introducing an aesthetic numeric analogue scale (ANA-scale). J Craniomaxillofac Surg. 2011;40:439–442.
Gossec L, Paternotte S, Aanerud GJ, et al. Finalisation and validation of the rheumatoid arthritis impact of disease score, a patient-derived composite measure of impact of rheumatoid arthritis: a EULAR initiative. Ann Rheum Dis. 2011;70:935–942.
Wig J, Chandrashekharappa KN, Yaddanapudi LN, et al. Effect of prophylactic odansetron on postoperative nausea and vomiting in patients on preoperative steroids undergoing craniotomy for supratnetorial tumors. J Neurosurg Anesthesiol. 2007;19:239–242.
Rabin R, De Charro F. EQ-5D: a measure of health status from the EuroQol Group. Ann Med. 2001;33:337–343.
Stermer E, Gaitini L, Yudashkin M, et al. Patient-controlled analgesia for conscious sedation during colonoscopy: a randomized controlled study. Gastrointest Endosc. 2000;5:278–281.
Spiegel B, Bolus R, Harris LA, et al. Measuring irritable bowel syndrome patient-reported outcomes with an abdominal pain numeric rating scale. Aliment Pharmacol Ther. 2009;30:1159–1170.
Livingston PM, Craike MJ, White VM, et al. A nurse-assisted screening and referral program for depression among survivors of colorectal cancer: feasibility study. Med J Aust. 2010;193:S83–S87.
Wilcox AR, Dragnev MCC, Darcey CJ, et al. A new tool to measure burden of Crohn’s disease and its treatment: do patient and physician perceptions match? Inflamm Bowel Dis. 2010;16:645–650.
Torrance GW. Measurement of health state utilities for economic appraisal. J Health Econ. 1986;5:1–30.
Best WR, Becktel JM, Singleton JW, et al. Development of a Crohn's disease activity index. National Cooperative Crohn's Disease Study. Gastroenterology. 1976;70:439–444.
Harvey RF, Bradshaw JM. A simple index of Crohn’s disease activity. Lancet. 1980;1:514.
Hlavaty T, Persoons P, Vermeire S, et al. Evaluation of short-term responsiveness and cutoff values for inflammatory bowel disease questionnaire in Crohn’s Disease. Inflamm Bowel Dis. 2006;12:199–204.
Guyatt G, Mitchell A, Irvine EJ, et al. A new measure of health status for clinical trials in inflammatory bowel disease. Gastroenterology. 1989;96:804–810.
Pallis AG, Mouzas IA, Vlachonikolis IG, et al. The inflammatory bowel disease questionnaire. A review of its national validation studies. Inflamm Bowel Dis. 2004;10:261–269.
Walmsley RS, Ayres RC, Pounder RE, et al. A simple clinical colitis activity index. Gut. 1998;43:29–32.
Cohen J. A power primer. Psychol Bull. 1992;112:155–159.
Irvine EJ, Feagan B, Rochon J, et al. Quality of life: a valid and reliable measure of therapeutic efficacy in the treatment of inflammatory bowel disease. Canadian Crohn’s relapse prevention trial study group. Gastroenterology. 1994;106:287–296.
Gray WN, Denson LA, Baldassano RN, et al. Disease activity, behavioral dysfunction, and health-related quality of life in adolescents with inflammatory bowel disease. Inflamm Bowel Dis. 2011;17:1581–1586.
Banovic I, Gilibert D, Cosnes J. Crohn’s disease and fatigue: constancy and co-variations of activity of the disease, depression, anxiety, and subjective quality of life. Psychol Health Med.. 2010;15:394–405.
Kanwal F, Hays RD, Kilbourne AM, et al. Are physician-derived disease severity indices associated with health-related quality of life in patients with end-stage liver disease? Am J Gastroenterol. 2004;99:1726–1732.
Borstlap M, van de Laar M, Zant J, et al. Components of health: an analysis in rheumatoid arthritis using quality of life questionnaires and clinical and laboratory variables. Ann Rheum Dis. 1993;52:650–654.
Bardsley MJ, Astell S, McCallum A, et al. The performance of three measures of health status in an outpatient diabetes population. Diabet Med. 1993;10:619–626.
Beto JA, Bansal VK. Quality of life in treatment of hypertension: a metaanalysis of clinical trials. Am J Hypertens. 1992;5:125–133.
Laupacis A, Wong C, Churchill D. The use of generic and specific quality of life measures in hemodialysis patients treated with erythropoietin. The Canadian Erythropoietin Study Group. Control Clin Trials. 1991;12:168S–179S.
Hjortswang H, Almer S, Strom M. The network: a strategy to describe the relationship between quality of life and disease activity. The case of inflammatory bowel disease. Eur J Gastroenterol Hepatol. 1999;11:1099–1104.
Drossman DA, Leserman J, Li Z, et al. The rating form of IBD patient concerns: a new measure of health status. Psychosom Med. 1991;53:701–712.
Denis MA, Reenaers C, Fontaine F, et al. Assessment of endoscopic activity index and biological inflammatory markers in clinically active Crohn’s disease with normal C-reactive protein serum level. Inflam Bowel Dis. 2007;13:1100–1105.
Lewis JD. The utility of biomarkers in the diagnosis and therapy of inflammatory bowel disease. Gastroenterology. 2011;140:1817–1826.
Cakal B, Akoz AG, Ustundag Y, et al. Red cell distribution width for assessment of activity of inflammatory bowel disease. Dig Dis Sci. 2009;54:842–847.
Rodgers AD, Cummins AG. CRP correlated with clinical score in ulcerative colitis but no in Crohn’s disease. Dig Dis Sci. 2007;52:2063–2068.
Drossman DA, Patrick DL, Mitchell CM, et al. Health related quality of life in inflammatory bowel disease: functional status and patient worries and concerns. Dig Dis Sci. 1989;34:1379–1386.
Rubin DT, Siegel CA, Kane SV, et al. Impact of ulcerative colitis from patients’ and physicians’ perspectives: results from the UC: NORMAL survey. Inflamm Bowel Dis. 2009;15:581–588.
Sewitch MJ, Abrahamowicz M, Dobkin PL, et al. Measuring differences between patients’ and physicians’ health perceptions: the patient-physician discordance scale. J Behav Med. 2003;26:245–264.
Westwood N, Travis SPL. Review article: what do patients with inflammatory bowel disease want for their clinical management? Aliment Pharmacol Ther. 2008;27:1–8.
Wolfe BJ, Sirois FM. Beyond standard quality of life measures: the subjective experiences of living with inflammatory bowel disease. Qual Life Res. 2009;17:877–886.
Leeb BF, Sautner J, Leeb BA, et al. Lack of agreement between patients’ and physicians’ perspectives of rheumatoid arthritis disease activity changes. Scand J Rheumatol. 2006;35:441–446.
Rothwell PM, MacDowell Z, Wong CK, et al. Doctors and patients don’t agree: cross sectional study of patients’ and doctors’ perceptions and assessments of disability in multiple sclerosis. Br Med J. 1997;314:1580–1583.
Leong KP, Chong EY, Kong KO, et al. Discordant assessment of lupus activity between patients and their physicians: the Singapore experience. Lupus. 2010;19:100–106.
Slevin ML, Plant H, Lynch D, et al. Who should measure quality of life, the doctor or the patient? Br J Cancer. 1988;57:109–112.
Acknowledgments
This study was funded in part by an unrestricted Investigator-Initiated grant from Centocor, Inc. The authors gratefully acknowledge Dr Fasiha Kanwal and Dr Ron Hays (UCLA) for methodologic input and Dr Patricio Ibanez (Cedars-Sinai) for assistance with patient recruitment. IBD Research at Cedars-Sinai that contributed to this study is supported by USPHS grant PO1DK046763, DK062413 and the Cedars-Sinai F. Widjaja Foundation Inflammatory Bowel and Immunobiology Research Institute Research Funds. Project investigators are support by The Helmsley Foundation, The European Union and the Crohn's and Colitis Foundation of America (D.P.B.M.), The Feintech Family Chair in IBD (S.R.T.), The Joshua L. and Lisa Z. Greer Chair in IBD Genetics (D.P.B.M.).
Conflict of interest
G.Y.M: Consultant (Amgen, Celgene, Janssen (formerly Centrocor), Given Imaging), non-CME speaker (Prometheus, Abbott), research support (Centocor).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Surti, B., Spiegel, B., Ippoliti, A. et al. Assessing Health Status in Inflammatory Bowel Disease Using a Novel Single-Item Numeric Rating Scale. Dig Dis Sci 58, 1313–1321 (2013). https://doi.org/10.1007/s10620-012-2500-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-012-2500-1