Abstract
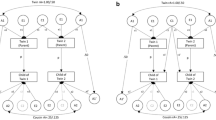
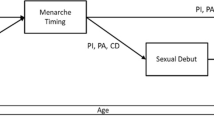
Age at menarche (AAM), time of first menstrual period, is an important developmental milestone in females. Follow-up data from 1,302 adolescent twins and their sisters were used to partition the normal variation in AAM. The proportion of censoring was 14.1%. Both a standard and a survival analysis method were used. The best fitting model from the survival analysis method was an ACE model, where 57% and 23% of the variance in AAM was explained by additive genetic and environmental effects, respectively. The best fitting model when using a standard variance decomposition method was an AE model, where 82% of the variance was explained by additive genetic effects. The lack of correspondence between the results of the two methods was an artefact of the different ascertainment of AAM reports from siblings and twins. After the removal of the sibling sample, both methods indicated that an ACE model was the most likely. Standard and survival analysis methods estimated the proportion of variance explained by additive effects to be 0.50 and 0.54, and common environmental effects to be 0.31 and 0.29, respectively. We conclude that variation in AAM can be explained by additive genetic and common environmental components.

Similar content being viewed by others
References
Anderson CA, McRae AF, Visscher PM (2006) A simple linear regression method for quantitative trait loci linkage analysis with censored observations. Genetics 173:1735–1745
Damon A, Bajema CJ (1974) Age at menarche of recall after 39 years. Human Biol 46:381–384
Damon A, Damon ST, Reed RB, Valadian I (1969) Age at menarche of mothers and daughters, with a note on accuracy of recall. Human Biol 41:161–182
Eaves L, Silberg J, Foley D, Bulik C, Maes H, Erkanli A, Angold A, Costello EJ, Worthman C (2004) Genetic and environmental influences on the relative timing of pubertal change. Twin Res 7:471–481
Kaltiala-Heino R, Kosunen E, Rimpela M (2003) Pubertal timing, sexual behaviour and self-reported depression in middle adolescence. J Adolesc 26:531–545
Kaltiala-Heino R, Rimpella M, Rissanen A, Rantanen P (2001) Early puberty and early sexual activity are associated with bulimic-type eating pathology in middle adolescence. J Adolesc Health 28:346–352
Kaprio J, Rimpela A, WinterT, Viken RJ, Rimpela M, Rose RJ (1995) Common genetic influences on BMI and menarche. Human Biol 67:739–753
Kirk KM, Blomberg SP, Duffy DL, Heath AC, Owens IPF, Martin NG (2001) Natural selection and quantitative genetics of life-history traits in western women: a twin study. Evolution 55:423–435
Klein JP, Moeschberger ML (1999) Survival analysis: techniques for censored and truncated data. Springer-Verlag, New York, NY, USA
Koeppen-Schomerus G, Spinath FM, Plomin R (2003) Twins and non-twin siblings: different estimates of shared environmental influence in early childhood. Twin Res 6:97–105
Koo MM, Rohan TE (1997) Accuracy of short-term recall of age at menarche. Ann Human Biol 24:61–64
Loesch DZ, Huggins R, Rogucka E, Hoang NH, Hopper JL (1995). Genetic correlates of menarcheal age: a multivariate twin study. Ann Human Biol 22:479–490
Meyer JM, Eaves LJ, Heath AC, Martin NG (1991) Estimating genetic influences on the age-at-menarche: a survival analysis approach. Am J Med Genet 39:148–154
Neale MC, Boker SM, Xie G, Maes HH (2002) MX: Statistical Modeling
R Development Core Team (2005) R: A language and environment for statistical computing
Ripatti S, Palmgren J (2000) Estimation of multivariate frailty models using penalized partial likelihood. Biometrics 56:1016–1022
Roche AF (1992) Growth, maturation and body composition: the Fels Longitudinal Study. Cambridge University Press, Cambridge
Schneider MdP, Strandberg E, Ducrocq V, Roth A (2005) Survival analysis applied to genetic evaluation for female fertility in dairy cattle. J Dairy Sci 88:2253–2259
Self SG, Liang K-Y (1987) Asymptotic properties of maximum likelihood estimators and likelihood ratio tests under nonstandard conditions. J Am Stat Assoc 82:605–610
Stram DO, Lee JW (1994) Variance components testing in the longitudinal mixed effects model. Biometrics 50:1171–1177
Therneau T (2003) On mixed-effect Cox models, sparse matrices, and modeling data from large pedigrees. Mayo Clinic, Rochester, USA
Towne B, Czerwinski SA, Demerath EW, Blangero J, Roche AF, Siervogel RM (2005) Heritability of age at menarche in girls from the Fels Longitudinal Study. Am J Phys Anthropol 128:210–219
Treloar SA, Martin NG (1990) Age at menarche as a fitness trait: nonadditive genetic variance detected in a large twin sample. Am J Human Genet 47:137–148
van den Berg SM, Boomsma DI (2007) The familial clustering of age at menarche in extended twin families. Behav Genet (Epub Ahead of Print)
van den Berg SM, Setiawan A, Bartels M, Polderman TJ, Vaart AWvd, Boomsma DI (2006) Individual differences in puberty onset in girls: Bayesian estimation of heritabilities and genetic correlations. Behav Genet 36:261–270
Velie EM, Nechuta S, Osuch JR (2006) Lifetime reproductive and anthropometric risk factors for breast cancer in postmenopausal women. Breast Dis 24:17–35
Wei LJ, Lin DY, Weissfeld L (1989) Regression analysis of multivariate incomplete failure time data by modelling marginal distributions. J Am Stat Assoc 84:1065–1073
Wright M, Geus Ed, Ando J, Luciano M, Posthuma D, Ono Y, Hansell N, Baal Cv, Hiraishi K, Hasegawa T, Smith G, Geffen G, Geffen L, Kanba S, Miyake A, Martin N, Boomsma D (2001) Genetics of cognition: outline of a collaborative twin study. Twin Res 4:48–56
Xue X, Brookmeyer R (1996) Bivariate frailty model for the analysis of multivariate survival times. Lifetime Data Anal 2:277–289
Yazdi MH, Visscher PM, Ducrocq V, Thompson R (2002) Heritability, reliability of genetic evaluations and response to selection in proportional hazards models. J Dairy Sci 85:1563–1577
Zhao JH (2005) Mixed effects Cox models of alcohol dependence in extended families. BMC Genet 6(Suppl 1):S127
Zhu G, Duffy DL, Eldridge A, Grace M, Mayne C, OGorman L, Aitken JF, Neale MC, Hayward NK, Green AC, Martin NG (1999) A major quantitative-trait locus for mole density is linked to the familial melanoma gene CDKN2A: a maximum-likelihood combined linkage and association analysis in twins and their sibs. Am J Human Genet 65:483–492
Acknowledgements
We would like to thank all the twins and their families for participating in the study and Ann Eldridge and Marlene Grace for ascertaining the age at menarche data. Thanks to Sri Shekar for an excellent introduction to MX. CAA was funded by a Medical Research Council (UK) postgraduate research studentship. The adolescent twin studies were supported by the Australian Research Council (A79600334, A79801419, A79906588, DP0212016), the Human Frontier Science Program (RG0154.1998-B), the NHMRC (950998, 981339, 241944), and the National Institutes of Health (CA88363). The work was also supported by an NHMRC Bioinformatics program grant awarded to PMV (389982). We would also like to thank the editor and three anonymous reviewers for constructive comments on an earlier version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by Dorret Boomsma
Rights and permissions
About this article
Cite this article
Anderson, C.A., Duffy, D.L., Martin, N.G. et al. Estimation of Variance Components for Age at Menarche in Twin Families. Behav Genet 37, 668–677 (2007). https://doi.org/10.1007/s10519-007-9163-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10519-007-9163-2