Abstract
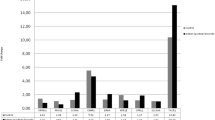
Autism spectrum disorders (ASD) comprise neurodevelopmental disorders with clinical onset during the first years of life. The identification of peripheral biomarkers could significantly impact diagnosis and an individualized, early treatment. Although the aetiology of ASD remains poorly understood, there is increasing evidence that neurotrophins and their receptors represent a group of candidate genes for ASD pathophysiology and biomarker research. Total messenger RNA (mRNA) from whole blood was obtained from adolescents and adults diagnosed as ASD (n = 21) according to DSM-IV criteria and confirmed by the Autism Diagnostic Observation Schedule (ADOS) and Autism Diagnostic Interview-Revised (ADI-R) algorithms, as well as healthy controls (n = 10). The mRNA expression of neurotrophins (BDNF, NT3 and NT4) and their receptors (TrkA, TrkB and p75 NTR) was determined by quantitative real-time polymerase chain reaction (qRT-PCR). Moreover, social cognition abilities of ASD patients and controls were determined according to three Theory of Mind (ToM) tests (Reading the Mind in the Eyes, Faux pas, and Happé stories). The NT3 and NT4 mRNA expression in the whole blood was significantly lower in ASD compared to healthy controls, while p75NTR was higher (P < 0.005). In addition, lower scores in three of the ToM tests were observed in ASD subjects compared to controls. A significant (P < 0.005) ToM impairment in Happé stories test was demonstrated in ASD. Nevertheless, no correlations were observed between neurotrophins and their receptors expressions and measures of ToM. Given their potential as peripheral blood-based biomarkers, NT3, NT4 and p75 NTR mRNA expression patterns may be useful tools for a more personalized diagnostics and therapy in ASD. Further investigations with larger numbers of samples are needed to verify these results.

Similar content being viewed by others
References
Baird G, Simonoff E, Pickles A, Chandler S, Loucas T, Meldrum D et al (2006) Prevalence of disorders of the autism spectrum in a population cohort of children in South Thames: the Special Needs and Autism Project (SNAP). Lancet 368(9531):210–215
Howlin P, Goode S, Hutton J, Rutter M (2004) Adult outcome for children with autism. J Child Psychol Psychiatry 45(2):212–229
Zwaigenbaum L, Bryson S, Rogers T, Roberts W, Brian J, Szatmari P (2005) Behavioral manifestations of autism in the first year of life. Int J Dev Neurosci 23(2–3):143–152
Volkmar FR, Lord C, Bailey A, Schultz RT, Klin A (2004) Autism and pervasive developmental disorders. J Child Psychol Psychiatry 45(1):135–170
Pardo CA, Eberhart CG (2007) The neurobiology of autism. Brain Pathol 17(4):434–447
Belmonte MK, Allen G, Beckel-Mitchener A, Boulanger LM, Carper RA, Webb SJ (2004) Autism and abnormal development of brain connectivity. J Neurosci 24(42):9228–9231
Polleux F, Lauder JM (2004) Toward a developmental neurobiology of autism. Ment Retard Dev Disabil Res Rev 10(4):303–317
Folstein SE, Rosen-Sheidley B (2001) Genetics of autism: complex aetiology for a heterogeneous disorder. Nat Rev Genet 2(12):943–955
Freitag CM, Agelopoulos K, Huy E, Rothermundt M, Krakowitzky P, Meyer J, Deckert J, von Gontard A, Hohoff C (2010) Adenosine A(2A) receptor gene (ADORA2A) variants may increase autistic symptoms and anxiety in autism spectrum disorder. Eur Child Adolesc Psychiatry 19(1):67–74
Ruggeri B, Sarkans U, Schumann G, Persico AM (2014) Biomarkers in autism spectrum disorder: the old and the new. Psychopharmacology (Berl) 231(6):1201–1216
Glatt SJ, Tsuang MT, Winn M, Chandler SD, Collins M, Lopez L, Weinfeld M, Carter C, Schork N, Pierce K, Courchesne E (2012) Blood-based gene expression signatures of infants and toddlers with autism. J Am Acad Child Adolesc Psychiatry 51(9):934.e2–944.e2
Kong SW, Collins CD, Shimizu-Motohashi Y, Holm IA, Campbell MG, Lee IH, Brewster SJ, Hanson E, Harris HK, Lowe KR, Saada A, Mora A, Madison K, Hundley R, Egan J, McCarthy J, Eran A, Galdzicki M, Rappaport L, Kunkel LM, Kohane IS (2012) Characteristics and predictive value of blood transcriptome signature in males with autism spectrum disorders. PLoS One 7(12):e4947
Nickl-Jockschat T, Michel TM (2011) The role of neurotrophic factors in autism. Mol Psychiatry 16(5):478–490
Lang UE, Jockers-Scherübl MC, Hellweg R (2004) State of the art of the neurotrophin hypothesis in psychiatric disorders: implications and limitations. J Neural Transm 111(3):387–411
Leibrock J, Lottspeich F, Hohn A, Hofer M, Hengerer B, Masiakowski P et al (1989) Molecular cloning and expression of brain-derived neurotrophic factor. Nature 341(6238):149–152
Ernfors P, Ibanez CF, Ebendal T, Olson L, Persson H (1990) Molecular cloning and neurotrophic activities of a protein with structural similarities to nerve growth factor: developmental and topographical expression in the brain. Proc Natl Acad Sci 87(14):5454–5458
Klein R, Nanduri V, Jing S, Lamballe F, Tapley P, Bryant S et al (1991) The trkB tyrosine protein kinase is a receptor for brain-derived neurotrophic factor and neurotrophin-3. Cell 66(2):395–403
Ip NY, Ibáñez CF, Nye SH, Mcclain J, Jones PF, Gies DR et al (1992) Mammalian neurotrophin-4: structure, chromosomal localization, tissue distribution, and receptor specificity. Proc Natl Acad Sci U S A 89(7):3060–3064
Klein R, Jing S, Nanduri V, O’Rourke E, Barbacid M (1991) The trk proto-oncogene encodes a receptor for nerve growth factor. Cell 65(1):189–197
Lamballe F, Klein R, Barbacid M (1991) trkC, a new member of the trk family of tyrosine protein kinases, is a receptor for neurotrophin-3. Cell 66(5):967–979
Soppet D, Escandon E, Maragos J, Middlemas DS, Reid SW, Blair J et al (1991) The neurotrophic factors brain-derived neurotrophic factor and neurotrophin-3 are ligands for the trkB tyrosine kinase receptor. Cell 65(5):895–903
Squinto SP, Stitt TN, Aldrich TH, Davis S, Bianco SM, Radziejewski C et al (1991) trkB encodes a functional receptor for brain-derived neurotrophic factor and neurotrophin-3 but not nerve growth factor. Cell 65(5):885–893
Hallböök F, Ibáñez CF, Persson H (1991) Evolutionary studies of the nerve growth factor family reveal a novel member abundantly expressed in Xenopus ovary. Neuron 6(5):845–858
Lee KF, Bachman K, Landis S, Jaenisch R (1994) Dependence on p75 for innervation of some sympathetic targets. Science 263(5152):1447–1449
Connolly AM, Chez M, Streif EM, Keeling RM, Golumbek PT, Kwon JM et al (2006) Brain-derived neurotrophic factor and autoantibodies to neural antigens in sera of children with autistic spectrum disorders, Landau-Kleffner syndrome, and epilepsy. Biol Psychiatry 59(4):354–363
Miyazaki K, Narita N, Sakuta R, Miyahara T, Naruse H, Okado N et al (2004) Serum neurotrophin concentrations in autism and mental retardation: a pilot study. Brain Dev 26(5):292–295
Nelson KB, Grether JK, Croen LA, Dambrosia JM, Dickens BF, Jelliffe LL et al (2001) Neuropeptides and neurotrophins in neonatal blood of children with autism or mental retardation. Ann Neurol 49(5):597–606
Perry EK (2001) Cholinergic activity in autism: abnormalities in the cerebral cortex and basal forebrain. Am J Psychiatry 158(7):1058–1066
Correia CT, Coutinho AM, Sequeira AF, Sousa IG, Lourenço Venda L, Almeida JP et al (2010) Increased BDNF levels and NTRK2 gene association suggest a disruption of BDNF/TrkB signaling in autism. Genes Brain Behav 9(7):841–848
Hashimoto K, Iwata Y, Nakamura K, Tsujii M, Tsuchiya KJ, Sekine Y et al (2006) Reduced serum levels of brain-derived neurotrophic factor in adult male patients with autism. Prog Neuropsychopharmacol Biol Psychiatry 30(8):1529–1531
Abdallah MW, Mortensen EL, Greaves-Lord K, Larsen N, Bonefeld-Jørgensen EC, Nørgaard-Pedersen B et al (2013) Neonatal levels of neurotrophic factors and risk of autism spectrum disorders. Acta Psychiatr Scand 128(1):61–69
Taurines R, Segura M, Schecklmann M, Albantakis L, Grünblatt E, Walitza S, Jans T, Lyttwin B, Haberhausen M, Theisen FM, Martin B, Briegel W, Thome J, Schwenck C, Romanos M, Gerlach M (2014) Altered peripheral BDNF mRNA expression and BDNF protein concentrations in blood of children and adolescents with autism spectrum disorder. J Neural Transm 121(9):1117–1128
Viding E, Blakemore SJ (2007) Endophenotype approach to developmental psychopathology: implications for autism research. Behav Genet 37(1):51–60
Baron-Cohen S (1991) The theory of mind deficit in autism: how specific is it? Br J Dev Psychol 9:301–314
Happé FG (1994) An advanced test of theory of mind: understanding of story characters’ thoughts and feelings by able autistic, mentally handicapped, and normal children and adults. J Autism Dev Disord 24(2):129–154
Dawson G, Carver L, Meltzoff AN, Panagiotides H, McPartland J, Webb SJ (2002) Neural correlates of face and object recognition in young children with autisms spectrum disorder, developmental delay, and typical development. Child Dev 73(3):700–717
Pierce K, Muller RA, Ambrose J, Allen G, Curchesne E (2001) Face processing occurs outside the fusiform “face area” in autism: evidence from function MRI. Brain 124(Pt 10):2059–2073
Karege F, Schwald M, Cisse M (2002) Postnatal developmental profile of brain-derived neurotrophic factor in rat brain and platelets. Neurosci Lett 328(3):261–264
Katoh-Semba R, Wakako R, Komori T, Shigemi H, Miyazaki N, Ito H, Kumagai T, Tsuzuki M, Shigemi K, Yoshida F, Nakayama A (2007) Age-related changes in BDNF protein levels in human serum: differences between autism cases and normal controls. Int J Dev Neurosci 25(6):367–372
Lord C, Rutter M, Couteur A (1994) Autism diagnostic interview-revised: a revised version of a diagnostic interview for caregivers of individuals with possible pervasive developmental disorders. J Autism Dev Disord 24(5):659–685
Lord C, Risi S, Lambrecht L, Cook EH, Leventhal BL, Dilavore PC et al (2000) The autism diagnostic observation schedule-generic: a standard measure of social and communication deficits associated with the spectrum of autism. J Autism Dev Disord 30(3):205–223
Baron-Cohen S, Wheelwright S, Hill J, Raste Y, Plumb I (2001) The “Reading the Mind in the Eyes” test revised version: a study with normal adults, and adults with Asperger syndrome or high-functioning autism. J Child Psychol Psychiatry 42(2):241–251
Baron-Cohen S, Ring HA, Wheelwright S, Bullmore ET, Brammer MJ, Simmons A et al (1999) Social intelligence in the normal and autistic brain: an fMRI study. Eur J Neurosci 11(6):1891–1898
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A et al (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3(7), RESEARCH0034
Tostes MH, Teixeira HC, Gattaz WF, Brandão MA, Raposo NR (2012) Altered neurotrophin, neuropeptide, cytokines and nitric oxide levels in autism. Pharmacopsychiatry 45(6):241–243
Sajdel-Sulkowska EM, Xu M, McGinnis W, Koibuchi N (2011) Brain region-specific changes in oxidative stress and neurotrophin levels in autism spectrum disorders (ASD). Cerebellum 10(1):43–48
Xu M, Sajdel-Sulkowska EM, Iwasaki T, Koibuchi N (2013) Aberrant cerebellar neurotrophin-3 expression induced by lipopolysaccharide exposure during brain development. Cerebellum 12(3):316–318
Sherrard RM, Bower AJ (2002) Climbing fiber development: do neurotrophins have a part to play? Cerebellum 1(4):265–275
Whitney ER, Kemper TL, Rosene DL, Bauman ML, Blatt GJ (2009) Density of cerebellar basket and stellate cells in autism: evidence for a late developmental loss of Purkinje cells. J Neurosci Res 87(10):2245–2254
Ritvo ER, Freeman BJ, Scheibel AB, Duong T, Robinson H, Guthrie D, Ritvo A (1986) Lower Purkinje cell counts in the cerebella of four autistic subjects: initial findings of the UCLA-NSAC Autopsy Research Report. Am J Psychiatry 143(7):862–866
Mount HT, Dreyfus CF, Black IB (1994) Neurotrophin-3 selectively increases cultured Purkinje cell survival. Neuroreport 5(18):2497–2500
Sudarov A (2013) Defining the role of cerebellar Purkinje cells in autism spectrum disorders. Cerebellum 12(6):950–955
Bauman ML, Kemper TL (2005) Neuroanatomic observations of the brain in autism: a review and future directions. Int J Dev Neurosci 23(2–3):183–187
Coulson EJ (1999) p75 neurotrophin receptor-mediated neuronal death is promoted by Bcl-2 and prevented by Bcl-xL. J Biol Chem [Internet] 274(23):16387–16391
Frade JM, Rodríguez-Tébar A, Barde YA (1996) Induction of cell death by endogenous nerve growth factor through its p75 receptor. Nature 383(6596):166–168
Friedman WJ, Greene LA (1999) Neurotrophin signaling via Trks and p75. Exp Cell Res 253(1):131–142
Lee R, Kermani P, Teng KK, Hempstead BL (2001) Regulation of cell survival by secreted proneurotrophins. Science 294(5548):1945–1948
Volosin M, Song W, Almeida RD, Kaplan DR, Hempstead BL, Friedman WJ (2006) Interaction of survival and death signaling in basal forebrain neurons: roles of neurotrophins and proneurotrophins. J Neurosci 26(29):7756–7766
De Graaf-Peters VB, Hadders-Algra M (2006) Ontogeny of the human central nervous system: what is happening when? Early Hum Dev 82(4):257–266
Wei H, Alberts I, Li X (2014) The apoptotic perspective of autism. Int J Dev Neurosci 36:13–18
Sheikh AM, Malik M, Wen G, Chauhan A, Chauhan V, Gong CX, Liu F, Brown WT, Li X (2010) BDNF-Akt-Bcl2 antiapoptotic signaling pathway is compromised in the brain of autistic subjects. J Neurosci Res 88(12):2641–2647
Siniscalco D, Sapone A, Giordano C, Cirillo A, de Novellis V, de Magistris L, Rossi F, Fasano A, Maione S, Antonucci N (2012) The expression of caspases is enhanced in peripheral blood mononuclear cells of autism spectrum disorder patients. J Autism Dev Disord 42(7):1403–1410
Malik M, Sheikh AM, Wen G, Spivack W, Brown WT, Li X (2011) Expression of inflammatory cytokines, Bcl2 and cathepsin D are altered in lymphoblasts of autistic subjects. Immunobiology 216(1–2):80–85
Benedetti M, Levi A, Chao MV (1993) Differential expression of nerve growth factor receptors leads to altered binding affinity and neurotrophin responsiveness. Proc Natl Acad Sci U S A 90(16):7859–7863
Verdi JM, Birren SJ, Ibáñez CF, Persson H, Kaplan DR, Benedetti M, Chao MV, Anderson DJ (1994) p75LNGFR regulates Trk signal transduction and NGF-induced neuronal differentiation in MAH cells. Neuron 12(4):733–745
Gottesman II, Gould TD (2003) The endophenotype concept in psychiatry: etymology and strategic intentions. Am J Psychiatry 160:636–645
Warrier V, Baron-Cohen S, Chakrabarti B (2013) Genetic variation in GABRB3 is associated with Asperger syndrome and multiple endophenotypes relevant to autism. Mol Autism 4(1):48
Baron-Cohen S, O’Riordan M, Stone V, Jones R, Plaisted K (1999) Recognition of faux pas by normally developing children and children with Asperger syndrome or high-functioning autism. J Autism Dev Disord 29(5):407–418
Kaland N, Moller-Nielsen A, Callesen K, Mortensen EL, Gottlieb D, Smith L (2002) A new ‘advanced’ test of theory of mind: evidence from children and adolescents with Asperger syndrome. J Child Psychol Psychiatry 43(4):517–528
Spek AA, Scholte EM, Van Berckelaer-Onnes IA (2010) Theory of mind in adults with HFA and Asperger syndrome. J Autism Dev Disord 40(3):280–289
Zalla T, Sav AM, Stopin A, Ahade S, Leboyer M (2009) Faux pas detection and intentional action in Asperger Syndrome. A replication on a french sample. J Autism Dev Disord 39(2):373–382
Tager-Flusberg H, Sullivan K (2000) A componential view of theory of mind: evidence from Williams syndrome. Cognition 76(1):59–90
Ponnet KS, Roeyers H, Buysse A, De Clercq A, Van der Heyden E (2004) Advanced mind-reading in adults with Asperger syndrome. Autism 8(3):249–266
Kaland N, Callesen K, Møller-Nielsen A, Mortensen EL, Smith L (2008) Performance of children and adolescents with Asperger syndrome or high-functioning autism on advanced theory of mind tasks. J Autism Dev Disord 38(6):1112–1123
Scaccianoce S, Del Bianco P, Paolone G, Caprioli D, Modafferi AM, Nencini P, Badiani A (2006) Social isolation selectively reduces hippocampal brain-derived neurotrophic factor without altering plasma corticosterone. Behav Brain Res 168(2):323–325
Hashimoto K, Shimizu E, Iyo M (2004) Critical role of brain-derived neurotrophic factor in mood disorders. Brain Res Brain Res Rev 45(2):104–114, Review
Buitelaar JK, van der Wees M, Swaab-Barneveld H, van der Gaag RJ (1999) Theory of mind and emotion-recognition functioning in autistic spectrum disorders and in psychiatric control and normal children. Dev Psychopathol 11(1):39–58
Geurts HM, Verté S, Oosterlaan J, Roeyers H, Hartman CA, Mulder EJ, Berckelaer-Onnes IA, Sergeant JA (2004) Can the children’s communication checklist differentiate between children with autism, children with ADHD, and normal controls? J Child Psychol Psychiatry 45(8):1437–1453
Sergeant JA, Geurts H, Oosterlaan J (2002) How specific is a deficit of executive functioning for attention-deficit/hyperactivity disorder? Behav Brain Res 130(1–2):3–28, Review
Dorris L, Espie CA, Knott F, Salt J (2004) Mind-reading difficulties in the siblings of people with Asperger’s syndrome: evidence for a genetic influence in the abnormal development of a specific cognitive domain. J Child Psychol Psychiatry 45(2):412–418
Castelli F, Frith C, Happé F, Frith U (2002) Autism, Asperger syndrome and brain mechanisms for the attribution of mental states to animated shapes. Brain 125(Pt 8):1839–1849
Sullivan PF, Fan C, Perou CM (2006) Evaluating the comparability of gene expression in blood and brain. Am J Med Genet B Neuropsychiatr Genet 141B(3):261–268
Rollins B, Martin MV, Morgan L, Vawter MP (2010) Analysis of whole genome biomarker expression in blood and brain. Am J Med Genet B Neuropsychiatr Genet 153B(4):919–936
Hayashi-Takagi A, Vawter MP, Iwamoto K (2014) Peripheral biomarkers revisited: integrative profiling of peripheral samples for psychiatric research. Biol Psychiatry 75(12):920–928
Acknowledgments
This research was supported by an unrestricted research grant from María Francisca de Roviralta Fundation (Madrid, Spain). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. This work has been also funded by the FI-DRG2010 grant and the BE-DRG2010 grant sponsored by the Departament d’Educació i Universitats de la Generalitat de Catalunya and European Social Fund. We are grateful to all patients and controls for their participation in our study, to clinical collaborators for patient’s assessment and to Irene Bolea for critical comments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Mònica Segura and Carla Pedreño contributed equally to this work.
Rights and permissions
About this article
Cite this article
Segura, M., Pedreño, C., Obiols, J. et al. Neurotrophin blood-based gene expression and social cognition analysis in patients with autism spectrum disorder. Neurogenetics 16, 123–131 (2015). https://doi.org/10.1007/s10048-014-0434-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10048-014-0434-9