Abstract
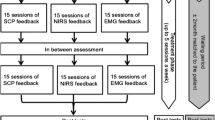
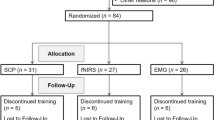
Despite sizeable short-term effects of neurofeedback (NF) therapy on attention-deficit and hyperactivity disorder (ADHD), longer-term clinical, comorbidity and self-regulation outcomes are less systematically studied. The aim of this largest NF follow-up to date was to evaluate these outcomes 6 months after NF compared to a semi-active control to disentangle specific from unspecific sustained effects. We performed a multicenter, randomized, parallel, controlled, clinical, superiority trial in five German university outpatient departments. Participants were eligible if they fulfilled DSM-IV-TR criteria for ADHD and were aged from 7 to 9 years. Participants were randomly assigned (1:1-ratio) to 25 sessions of slow cortical potential (SCP)-NF or electromyogram biofeedback (EMG-BF). Participants were not blinded, since they received instructions according to each treatment setting. Primary outcomes were parent ratings of ADHD. The trial was registered, number ISRCTN761871859. Both groups showed improvement of ADHD symptoms compared to baseline at 6-months follow-up with large effect sizes for SCP-NF (d = 1.04) and EMG-BF (d = 0.85), but without group differences. When analyzing all assessments (pre-test, post-test-1, post-test-2 and follow-up), a group-by-time interaction emerged (p = 0.0062), with SCP-NF showing stable improvement following treatment but EMG-BF showing a relapse from post-test-1 to post-test-2, and subsequent remission at follow-up. Six months after the end of treatment, improvement after SCP-NF remained large and stable. However, the lack of group differences at follow-up suggests shared specific and unspecific effects contributing to this clinical outcome. Our correlational results indicate specificity of SCP-NF for selected subscales after training, but not at follow-up.


Similar content being viewed by others
References
Polanczyk GV, Salum GA, Sugaya LS, Caye A, Rohde LA (2015) Annual research review: a meta-analysis of the worldwide prevalence of mental disorders in children and adolescents. J Child Psychol Psychiatry 56(3):345–365
Spence T, Biederman J, Wilens T, Harding M, O’Donnell D, Griffin S (1996) Pharmacotherapy of attention-deficit hyperactivity disorder across the life cycle. J Am Acad Child Adolesc Psychiatry 35(4):409–432
Graham J, Banaschewski T, Buitelaar J, Coghill D, Danckaerts M, Dittmann RW et al (2011) European guidelines on managing adverse effects of medication for ADHD. Eur Child Adolesc Psychiatry 20(1):17–37
Berger I, Dor T, Nevo Y, Goldzweig G (2008) Attitudes toward attention-deficit hyperactivity disorder (ADHD) treatment: parents’ and children’s perspectives. J Child Neurol 23(9):1036–1042
van de Loo-Neus GHH, Rommelse N, Buitelaar JK (2011) To stop or not to stop? How long should medication treatment of attention-deficit hyperactivity disorder be extended? Eur Neuropsychopharmacol 21(8):584–599
Sherlin LH, Arns M, Lubar J, Heinrich H, Kerson C, Strehl U et al (2011) Neurofeedback and basic learning theory: implications for research and practice. J Neurother 15(4):292–304
Arns M, de Ridder S, Strehl U, Breteler M, Coenen A (2009) Efficacy of neurofeedback treatment in ADHD: the effects on inattention, impulsivity and hyperactivity: a meta-analysis. Clin EEG Neurosci 40(3):180–189
Cortese S, Ferrin M, Brandeis D, Holtmann M, Aggensteiner P, Daley D, Santosh P, Simonoff E, Stevenson J, Stringaris A, Sonuga-Barke EJ (2016) Neurofeedback for attention-deficit/hyperactivity disorder: meta-analysis of clinical and neuropsychological outcomes from randomized controlled trials. J Am Acad Child Adolesc Psychiatry 55(6):444–455
Doren JV, Arns M, Heinrich H, Vollebregt MA, Strehl U, Loo SK (2018) Sustained effects of neurofeedback in ADHD: a systematic review and meta-analysis. Eur Child Adolesc Psychiatry 14:1–13
Saylor KE, Amann BH (2016) Impulsive aggression as a comorbidity of attention-deficit/hyperactivity disorder in children and adolescents. J Child Adolesc Psychopharmacol 26(1):19–25
Daley D, van der Oord S, Ferrin M, Danckaerts M, Doepfner M, Cortese S et al (2014) Behavioral interventions in attention-deficit/hyperactivity disorder: a meta-analysis of randomized controlled trials across multiple outcome domains. J Am Acad Child Adolesc Psychiatry 53(8):835–847.e5
Rockstroh B, Elbert T, Lutzenberger W, Birbaumer N (1990) Biofeedback: evaluation and therapy in children with attentional dysfunctions. In: Rothenberger A (ed) Brain and Behavior in Child Psychiatry. Springer, Berlin, pp 345–357
Albrecht B, Brandeis D, Uebel H, Heinrich H, Mueller UC, Hasselhorn M et al (2008) Action monitoring in boys with attention-deficit/hyperactivity disorder, their nonaffected siblings, and normal control subjects: evidence for an endophenotype. Biol Psychiatry 64(7):615–625
Drechsler R, Straub M, Doehnert M, Heinrich H, Steinhausen H-C, Brandeis D (2007) Controlled evaluation of a neurofeedback training of slow cortical potentials in children with Attention Deficit/Hyperactivity Disorder (ADHD). Behav Brain Funct 26(3):35
Gevensleben H, Holl B, Albrecht B, Vogel C, Schlamp D, Kratz O et al (2009) Is neurofeedback an efficacious treatment for ADHD? A randomised controlled clinical trial. J Child Psychol Psychiatry 50(7):780–789
Holtmann M, Stadler C, Leins U, Strehl U, Birbaumer N, Poustka F (2004) Neurofeedback for the treatment of attention-deficit/hyperactivity disorder (ADHD) in childhood and adolescence. Z Für Kinder Jugendpsychiatrie Psychother 32(3):187–200
Strehl U, Leins U, Goth G, Klinger C, Hinterberger T, Birbaumer N (2006) Self-regulation of slow cortical potentials: a new treatment for children with attention-deficit/hyperactivity disorder. Pediatrics 118(5):e1530–e1540
Strehl U, Aggensteiner P, Wachtlin D, Brandeis D, Albrecht B, Arana M, Bach C, Banaschewski T, Bogen T, Flaig-Röhr A, Freitag CM, Fuchsenberger Y, Gest S, Gevensleben H, Herde L, Hohmann S, Legenbauer T, Marx AM, Millenet S, Pniewski B, Rothenberger A, Ruckes C, Wörz S, Holtmann M (2017) Neurofeedback of slow cortical potentials in children with attention-deficit/hyperactivity disorder: a multicenter randomized trial controlling for unspecific effects. Front Hum Neurosci 11:135
Meisel V, Servera M, Garcia-Banda G, Cardo E, Moreno I (2013) Neurofeedback and standard pharmacological intervention in ADHD: a randomized controlled trial with six-month follow-up. Biol Psychol 94(1):12–21
Konicar L, Veit R, Eisenbarth H, Barth B, Tonin P, Strehl U, Birbaumer N (2015) Brain self-regulation in criminal psychopaths. Sci Rep 5:9426
Janssen TWP, Bink M, Weeda WD, Geladé K, van Mourik R, Maras A et al (2016) Learning curves of theta/beta neurofeedback in children with ADHD. Eur Child Adolesc Psychiatry 19:1–10
Holtmann M, Pniewski B, Wachtlin D, Worz S, Strehl U (2014) Neurofeedback in children with attention-deficit/hyperactivity disorder (ADHD)—a controlled multicenter study of a non-pharmacological treatment approach. BMC Pediatr 13(14):202
Monastra VJ, Monastra DM, George S (2002) The effects of stimulant therapy, EEG biofeedback, and parenting style on the primary symptoms of attention-deficit/hyperactivity disorder. Appl Psychophysiol Biofeedback 27(4):231–249
Biederman J, Mick E, Faraone SV (2000) Age-dependent decline of symptoms of attention deficit hyperactivity disorder: impact of remission definition and symptom type. Am J Psychiatry 157(5):816–818
Maurizio S, Liechti MD, Heinrich H, Jäncke L, Steinhausen HC, Walitza S et al (2014) Comparing tomographic EEG neurofeedback and EMG biofeedback in children with attention-deficit/ hyperactivity disorder. Biol Psychol 95:31–44
Barth B, Mayer K, Strehl U, Fallgatter AJ, Ehlis A-C (2017) EMG biofeedback training in adult attention-deficit/hyperactivity disorder: an active (control) training? Behav Brain Res 30(329):58–66
Geladé K, Janssen TWP, Bink M, Twisk JWR, van Mourik R, Maras A et al (2017) A 6-month follow-up of an RCT on behavioral and neurocognitive effects of neurofeedback in children with ADHD. Eur Child Adolesc Psychiatry 2:1–13
Minder F, Zuberer A, Brandeis D, Drechsler R (2018) Informant-related effects of neurofeedback and cognitive training in children with ADHD including a waiting control phase: a randomized-controlled trial. Eur Child Adolesc Psychiatry 27(8):1055–1066
Takahashi J, Yasumura A, Nakagawa E, Inagaki M (2014) Changes in negative and positive EEG shifts during slow cortical potential training in children with attention-deficit/hyperactivity disorder: a preliminary investigation. NeuroReport 25:618–624
Baumeister S, Wolf I, Holz N, Boecker R, Adamo N, Holtmann M et al (2016) Neurofeedback training effects on inhibitory brain activation in ADHD: a matter of learning? Neuroscience 378:89–99
Acknowledgements
We thank all children and their parents who participated in the study. We thank our study nurse Brigitta Gehrig, physiotherapist Doris Brötz, for help with the EMG feedback, Christina Schwenck, Ph.D., for supervision of clinical diagnosis and recruitment, our medical and psychology students for their help in data collection and management, and neuroCare for technical support.
Funding
This study was funded by the German Research Foundation DFG HO 2503/4-1 and 2503/4-2. The funding body had no influence on the study design, collection, analysis and interpretation of data, the writing of the report, or the decision to submit the paper for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
AR is member of an advisory board and speakers’ bureau of Lilly, Shire, Medice and Novartis. He has received research and travel support and an educational grant from Shire. CF has received speaker’s fees from Eli Lilly and Shire. TB has served in an advisory or consultancy role for Hexal Pharma, Lilly, Medice, Novartis, Otsuka, Oxford Outcomes, PCM Scientific, Shire and Vifor Pharma. He has received conference attendance support and conference support or received speaker’s fees from Lilly, Medice, Novartis and Shire. He is/has been involved in clinical trials conducted by Lilly, Shire and Vifor Pharma. MH has served in an advisory or consultancy role for Medice and Shire, and has received conference attendance support or was paid for public speaking by Lilly, Medice, neuroConn and Shire. US has been paid for public speaking by neuroCare, the German Society for Biofeedback. DB serves as an unpaid scientific consultant of an EU-funded neurofeedback trial. The other authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Aggensteiner, PM., Brandeis, D., Millenet, S. et al. Slow cortical potentials neurofeedback in children with ADHD: comorbidity, self-regulation and clinical outcomes 6 months after treatment in a multicenter randomized controlled trial. Eur Child Adolesc Psychiatry 28, 1087–1095 (2019). https://doi.org/10.1007/s00787-018-01271-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00787-018-01271-8