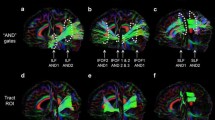
Abstract
While there is ample evidence that the structure and function of visual cortical areas are affected by early visual deprivation, little is known of how early blindness modifies subcortical relay and association thalamic nuclei, as well as mesencephalic structures. Therefore, in the present multicenter study, we used MRI to measure volume of the superior and inferior colliculi, as well as of the thalamic nuclei relaying sensory and motor information to the neocortex, parcellated according to atlas-based thalamo-cortical connections, in 29 individuals with congenital blindness of peripheral origin (17 M, age 35.7 ± 14.3 years) and 29 sighted subjects (17 M, age 31.9 ± 9.0). Blind participants showed an overall volume reduction in the left (p = 0.008) and right (p = 0.007) thalami, as compared to the sighted individuals. Specifically, the lateral geniculate (i.e., primary visual thalamic relay nucleus) was 40 % reduced (left: p = 4 × 10−6, right: p < 1 × 10−6), consistent with findings from animal studies. In addition, associated thalamic nuclei that project to temporal (left: p = 0.005, right: p = 0.005), prefrontal (left: p = 0.010, right: p = 0.014), occipital (left: p = 0.005, right: p = 0.023), and right premotor (p = 0.024) cortical regions were also significantly reduced in the congenitally blind group. Conversely, volumes of the relay nuclei directly involved in auditory, motor, and somatosensory processing were not affected by visual deprivation. In contrast, no difference in volume was observed in either the superior or the inferior colliculus between the two groups. Our findings indicate that visual loss since birth leads to selective volumetric changes within diencephalic, but not mesencephalic, structures. Both changes in reciprocal cortico-thalamic connections or modifications in the intrinsic connectivity between relay and association nuclei of the thalamus may contribute to explain these alterations in thalamic volumes. Sparing of the superior colliculi is in line with their composite, multisensory projections, and with their not exclusive visual nature.




Similar content being viewed by others
References
Andrews TJ, Halpern SD, Purves D (1997) Correlated size variations in human visual cortex, lateral geniculate nucleus, and optic tract. J Neurosci 17:2859–2868
Barnes GR, Li X, Thompson B, Singh KD, Dumoulin SO, Hess RF (2010) Decreased gray matter concentration in the lateral geniculate nuclei in human amblyopes. Invest Ophthalmol Vis Sci 51:1432–1438. doi:10.1167/iovs.09-3931
Bavelier D, Neville HJ (2002) Cross-modal plasticity: where and how? Nat Rev Neurosci 3:443–452. doi:10.1038/nrn848
Behrens TE et al (2003) Non-invasive mapping of connections between human thalamus and cortex using diffusion imaging. Nat Neurosci 6:750–757. doi:10.1038/nn1075
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc B 57:289–300
Berardi N, Pizzorusso T, Maffei L (2000) Critical periods during sensory development. Curr Opin Neurobiol 10:138–145
Bonino D et al (2008) Tactile spatial working memory activates the dorsal extrastriate cortical pathway in congenitally blind individuals. Arch Ital Biol 146:133–146
Bridge H, Cowey A, Ragge N, Watkins K (2009) Imaging studies in congenital anophthalmia reveal preservation of brain architecture in ‘visual’ cortex. Brain 132:3467–3480. doi:10.1093/brain/awp279
Burgel U, Schormann T, Schleicher A, Zilles K (1999) Mapping of histologically identified long fiber tracts in human cerebral hemispheres to the MRI volume of a reference brain: position and spatial variability of the optic radiation. NeuroImage 10:489–499. doi:10.1006/nimg.1999.0497
Burgel U, Amunts K, Hoemke L, Mohlberg H, Gilsbach JM, Zilles K (2006) White matter fiber tracts of the human brain: three-dimensional mapping at microscopic resolution, topography and intersubject variability. NeuroImage 29:1092–1105. doi:10.1016/j.neuroimage.2005.08.040
Burnett LR, Stein BE, Chaponis D, Wallace MT (2004) Superior colliculus lesions preferentially disrupt multisensory orientation. Neuroscience 124:535–547. doi:10.1016/j.neuroscience.2003.12.026
Cappe C, Morel A, Barone P, Rouiller EM (2009) The thalamocortical projection systems in primate: an anatomical support for multisensory and sensorimotor interplay. Cereb Cortex 19:2025–2037. doi:10.1093/cercor/bhn228
Cattaneo Z, Vecchi T, Cornoldi C, Mammarella I, Bonino D, Ricciardi E, Pietrini P (2008) Imagery and spatial processes in blindness and visual impairment. Neurosci Biobehav Rev 32:1346–1360. doi:10.1016/j.neubiorev.2008.05.002
Chabot N, Robert S, Tremblay R, Miceli D, Boire D, Bronchti G (2007) Audition differently activates the visual system in neonatally enucleated mice compared with anophthalmic mutants. Eur J Neurosci 26:2334–2348. doi:10.1111/j.1460-9568.2007.05854.x
Chebat DR, Chen JK, Schneider F, Ptito A, Kupers R, Ptito M (2007) Alterations in right posterior hippocampus in early blind individuals. NeuroReport 18:329–333. doi:10.1097/WNR.0b013e32802b70f8
Chen Z, Wang J, Lin F, Dai H, Mu K, Zhang H (2013) Correlation between lateral geniculate nucleus atrophy and damage to the optic disc in glaucoma. J Neuroradiol 40:281–287. doi:10.1016/j.neurad.2012.10.004
Collignon O, Dormal G, Albouy G, Vandewalle G, Voss P, Phillips C, Lepore F (2013) Impact of blindness onset on the functional organization and the connectivity of the occipital cortex. Brain 136:2769–2783. doi:10.1093/brain/awt176
Crish SD, Dengler-Crish CM, Catania KC (2006) Central visual system of the naked mole-rat (Heterocephalus glaber). Anat Record Part A Discov Mol Cell Evol Biol 288:205–212. doi:10.1002/ar.a.20288
Cullen MJ, Kaiserman-Abramof IR (1976) Cytological organization of the dorsal lateral geniculate nuclei in mutant anophthalmic and postnatally enucleated mice. J Neurocytol 5:407–424
Dai H et al (2011) Assessment of lateral geniculate nucleus atrophy with 3T MR imaging and correlation with clinical stage of glaucoma. AJNR Am J Neuroradiol 32:1347–1353. doi:10.3174/ajnr.A2486
Desgent S, Ptito M (2012) Cortical GABAergic interneurons in cross-modal plasticity following early blindness. Neural Plasticity 2012:590725. doi:10.1155/2012/590725
DuBois RM, Cohen MS (2000) Spatiotopic organization in human superior colliculus observed with fMRI. NeuroImage 12:63–70. doi:10.1006/nimg.2000.0590
Ehrsson HH (2007) The experimental induction of out-of-body experiences. Science 317:1048. doi:10.1126/science.1142175
Eickhoff SB, Stephan KE, Mohlberg H, Grefkes C, Fink GR, Amunts K, Zilles K (2005) A new SPM toolbox for combining probabilistic cytoarchitectonic maps and functional imaging data. NeuroImage 25:1325–1335. doi:10.1016/j.neuroimage.2004.12.034
Felleman DJ, Van Essen DC (1991) Distributed hierarchical processing in the primate cerebral cortex. Cereb Cortex 1:1–47
Fonov V, Evans AC, Botteron K, Almli CR, McKinstry RC, Collins DL, Brain Development Cooperative Group (2011) Unbiased average age-appropriate atlases for pediatric studies. NeuroImage 54:313–327. doi:10.1016/j.neuroimage.2010.07.033
Fortin M et al (2008) Wayfinding in the blind: larger hippocampal volume and supranormal spatial navigation. Brain 131:2995–3005. doi:10.1093/brain/awn250
Frost DO, Boire D, Gingras G, Ptito M (2000) Surgically created neural pathways mediate visual pattern discrimination. Proc Natl Acad Sci USA 97:11068–11073. doi:10.1073/pnas.190179997
Ghazanfar AA, Schroeder CE (2006) Is neocortex essentially multisensory? Trends Cogn Sci 10:278–285. doi:10.1016/j.tics.2006.04.008
Gupta N, Greenberg G, de Tilly LN, Gray B, Polemidiotis M, Yucel YH (2009) Atrophy of the lateral geniculate nucleus in human glaucoma detected by magnetic resonance imaging. Br J Ophthalmol 93:56–60. doi:10.1136/bjo.2008.138172
Headon MP, Powell TP (1973) Cellular changes in the lateral geniculate nucleus of infant monkeys after suture of the eyelids. J Anat 116:135–145
Heil P, Bronchti G, Wollberg Z, Scheich H (1991) Invasion of visual cortex by the auditory system in the naturally blind mole rat. NeuroReport 2:735–738
Hendelman WJ (2005) Atlas of functional neuroanatomy, 2nd edn. CRC Press, Boca Raton
Hernowo AT, Boucard CC, Jansonius NM, Hooymans JM, Cornelissen FW (2011) Automated morphometry of the visual pathway in primary open-angle glaucoma. Invest Ophthalmol Vis Sci 52:2758–2766. doi:10.1167/iovs.10-5682
Heumann D, Rabinowicz T (1980) Postnatal development of the dorsal lateral geniculate nucleus in the normal and enucleated albino mouse. Exp Brain Res 38:75–85
Hilbig H, Bidmon HJ, Zilles K, Busecke K (1999) Neuronal and glial structures of the superficial layers of the human superior colliculus. Anat Embryol 200:103–115
Iglesias JE, Liu CY, Thompson PM, Tu Z (2011) Robust brain extraction across datasets and comparison with publicly available methods. IEEE Trans Med Imaging 30:1617–1634. doi:10.1109/TMI.2011.2138152
Ioannides AA, Liu L, Poghosyan V, Saridis GA, Gjedde A, Ptito M, Kupers R (2013) MEG reveals a fast pathway from somatosensory cortex to occipital areas via posterior parietal cortex in a blind subject. Front Hum Neurosci 7:429. doi:10.3389/fnhum.2013.00429
Jenkinson M, Bannister P, Brady M, Smith S (2002) Improved optimization for the robust and accurate linear registration and motion correction of brain images. NeuroImage 17:825–841
Jenkinson M, Beckmann CF, Behrens TE, Woolrich MW, Smith SM (2012) Fsl. NeuroImage 62:782–790. doi:10.1016/j.neuroimage.2011.09.015
Jiang J et al (2009) Thick visual cortex in the early blind. J Neurosci 29:2205–2211. doi:10.1523/JNEUROSCI.5451-08.2009
Johansen-Berg H, Behrens TE, Sillery E, Ciccarelli O, Thompson AJ, Smith SM, Matthews PM (2005) Functional-anatomical validation and individual variation of diffusion tractography-based segmentation of the human thalamus. Cereb Cortex 15:31–39. doi:10.1093/cercor/bhh105
Kahn DM, Krubitzer L (2002) Retinofugal projections in the short-tailed opossum (Monodelphis domestica). J Comp Neurol 447:114–127. doi:10.1002/cne.10206
Kang DH, Kwon KW, Gu BM, Choi JS, Jang JH, Kwon JS (2008) Structural abnormalities of the right inferior colliculus in schizophrenia. Psychiatry Res 164:160–165. doi:10.1016/j.pscychresns.2007.12.023
Karlen SJ, Krubitzer L (2009) Effects of bilateral enucleation on the size of visual and nonvisual areas of the brain. Cereb Cortex 19:1360–1371. doi:10.1093/cercor/bhn176
Karlen SJ, Kahn DM, Krubitzer L (2006) Early blindness results in abnormal corticocortical and thalamocortical connections. Neuroscience 142:843–858. doi:10.1016/j.neuroscience.2006.06.055
Katyal S, Zughni S, Greene C, Ress D (2010) Topography of covert visual attention in human superior colliculus. J Neurophysiol 104:3074–3083. doi:10.1152/jn.00283.2010
Klinge C, Eippert F, Roder B, Buchel C (2010) Corticocortical connections mediate primary visual cortex responses to auditory stimulation in the blind. J Neurosci 30:12798–12805. doi:10.1523/JNEUROSCI.2384-10.2010
Korsholm K, Madsen KH, Frederiksen JL, Skimminge A, Lund TE (2007) Recovery from optic neuritis: an ROI-based analysis of LGN and visual cortical areas. Brain 130:1244–1253. doi:10.1093/brain/awm045
Krebs RM et al (2010) High-field FMRI reveals brain activation patterns underlying saccade execution in the human superior colliculus. PLoS ONE 5:e8691. doi:10.1371/journal.pone.0008691
Kupers R, Ptito M (2011) Insights from darkness: what the study of blindness has taught us about brain structure and function. Prog Brain Res 192:17–31. doi:10.1016/B978-0-444-53355-5.00002-6
Kupers R, Ptito M (2014) Compensatory plasticity and cross-modal reorganization following early visual deprivation. Neurosci Biobehav Rev 41:36–52. doi:10.1016/j.neubiorev.2013.08.001
Kupers R, Fumal A, de Noordhout AM, Gjedde A, Schoenen J, Ptito M (2006) Transcranial magnetic stimulation of the visual cortex induces somatotopically organized qualia in blind subjects. Proc Natl Acad Sci USA 103:13256–13260. doi:10.1073/pnas.0602925103
Kupers R, Beaulieu-Lefebvre M, Schneider FC, Kassuba T, Paulson OB, Siebner HR, Ptito M (2011) Neural correlates of olfactory processing in congenital blindness. Neuropsychologia 49:2037–2044. doi:10.1016/j.neuropsychologia.2011.03.033
Lee JY et al (2014) An investigation of lateral geniculate nucleus volume in patients with primary open-angle glaucoma using 7 tesla magnetic resonance imaging. Invest Ophthalmol Vis Sci 55:3468–3476. doi:10.1167/iovs.14-13902
Leh SE, Johansen-Berg H, Ptito A (2006) Unconscious vision: new insights into the neuronal correlate of blindsight using diffusion tractography. Brain 129:1822–1832. doi:10.1093/brain/awl111
Leh SE, Ptito A, Schonwiesner M, Chakravarty MM, Mullen KT (2010) Blindsight mediated by an S-cone-independent collicular pathway: an fMRI study in hemispherectomized subjects. J Cogn Neurosci 22:670–682. doi:10.1162/jocn.2009.21217
Leo A, Bernardi G, Handjaras G, Bonino D, Ricciardi E, Pietrini P (2012) Increased BOLD variability in the parietal cortex and enhanced parieto-occipital connectivity during tactile perception in congenitally blind individuals. Neural Plasticity 2012:720278. doi:10.1155/2012/720278
Lepore N et al (2009) Pattern of hippocampal shape and volume differences in blind subjects. NeuroImage 46:949–957. doi:10.1016/j.neuroimage.2009.01.071
Lepore N et al (2010) Brain structure changes visualized in early- and late-onset blind subjects. NeuroImage 49:134–140. doi:10.1016/j.neuroimage.2009.07.048
Li M et al (2012) Quantification of the human lateral geniculate nucleus in vivo using MR imaging based on morphometry: volume loss with age. AJNR Am J Neuroradiol 33:915–921. doi:10.3174/ajnr.A2884
Limbrick-Oldfield EH, Brooks JC, Wise RJ, Padormo F, Hajnal JV, Beckmann CF, Ungless MA (2012) Identification and characterisation of midbrain nuclei using optimised functional magnetic resonance imaging. NeuroImage 59:1230–1238. doi:10.1016/j.neuroimage.2011.08.016
Liu Y et al (2007) Whole brain functional connectivity in the early blind. Brain 130:2085–2096. doi:10.1093/brain/awm121
Lund RD, Lund JS (1971) Synaptic adjustment after deafferentation of the superior colliculus of the rat. Science 171:804–807
Masucci EF, Borts FT, Perl SM, Wener L, Schwankhaus J, Kurtzke JF (1995) MR vs CT in progressive supranuclear palsy Computerized medical imaging and graphics : the official journal of the Computerized Medical Imaging Society 19:361–368
May PJ (2006) The mammalian superior colliculus: laminar structure and connections. Prog Brain Res 151:321–378. doi:10.1016/S0079-6123(05)51011-2
Merabet LB, Pascual-Leone A (2010) Neural reorganization following sensory loss: the opportunity of change. Nat Rev Neurosci 11:44–52. doi:10.1038/nrn2758
Naidich TP, Duvernoy HM, Delman BN, Sorensen AG, Kollias SS, Haacke EM (2009) Duvernoy’s atlas of the human brain stem and cerebellum. Springer, Wien
Nichols TE, Holmes AP (2002) Nonparametric permutation tests for functional neuroimaging: a primer with examples. Hum Brain Mapp 15:1–25
Noppeney U (2007) The effects of visual deprivation on functional and structural organization of the human brain. Neurosci Biobehav Rev 31:1169–1180. doi:10.1016/j.neubiorev.2007.04.012
Noppeney U, Friston KJ, Ashburner J, Frackowiak R, Price CJ (2005) Early visual deprivation induces structural plasticity in gray and white matter. Curr Biol 15:R488–490. doi:10.1016/j.cub.2005.06.053
Pan WJ, Wu G, Li CX, Lin F, Sun J, Lei H (2007) Progressive atrophy in the optic pathway and visual cortex of early blind Chinese adults: a voxel-based morphometry magnetic resonance imaging study. NeuroImage 37:212–220. doi:10.1016/j.neuroimage.2007.05.014
Park HJ, Lee JD, Kim EY, Park B, Oh MK, Lee S, Kim JJ (2009) Morphological alterations in the congenital blind based on the analysis of cortical thickness and surface area. NeuroImage 47:98–106. doi:10.1016/j.neuroimage.2009.03.076
Patenaude B, Smith SM, Kennedy DN, Jenkinson M (2011) A Bayesian model of shape and appearance for subcortical brain segmentation. NeuroImage 56:907–922. doi:10.1016/j.neuroimage.2011.02.046
Pavani F, Spence C, Driver J (2000) Visual capture of touch: out-of-the-body experiences with rubber gloves. Psychol Sci 11:353–359
Pietrini P et al (2004) Beyond sensory images: object-based representation in the human ventral pathway. Proc Natl Acad Sci USA 101:5658–5663. doi:10.1073/pnas.0400707101
Ptito M, Desgent S (2006) Sensory Input-Based Adaptation and Brain Architecture. In: Baltes PB, Reuter-Lorenz PA, Rosler F (eds) Lifespan development and the brain. Cambridge University Press, Cambridge, pp 111–133
Ptito M, Kupers R (2005) Cross-modal plasticity in early blindness. J Integr Neurosci 4:479–488
Ptito M, Moesgaard SM, Gjedde A, Kupers R (2005) Cross-modal plasticity revealed by electrotactile stimulation of the tongue in the congenitally blind. Brain 128:606–614. doi:10.1093/brain/awh380
Ptito M, Schneider FC, Paulson OB, Kupers R (2008) Alterations of the visual pathways in congenital blindness. Exp Brain Res 187:41–49. doi:10.1007/s00221-008-1273-4
Qin W, Xuan Y, Liu Y, Jiang T, Yu C (2014) Functional connectivity density in congenitally and late blind subjects. Cereb Cortex. doi:10.1093/cercor/bhu051
Rademacher J, Burgel U, Zilles K (2002) Stereotaxic localization, intersubject variability, and interhemispheric differences of the human auditory thalamocortical system. NeuroImage 17:142–160
Rhoades RW (1980) Effects of neonatal enucleation on the functional organization of the superior colliculus in the golden hamster. J Physiol 301:383–399
Ricciardi E, Pietrini P (2011) New light from the dark: what blindness can teach us about brain function. Curr Opin Neurol 24:357–363. doi:10.1097/WCO.0b013e328348bdbf
Ricciardi E et al (2007) The effect of visual experience on the development of functional architecture in hMT+. Cereb Cortex 17:2933–2939. doi:10.1093/cercor/bhm018
Ricciardi E et al (2009) Do we really need vision? How blind people “see” the actions of others. J Neurosci 29:9719–9724. doi:10.1523/JNEUROSCI.0274-09.2009
Ricciardi E, Handjaras G, Bonino D, Vecchi T, Fadiga L, Pietrini P (2013) Beyond motor scheme: a supramodal distributed representation in the action-observation network. PLoS ONE 8:e58632. doi:10.1371/journal.pone.0058632
Ricciardi E, Bonino D, Pellegrini S, Pietrini P (2014) Mind the blind brain to understand the sighted one! Is there a supramodal cortical functional architecture? Neurosci Biobehav Rev 41:64–77. doi:10.1016/j.neubiorev.2013.10.006
Sabanciogullari V, Salk I, Balaban H, Oztoprak I, Kelkit S, Cimen M (2013) Magnetic resonance imaging mesencephalic tectum dimensions according to age and gender. Neurosciences 18:33–39
Sakakura H, Iwama K (1967) Effects of bilateral eye enucleation upon single unit activity of the lateral geniculate body in free behaving cats. Brain Res 6:667–678
Schiller PH (1977) The effect of superior colliculus ablation on saccades elicted by cortical stimulation. Brain Res 122:154–156
Schmid B, Schindelin J, Cardona A, Longair M, Heisenberg M (2010) A high-level 3D visualization API for Java and ImageJ. BMC Bioinform 11:274. doi:10.1186/1471-2105-11-274
Schneider KA, Kastner S (2009) Effects of sustained spatial attention in the human lateral geniculate nucleus and superior colliculus. J Neurosci 29:1784–1795. doi:10.1523/JNEUROSCI.4452-08.2009
Sharma J, Angelucci A, Sur M (2000) Induction of visual orientation modules in auditory cortex. Nature 404:841–847. doi:10.1038/35009043
Sherman JL, Citrin CM, Barkovich AJ, Bowen BJ (1987) MR imaging of the mesencephalic tectum: normal and pathologic variations. AJNR Am J Neuroradiol 8:59–64
Shimony JS, Burton H, Epstein AA, McLaren DG, Sun SW, Snyder AZ (2006) Diffusion tensor imaging reveals white matter reorganization in early blind humans. Cereb Cortex 16:1653–1661. doi:10.1093/cercor/bhj102
Shu N, Liu Y, Li J, Li Y, Yu C, Jiang T (2009) Altered anatomical network in early blindness revealed by diffusion tensor tractography. PLoS ONE 4:e7228. doi:10.1371/journal.pone.0007228
Smith SA, Bedi KS (1997) Unilateral eye enucleation in adult rats causes neuronal loss in the contralateral superior colliculus. J Anat 190(Pt 4):481–490
Smith SM, Nichols TE (2009) Threshold-free cluster enhancement: addressing problems of smoothing, threshold dependence and localisation in cluster inference. NeuroImage 44:83–98. doi:10.1016/j.neuroimage.2008.03.061
Smith SM et al (2004) Advances in functional and structural MR image analysis and implementation as FSL. NeuroImage 23(Suppl 1):S208–219. doi:10.1016/j.neuroimage.2004.07.051
Sparks DL, Hartwich-Young R (1989) The deep layers of the superior colliculus. Rev Oculomot Res 3:213–255
Sur M, Garraghty PE, Roe AW (1988) Experimentally induced visual projections into auditory thalamus and cortex. Science 242:1437–1441
Sylvester R, Josephs O, Driver J, Rees G (2007) Visual FMRI responses in human superior colliculus show a temporal-nasal asymmetry that is absent in lateral geniculate and visual cortex. J Neurophysiol 97:1495–1502. doi:10.1152/jn.00835.2006
Theoret H, Boire D, Herbin M, Ptito M (2001) Anatomical sparing in the superior colliculus of hemispherectomized monkeys. Brain Res 894:274–280
Tiao YC, Blakemore C (1976) Functional organization in the superior colliculus of the golden hamster. J Comp Neurol 168:483–503. doi:10.1002/cne.901680404
Tomaiuolo F et al (2014) Morphometric changes of the corpus callosum in congenital blindness. PLoS ONE 9:e107871. doi:10.1371/journal.pone.0107871
Trampel R, Ott DV, Turner R (2011) Do the congenitally blind have a stria of Gennari? First intracortical insights in vivo. Cereb Cortex 21:2075–2081. doi:10.1093/cercor/bhq282
Van Essen DC, Drury HA, Dickson J, Harwell J, Hanlon D, Anderson CH (2001) An integrated software suite for surface-based analyses of cerebral cortex. J Am Med Inf Assoc JAMIA 8:443–459
von Melchner L, Pallas SL, Sur M (2000) Visual behaviour mediated by retinal projections directed to the auditory pathway. Nature 404:871–876. doi:10.1038/35009102
Wallace MT, Stein BE (1994) Cross-modal synthesis in the midbrain depends on input from cortex. J Neurophysiol 71:429–432
Wallace MT, Wilkinson LK, Stein BE (1996) Representation and integration of multiple sensory inputs in primate superior colliculus. J Neurophysiol 76:1246–1266
Wang D, Qin W, Liu Y, Zhang Y, Jiang T, Yu C (2014) Altered resting-state network connectivity in congenital blind. Hum Brain Mapp 35:2573–2581. doi:10.1002/hbm.22350
Warmuth-Metz M, Naumann M, Csoti I, Solymosi L (2001) Measurement of the midbrain diameter on routine magnetic resonance imaging: a simple and accurate method of differentiating between Parkinson disease and progressive supranuclear palsy. Arch Neurol 58:1076–1079
White BJ, Munoz DP (2011) The superior colliculus. In: Liversedge S, Gilchrist I, Everling S (eds) Oxford handbook of eye movements, 1st edn. Oxford University Press, New York, pp 195–213
Winer JA (1984) The human medial geniculate body. Hear Res 15:225–247
Winkler AM et al (2012) Measuring and comparing brain cortical surface area and other areal quantities. NeuroImage 61:1428–1443. doi:10.1016/j.neuroimage.2012.03.026
Yu C, Shu N, Li J, Qin W, Jiang T, Li K (2007) Plasticity of the corticospinal tract in early blindness revealed by quantitative analysis of fractional anisotropy based on diffusion tensor tractography. NeuroImage 36:411–417. doi:10.1016/j.neuroimage.2007.03.003
Zhang Y, Brady M, Smith S (2001) Segmentation of brain MR images through a hidden Markov random field model and the expectation-maximization algorithm. IEEE Trans Med Imaging 20:45–57. doi:10.1109/42.906424
Zhang D, Snyder AZ, Fox MD, Sansbury MW, Shimony JS, Raichle ME (2008) Intrinsic functional relations between human cerebral cortex and thalamus. J Neurophysiol 100:1740–1748. doi:10.1152/jn.90463.2008
Zvorykin VP (1980) New data on individual quantitative features of the human lateral geniculate body. Arkhiv anatomii gistologii i embriologii 78:24–27
Acknowledgments
LC would like to thank Dr. Francesco Tomaiuolo and Dr. Ludwig Barbaro for the helpful comments and suggestions provided. This work was supported by grants from the Italian Ministero dell’Istruzione, dell’Università e della Ricerca (PP, ER, LC, GH), the Lundbeck foundation (RK), the Danish Medical Research Council (MP), and the Harland Sanders Chair in Visual Science (Canada). The authors note professional manuscript proofreading by Inglewood Biomedical Editing.
Conflict of interest
None of the authors have any potential conflict of interest related to this manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cecchetti, L., Ricciardi, E., Handjaras, G. et al. Congenital blindness affects diencephalic but not mesencephalic structures in the human brain. Brain Struct Funct 221, 1465–1480 (2016). https://doi.org/10.1007/s00429-014-0984-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-014-0984-5