Abstract
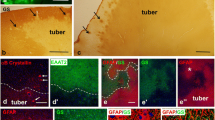
Tubers are cerebral cortical developmental malformations associated with epilepsy and autism in tuberous sclerosis complex (TSC). The disparity between tuber number and severity of neurological impairment often observed in TSC led us to hypothesize that microscopic structural abnormalities distinct from tubers may occur in TSC. Serial frontal to occipital lobe sections were prepared from five postmortem TSC brain specimens. Sections were probed with cresyl violet stain or NeuN antibodies to define cytoarchitectural abnormalities and phospho-S6 (Ser235/236) antibodies to define mammalian target of rapamycin complex 1 (mTORC1) pathway activation. Tubers identified in all specimens (mean, 5 tubers per brain specimen) were defined by abnormal cortical lamination, dysmorphic neurons, and giant cells (GCs) and exhibited robust phospho-S6 immunolabeling. Histopathological analysis of non-tuber cortices demonstrated that 32% of the sections exhibited microscopic cytoarchitectural alterations, whereas 68% of the sections did not. Four types of morphological abnormalities were defined including: (1) focal dyslamination, (2) heterotopic neurons, (3) small collections of giant cells (GCs) and neurons we termed “microtubers”, (4) isolated GCs we termed “sentinel” cells. When compared with control cortex, phospho-S6 labeling was enhanced in microtubers and sentinel cells and in some but not all areas of dyslamination. There are microscopic cytoarchitectural abnormalities identified in postmortem TSC brain specimens that are distinct from tubers. mTORC1 cascade activation in these areas supports a widespread effect of TSC1 or TSC2 mutations on brain development. Tubers may represent the most dramatic developmental abnormality in TSC; however, more regionally pervasive yet subtle abnormalities may contribute to neurological disability in TSC.







Similar content being viewed by others
References
Arulrajah S, Ertan G, Jordan L et al (2009) Magnetic resonance imaging and diffusion-weighted imaging of normal-appearing white matter in children and young adults with tuberous sclerosis complex. Neuroradiology 51:781–786
Baybis M, Yu J, Lee A et al (2004) mTOR cascade activation distinguishes tubers from focal cortical dysplasia. Ann Neurol 56:478–487
Boer K, Troost D, Jansen F et al (2008) Clinicopathological and immunohistochemical findings in an autopsy case of tuberous sclerosis complex. Neuropathology 28:577–590
Bollo RJ, Kalhorn SP, Carlson C, Haegeli V, Devinsky O, Weiner HL (2008) Epilepsy surgery and tuberous sclerosis complex: special considerations. Neurosurg Focus 25:E13
Chan JA, Zhang H, Roberts PS et al (2004) Pathogenesis of tuberous sclerosis subependymal giant cell astrocytomas: biallelic inactivation of TSC1 or TSC2 leads to mTOR activation. J Neuropathol Exp Neurol 63:1236–1242
Chou IJ, Lin KL, Wong AM et al (2008) Neuroimaging correlation with neurological severity in tuberous sclerosis complex. Eur J Paediatr Neurol 12:108–112
Crino PB, Nathanson KL, Henske EP (2006) The tuberous sclerosis complex. N Engl J Med 355:1345–1356
Doherty C, Goh S, Young Poussaint T, Erdag N, Thiele EA (2005) Prognostic significance of tuber count and location in tuberous sclerosis complex. J Child Neurol 20:837–841
Eberhart CG, Copeland J, Abel TW (2006) Brief report: S6 ribosomal protein phosphorylation in autistic frontal cortex and cerebellum: a tissue array analysis. J Autism Dev Disord 36:1131–1135
Ehninger D, de Vries PJ, Silva AJ (2009) From mTOR to cognition: molecular and cellular mechanisms of cognitive impairments in tuberous sclerosis. J Intellect Disabil Res 53:838–851
Fukutani Y, Yasuda M, Saitoh C et al (1992) An autopsy case of tuberous sclerosis. Histological and immunohistochemical study. Histol Histopathol 7:709–714
Gallagher A, Grant EP, Madan N, Jarrett DY, Lyczkowski DA, Thiele EA (2010) MRI findings reveal three different types of tubers in patients with tuberous sclerosis complex. J Neurol 257:1373–1381
Major P, Rakowski S, Simon MV (2009) Are cortical tubers epileptogenic? Evidence from electrocorticography. Epilepsia 50:147–154
Miyata H, Chiang AC, Vinters HV (2004) Insulin signaling pathways in cortical dysplasia and TSC-tubers: tissue microarray analysis. Ann Neurol 56:510–519
Mizuguchi M, Takashima S (2001) Neuropathology of tuberous sclerosis. Brain Dev 23(7):508–515
Naramoto A, Itoh N, Nakano M, Shigematsu H (1989) An autopsy case of tuberous sclerosis associated with primary pericardial mesothelioma. Acta Pathol Jpn 39:400–406
Nixon JR, Miller GM, Okazaki H, Gomez MR (1989) Cerebral tuberous sclerosis: postmortem magnetic resonance imaging and pathologic anatomy. Mayo Clin Proc 64:305–311
Pou-Serradell A, Pascual J, Ugarte A, Florensa R, Soler L (1979) A study of 14 cases of Bourneville’s tuberous sclerosis, including two pathological reports and seven cases investigated by computer tomography (author’s transl). Rev Neurol (Paris) 135:693–703
Ridler K, Bullmore ET, De Vries PJ (2001) Widespread anatomical abnormalities of grey and white matter structure in tuberous sclerosis. Psychol Med 31:1437–1446
Ridler K, Suckling J, Higgins NJ (2007) Neuroanatomical correlates of memory deficits in tuberous sclerosis complex. Cereb Cortex 17:261–271
Raznahan A, Higgins NP, Griffiths PD, Humphrey A, Yates JR, Bolton PF (2007) Biological markers of intellectual disability in tuberous sclerosis. Psychol Med 37:1293–1304
Scheithauer BW, Reagan TJ (1999) Neuropathology. In: Gomez MR (ed) Tuberous sclerosis complex, 3rd edn. Oxford University Press, Oxford, pp 101–144
Talos DM, Kwiatrowski DJ, Cordero K, Black PM, Jensen FE (2008) Cell-specific alterations of glutamate receptor expression in tuberous sclerosis complex cortical tubers. Ann Neurol 63:454–465
Wong V, Khong PL (2006) Tuberous sclerosis complex: correlation of magnetic resonance imaging (MRI) findings with comorbidities. J Child Neurol 21:99–105
Zaroff CM, Bar WB, Carlson C et al (2006) Mental retardation and relation to seizure and tuber burden in tuberous sclerosis complex. Seizure 15:558–562
Acknowledgments
This work was supported by NS045877, NS045021, and Department of Defense CDMRP-TSC Program (PBC); the National Epilepsy Fund-“Power of the Small,” Hersenstichting Nederland (NEF 02-10 and NEF 05-11) and Stichting Michelle (M06.011; EA). We humbly thank TSC patients and families who sponsored brain donation for research and the Brain and Tissue Bank of the University of Maryland.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marcotte, L., Aronica, E., Baybis, M. et al. Cytoarchitectural alterations are widespread in cerebral cortex in tuberous sclerosis complex. Acta Neuropathol 123, 685–693 (2012). https://doi.org/10.1007/s00401-012-0950-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-012-0950-3