Abstract
Purpose
To evaluate the detection rate among three different targeted biopsy approaches of robot-assisted MRI/TRUS fusion (RA-TB), mpMRI in-bore (MRGB), cognitive fusion guidance biopsy (COG-TB) for the detection of prostate cancer (PC) and clinically significant PC (csPC).
Methods
Between 2014 and 2016, 156 patients with a lesion on mpMRI, performed in accordance with ESUR guidelines, due to cancer suspicion or on-going cancer suspicion after prior negative prostate biopsy, underwent targeted biopsy with RA-TB, MRGB or COG-TB. All lesions were rated according to PI-RADS v2. We compared detection rates between techniques. Models were constructed to predict the detection of overall PC and csPC and using a 1000 boot-strap sample.
Results
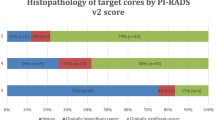
In the all cohort, 73, 45 and 38 patients underwent RA-TB, MRGB or COG-TB, respectively. Overall PC was found in 39 (52.42%), 23 (51.11%) and 11 (28.95%) (p = 0.04) patients of RA-TB, MRGB and COG-TB arm, respectively. As concerning the detection of csPC, it was found in 26 (35.62%),18 (40.0%) and 9 (23.68%) patients of RA-TB, MRGB and COG-TB arm (p = 0.27). Model 1 showed that RA-TB [OR: 10.08 (95% CI 1.95–51.97); p < 0.01] and MRGB [OR: 12.88 (95% CI 2.36–70.25); p < 0.01] were associated with overall PC detection in TB, while only MRGB was associated with csPC at TB (model 2) [OR: 5.72; (95% CI 1.40–23.35); p < 0.01]. The c-index for model 1 and model 2 was 0.86 and 0.85, respectively. We did not report significant complications between groups.
Conclusion
In-bore biopsy and MRI/TRUS fusion-guided biopsy showed greater accuracy in detecting PC compared to cognitive fusion as modeled in a newly established normogram.


Similar content being viewed by others
References
Dy GW, Gore JL, Forouzanfar MH, Naghavi M, Fitzmaurice C (2017) Global burden of urologic cancers, 1990–2013. Eur Urol 71(3):437–446. https://doi.org/10.1016/j.eururo.2016.10.008
Mottet N, Bellmunt J, Bolla M, Briers E, Cumberbatch MG, De Santis M, Fossati N, Gross T, Henry AM, Joniau S, Lam TB, Mason MD, Matveev VB, Moldovan PC, van den Bergh RCN, van den Broeck T, van den Poel HG, van den Kwast TH, Rouviere O, Schoots IG, Wiegel T, Cornford P (2017) EAU-ESTRO-SIOG guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol 71(4):618–629. https://doi.org/10.1016/j.eururo.2016.08.003
Moldovan PC, Van den Broeck T, Sylvester R, Marconi L, Bellmunt J, van den Bergh RCN, Bolla M, Briers E, Cumberbatch MG, Fossati N, Gross T, Henry AM, Joniau S, van der Kwast TH, Matveev VB, van der Poel HG, De Santis M, Schoots IG, Wiegel T, Yuan CY, Cornford P, Mottet N, Lam TB, Rouviere O (2017) What is the negative predictive value of multiparametric magnetic resonance imaging in excluding prostate cancer at biopsy? A systematic review and meta-analysis from the european association of urology prostate cancer guidelines panel. Eur Urol 72(2):250–266. https://doi.org/10.1016/j.eururo.2017.02.026
Futterer JJ, Briganti A, De Visschere P, Emberton M, Giannarini G, Kirkham A, Taneja SS, Thoeny H, Villeirs G, Villers A (2015) Can clinically significant prostate cancer be detected with multiparametric magnetic resonance imaging? A systematic review of the literature. Eur Urol 68(6):1045–1053. https://doi.org/10.1016/j.eururo.2015.01.013
Schiavina R, Vagnoni V, D’Agostino D, Borghesi M, Salvaggio A, Giampaoli M, Pultrone CV, Saraceni G, Gaudiano C, Vigo M, Bianchi L, Dababneh H, La Manna G, Chessa F, Romagnoli D, Martorana G, Brunocilla E, Porreca A (2017) “In-bore” MRI-guided prostate biopsy using an endorectal nonmagnetic device: a prospective study of 70 consecutive patients. Clin Genitourin Cancer 15(3):417–427. https://doi.org/10.1016/j.clgc.2017.01.013
Mischinger J, Kaufmann S, Russo GI, Harland N, Rausch S, Amend B, Scharpf M, Loewe L, Todenhoefer T, Notohamiprodjo M, Nikolaou K, Stenzl A, Bedke J, Kruck S (2017) Targeted versus systematic robot-assisted transperineal MRI-TRUS fusion prostate biopsy. BJU Int. https://doi.org/10.1111/bju.14089
Filson CP, Natarajan S, Margolis DJ, Huang J, Lieu P, Dorey FJ, Reiter RE, Marks LS (2016) Prostate cancer detection with magnetic resonance-ultrasound fusion biopsy: the role of systematic and targeted biopsies. Cancer 122(6):884–892. https://doi.org/10.1002/cncr.29874
Kaufmann S, Mischinger J, Amend B, Rausch S, Adam M, Scharpf M, Fend F, Kramer U, Notohamiprodjo M, Nikolaou K, Stenzl A, Bedke J, Kruck S (2017) First report of robot-assisted transperineal fusion versus off-target biopsy in patients undergoing repeat prostate biopsy. World J Urol 35(7):1023–1029. https://doi.org/10.1007/s00345-016-1970-8
Sonn GA, Margolis DJ, Marks LS (2014) Target detection: magnetic resonance imaging-ultrasound fusion-guided prostate biopsy. Urol Oncol 32(6):903–911. https://doi.org/10.1016/j.urolonc.2013.08.006
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213
Barentsz JO, Richenberg J, Clements R, Choyke P, Verma S, Villeirs G, Rouviere O, Logager VV, Futterer JJ, European Society of Urogenital R (2012) ESUR prostate MR guidelines 2012. Eur Radiol 22(4):746–757. https://doi.org/10.1007/s00330-011-2377-y
Weinreb JC, Barentsz JO, Choyke PL, Cornud F, Haider MA, Macura KJ, Margolis D, Schnall MD, Shtern F, Tempany CM, Thoeny HC, Verma S (2016) PI-RADS prostate imaging-reporting and data system: 2015, version 2. Eur Urol 69(1):16–40. https://doi.org/10.1016/j.eururo.2015.08.052
Moore CM, Kasivisvanathan V, Eggener S, Emberton M, Futterer JJ, Gill IS, Grubb Iii RL, Hadaschik B, Klotz L, Margolis DJ, Marks LS, Melamed J, Oto A, Palmer SL, Pinto P, Puech P, Punwani S, Rosenkrantz AB, Schoots IG, Simon R, Taneja SS, Turkbey B, Ukimura O, van der Meulen J, Villers A, Watanabe Y, Consortium S (2013) Standards of reporting for MRI-targeted biopsy studies (START) of the prostate: recommendations from an International Working Group. Eur Urol 64(4):544–552. https://doi.org/10.1016/j.eururo.2013.03.030
Puech P, Rouviere O, Renard-Penna R, Villers A, Devos P, Colombel M, Bitker MO, Leroy X, Mege-Lechevallier F, Comperat E, Ouzzane A, Lemaitre L (2013) Prostate cancer diagnosis: multiparametric MR-targeted biopsy with cognitive and transrectal US-MR fusion guidance versus systematic biopsy–prospective multicenter study. Radiology 268(2):461–469. https://doi.org/10.1148/radiol.13121501
Labanaris AP, Engelhard K, Zugor V, Nutzel R, Kuhn R (2010) Prostate cancer detection using an extended prostate biopsy schema in combination with additional targeted cores from suspicious images in conventional and functional endorectal magnetic resonance imaging of the prostate. Prostate Cancer Prostatic Dis 13(1):65–70. https://doi.org/10.1038/pcan.2009.41
Kaufmann S, Kruck S, Kramer U, Gatidis S, Stenzl A, Roethke M, Scharpf M, Schilling D (2015) Direct comparison of targeted MRI-guided biopsy with systematic transrectal ultrasound-guided biopsy in patients with previous negative prostate biopsies. Urol Int 94(3):319–325. https://doi.org/10.1159/000365397
Kuru TH, Herden J, Zugor V, Akbarov I, Pfister D, Porres D, Heidenreich A (2016) How to perform image-guided prostate biopsy: in-bore and fusion approaches. Eur Urol Focus 2(2):151–153. https://doi.org/10.1016/j.euf.2016.03.016
Roethke M, Anastasiadis AG, Lichy M, Werner M, Wagner P, Kruck S, Claussen CD, Stenzl A, Schlemmer HP, Schilling D (2012) MRI-guided prostate biopsy detects clinically significant cancer: analysis of a cohort of 100 patients after previous negative TRUS biopsy. World J Urol 30(2):213–218. https://doi.org/10.1007/s00345-011-0675-2
Venderink W, van der Leest M, van Luijtelaar A, van de Ven WJM, Futterer JJ, Sedelaar JPM, Huisman HJ (2017) Retrospective comparison of direct in-bore magnetic resonance imaging (MRI)-guided biopsy and fusion-guided biopsy in patients with MRI lesions which are likely or highly likely to be clinically significant prostate cancer. World J Urol. https://doi.org/10.1007/s00345-017-2085-6
Kryvenko ON, Carter HB, Trock BJ, Epstein JI (2014) Biopsy criteria for determining appropriateness for active surveillance in the modern era. Urology 83(4):869–874. https://doi.org/10.1016/j.urology.2013.12.054
Vickers AJ, Elkin EB (2006) Decision curve analysis: a novel method for evaluating prediction models. Med Decis Making 26(6):565–574. https://doi.org/10.1177/0272989X06295361
Lemeshow S, Hosmer DW Jr (1982) A review of goodness of fit statistics for use in the development of logistic regression models. Am J Epidemiol 115(1):92–106
Schoots IG, Roobol MJ, Nieboer D, Bangma CH, Steyerberg EW, Hunink MG (2015) Magnetic resonance imaging-targeted biopsy may enhance the diagnostic accuracy of significant prostate cancer detection compared to standard transrectal ultrasound-guided biopsy: a systematic review and meta-analysis. Eur Urol 68(3):438–450. https://doi.org/10.1016/j.eururo.2014.11.037
Kroenig M, Schaal K, Benndorf M, Soschynski M, Lenz P, Krauss T, Drendel V, Kayser G, Kurz P, Werner M, Wetterauer U, Schultze-Seemann W, Langer M, Jilg CA (2016) Diagnostic Accuracy of robot-guided, software based transperineal MRI/TRUS fusion biopsy of the prostate in a high risk population of previously biopsy negative men. Biomed Res Int 2016:2384894. https://doi.org/10.1155/2016/2384894
Kaye DR, Stoianovici D, Han M (2014) Robotic ultrasound and needle guidance for prostate cancer management: review of the contemporary literature. Curr Opin Urol 24(1):75–80. https://doi.org/10.1097/MOU.0000000000000011
Wegelin O, van Melick HHE, Hooft L, Bosch J, Reitsma HB, Barentsz JO, Somford DM (2017) Comparing three different techniques for magnetic resonance imaging-targeted prostate biopsies: a systematic review of in-bore versus magnetic resonance imaging-transrectal ultrasound fusion versus cognitive registration. is there a preferred technique? Eur Urol 71(4):517–531. https://doi.org/10.1016/j.eururo.2016.07.041
Cantiello F, Russo GI, Cicione A, Ferro M, Cimino S, Favilla V, Perdona S, De Cobelli O, Magno C, Morgia G, Damiano R (2016) PHI and PCA3 improve the prognostic performance of PRIAS and epstein criteria in predicting insignificant prostate cancer in men eligible for active surveillance. World J Urol 34(4):485–493. https://doi.org/10.1007/s00345-015-1643-z
Cantiello F, Russo GI, Ferro M, Cicione A, Cimino S, Favilla V, Perdona S, Bottero D, Terracciano D, De Cobelli O, Morgia G, Damiano R (2015) Prognostic accuracy of prostate health index and urinary prostate cancer antigen 3 in predicting pathologic features after radical prostatectomy. Urol Oncol 33(4):163. https://doi.org/10.1016/j.urolonc.2014.12.002 (e115-123)
Russo GI, Regis F, Castelli T, Favilla V, Privitera S, Giardina R, Cimino S, Morgia G (2017) A systematic review and meta-analysis of the diagnostic accuracy of prostate health index and 4-kallikrein panel score in predicting overall and high-grade prostate cancer. Clin Genitourin Cancer 15(4):429–439. https://doi.org/10.1016/j.clgc.2016.12.022 (e421)
Acknowledgements
We thank Johannes Mischinger for helpful discussion of the manuscript.
Funding
No funding was received for this work. Stephan Kruck received travel Grant from Biobot Surgical Company.
Author information
Authors and Affiliations
Contributions
SK protocol/project development, data collection, manuscript writing. GIR manuscript writing, data analysis. FB protocol/project development. LL protocol/project development. GM protocol/project development. KN protocol/project development. AS protocol/project development. SK protocol/project development, data collection, manuscript writing. JB protocol/project development, manuscript writing.
Corresponding authors
Ethics declarations
Conflict of interest
Authors disclose no conflict of interest.
Research involving human participants and/or animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The local ethic committee approved the study (397/2012R).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kaufmann, S., Russo, G.I., Bamberg, F. et al. Prostate cancer detection in patients with prior negative biopsy undergoing cognitive-, robotic- or in-bore MRI target biopsy. World J Urol 36, 761–768 (2018). https://doi.org/10.1007/s00345-018-2189-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-018-2189-7