Abstract
Purpose
We performed a retrospective study to evaluate the efficacy of cetuximab plus chemotherapy in metastatic gastric cancer (MGC) patients previously treated with chemotherapy and to investigate potential predictors of treatment efficacy in those patients.
Methods
Thirty-two patients with MGC were included in this study. Cetuximab was delivered, often combined with irinotecan-based chemotherapy. Thirty patients were analyzed for K-ras mutations via direct sequencing of the tumor DNA.
Results
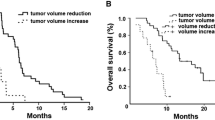
Patients were heavily pretreated with a median number of three previous lines of palliative chemotherapy (56% of the patients were refractory to all of the following drugs: fluoropyrimidines, cisplatin, irinotecan, oxaliplatin, and docetaxel) and 53% of the patients displayed poor performance status. Of 28 response-assessable patients, the overall response rate to cetuximab plus chemotherapy was 3.6% [95% confidence interval (CI) 0–10.5%] and the disease control rate was 28.6%. The median progression-free survival (PFS) was 1.7 months (95% CI 1.3–2.1 months), and the median overall survival (OS) was 3.2 months (95% CI 1.4–5.0 months). Multivariate analyses revealed that skin rash and performance status were significantly associated with PFS and OS. The presence of a K-ras mutation (13.3%) was not associated with either PFS or OS.
Conclusion
Our study suggests that MGC patients with good performance status and skin rash benefit most from salvage cetuximab combined with chemotherapy, even in heavily pretreated status.



Similar content being viewed by others
References
Parkin DM, Bray F, Ferlay J, Pisani P (2005) Global cancer statistics, 2002. CA Cancer J Clin 55:74–108
Glimelius B, Ekstrom K, Hoffman K et al (1997) Randomized comparison between chemotherapy plus best supportive care with best supportive care in advanced gastric cancer. Ann Oncol 8:163–168
Pyrhonen S, Kuitunen T, Nyandoto P, Kouri M (1995) Randomised comparison of fluorouracil, epidoxorubicin and methotrexate (FEMTX) plus supportive care with supportive care alone in patients with non-resectable gastric cancer. Br J Cancer 71:587–591
Wagner AD, Grothe W, Haerting J, Kleber G, Grothey A, Fleig WE (2006) Chemotherapy in advanced gastric cancer: a systematic review and meta-analysis based on aggregate data. J Clin Oncol 24:2903–2909
Dank M, Zaluski J, Barone C et al (2008) Randomized phase III study comparing irinotecan combined with 5-fluorouracil and folinic acid to cisplatin combined with 5-fluorouracil in chemotherapy naive patients with advanced adenocarcinoma of the stomach or esophagogastric junction. Ann Oncol 19:1450–1457
Al-Batran SE, Hartmann JT, Probst S et al (2008) Phase III trial in metastatic gastroesophageal adenocarcinoma with fluorouracil, leucovorin plus either oxaliplatin or cisplatin: a study of the Arbeitsgemeinschaft Internistische Onkologie. J Clin Oncol 26:1435–1442
Kang Y, Kang WK, Shin DB et al (2006) Randomized phase III trial of capecitabine/cisplatin (XP) vs. continuous infusion of 5-FU/cisplatin (FP) as first-line therapy in patients (pts) with advanced gastric cancer (AGC): efficacy and safety results. Proc Am Soc Clin Oncol 24:183s (abstr LBA4018)
Van Cutsem E, Moiseyenko VM, Tjulandin S et al (2006) Phase III study of docetaxel and cisplatin plus fluorouracil compared with cisplatin and fluorouracil as first-line therapy for advanced gastric cancer: a report of the V325 Study Group. J Clin Oncol 24:4991–4997
Cunningham D, Starling N, Rao S et al (2008) Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med 358:36–46
Takehana T, Kunitomo K, Suzuki S et al (2003) Expression of epidermal growth factor receptor in gastric carcinomas. Clin Gastroenterol Hepatol 1:438–445
Tokunaga A, Onda M, Okuda T et al (1995) Clinical significance of epidermal growth factor (EGF), EGF receptor, and c-erbB-2 in human gastric cancer. Cancer 75:1418–1425
Kim MA, Lee HS, Lee HE, Jeon YK, Yang HK, Kim WH (2008) EGFR in gastric carcinomas: prognostic significance of protein overexpression and high gene copy number. Histopathology 52:738–746
Matsubara J, Yamada Y, Hirashima Y et al (2008) Impact of insulin-like growth factor type 1 receptor, epidermal growth factor receptor, and HER2 expressions on outcomes of patients with gastric cancer. Clin Cancer Res 14:3022–3029
Gamboa-Dominguez A, Dominguez-Fonseca C, Quintanilla-Martinez L et al (2004) Epidermal growth factor receptor expression correlates with poor survival in gastric adenocarcinoma from Mexican patients: a multivariate analysis using a standardized immunohistochemical detection system. Mod Pathol 17:579–587
Kim JS, Kim MA, Kim TM et al (2009) Biomarker analysis in stage III-IV (M0) gastric cancer patients who received curative surgery followed by adjuvant 5-fluorouracil and cisplatin chemotherapy: epidermal growth factor receptor (EGFR) associated with favourable survival. Br J Cancer 100:732–738
Hirsch FR, Varella-Garcia M, Bunn PA Jr et al (2003) Epidermal growth factor receptor in non-small-cell lung carcinomas: correlation between gene copy number and protein expression and impact on prognosis. J Clin Oncol 21:3798–3807
Mahtani RL, Macdonald JS (2008) Synergy between cetuximab and chemotherapy in tumors of the gastrointestinal tract. Oncologist 13:39–50
Vincenzi B, Schiavon G, Silletta M, Santini D, Tonini G (2008) The biological properties of cetuximab. Crit Rev Oncol Hematol 68:93–106
Cunningham D, Humblet Y, Siena S et al (2004) Cetuximab monotherapy and cetuximab plus irinotecan in irinotecan-refractory metastatic colorectal cancer. N Engl J Med 351:337–345
Saltz LB, Meropol NJ, Loehrer PJ Sr, Needle MN, Kopit J, Mayer RJ (2004) Phase II trial of cetuximab in patients with refractory colorectal cancer that expresses the epidermal growth factor receptor. J Clin Oncol 22:1201–1208
Lievre A, Bachet JB, Boige V et al (2008) KRAS mutations as an independent prognostic factor in patients with advanced colorectal cancer treated with cetuximab. J Clin Oncol 26:374–379
Lievre A, Bachet JB, Le Corre D et al (2006) KRAS mutation status is predictive of response to cetuximab therapy in colorectal cancer. Cancer Res 66:3992–3995
Van Cutsem E, Kohne CH, Hitre E et al (2009) Cetuximab and chemotherapy as initial treatment for metastatic colorectal cancer. N Engl J Med 360:1408–1417
Bokemeyer C, Bondarenko I, Makhson A et al (2009) Fluorouracil, leucovorin, and oxaliplatin with and without cetuximab in the first-line treatment of metastatic colorectal cancer. J Clin Oncol 27:663–671
Pinto C, Di Fabio F, Siena S et al (2007) Phase II study of cetuximab in combination with FOLFIRI in patients with untreated advanced gastric or gastroesophageal junction adenocarcinoma (FOLCETUX study). Ann Oncol 18:510–517
Lordick F, Lorenzen S, Hegewisch-Becker S et al (2007) Cetuximab plus weekly oxaliplatin/5FU/FA (FUFOX) in 1st line metastatic gastric cancer, Final results from a multicenter phase II study of the AIO upper GI study group. Proc Am Soc Clin Oncol 25:204s (abstr 4526)
Zhang X, Xu J, Shen L et al (2009) A phase II study of cetuximab with cisplatin and capecitabine as first-line treatment in advanced gastric cancer. In: Proceedings of American society of clinical oncology 2009 gastrointestinal cancers symposium, 15–17 Jan 2009 (abstr LBA39)
Kim C, Lee J, Ryu M et al (2009) A prospective phase II study of cetuximab in combination with capecitabine and oxaliplatin (XELOX) in patients with metastatic and/or recurrent advanced gastric cancer. In: Proceedings of American society of clinical oncology 2009 gastrointestinal cancers symposium, 15–17 Jan 2009 (abstr 89)
Yeh K, Hsu C, Hsu C et al (2009) Phase II study of cetuximab plus weekly cisplatin and 24-hour infusion of high-dose 5-fluorouracil and leucovorin for the first-line treatment of advanced gastric cancer. In: Proceedings of American society of clinical oncology 2009 gastrointestinal cancers symposium, 15–17 Jan 2009 (abstr 60)
Woell E, Greil R, Eisterer W et al (2008) Oxaliplatin, irinotecan and cetuximab in advanced gastric cancer: first results of a multicenter phase II trial (AGMT Gastric-2). Proc Am Soc Clin Oncol 26:662s (abstr 15587)
AIO Gastric group, Germany, Moehler MH, Trarbach T et al (2008) Cetuximab with irinotecan/Na-Fa/5-FU as first-line treatment in advanced gastric cancer: preliminary results of a nonrandomised multi-centre AIO phase II study. In: Proceedings of American society of clinical oncology 2008 gastrointestinal cancers symposium, 25–27 Jan 2008 (abstract 102)
Han SW, Oh DY, Im SA et al (2009) Phase II study and biomarker analysis of cetuximab combined with modified FOLFOX6 in advanced gastric cancer. Br J Cancer 100:298–304
Therasse P, Arbuck SG, Eisenhauer EA et al (2000) New guidelines to evaluate the response to treatment in solid tumors, European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216
Frattini M, Balestra D, Suardi S et al (2004) Different genetic features associated with colon and rectal carcinogenesis. Clin Cancer Res 10:4015–4021
Tebbutt NC, Sourjina T, Strickland AH et al (2008) ATTAX2: docetaxel plus cetuximab as second-line treatment for docetaxel-refractory oesophago-gastric cancer—final results of a multicentre phase II trial by the AGITG. Proc Am Soc Clin Oncol 26:659s (abstr 15554)
Burtness B, Goldwasser MA, Flood W, Mattar B, Forastiere AA (2005) Phase III randomized trial of cisplatin plus placebo compared with cisplatin plus cetuximab in metastatic/recurrent head and neck cancer: an Eastern Cooperative Oncology Group study. J Clin Oncol 23:8646–8654
Xiong HQ, Rosenberg A, LoBuglio A et al (2004) Cetuximab, a monoclonal antibody targeting the epidermal growth factor receptor, in combination with gemcitabine for advanced pancreatic cancer: a multicenter phase II trial. J Clin Oncol 22:2610–2616
Perez-Soler R, Saltz L (2005) Cutaneous adverse effects with HER1/EGFR-targeted agents: is there a silver lining? J Clin Oncol 23:5235–5246
Lee SH, Lee JW, Soung YH et al (2003) BRAF and KRAS mutations in stomach cancer. Oncogene 22:6942–6945
Kim IJ, Park JH, Kang HC et al (2003) Mutational analysis of BRAF and K-ras in gastric cancers: absence of BRAF mutations in gastric cancers. Hum Genet 114:118–120
Russo A, Bazan V, Migliavacca M et al (2001) DNA aneuploidy and high proliferative activity but not K-ras-2 mutations as independent predictors of clinical outcome in operable gastric carcinoma: results of a 5-year Gruppo Oncologico dell’Italia Meridonale (GDIM) prospective study. Cancer 92:294–302
Yoo J, Park SY, Robinson RA, Kang SJ, Ahn WS, Kang CS (2002) ras Gene mutations and expression of Ras signal transduction mediators in gastric adenocarcinomas. Arch Pathol Lab Med 126:1096–1100
Hongyo T, Buzard GS, Palli D et al (1995) Mutations of the K-ras and p53 genes in gastric adenocarcinomas from a high-incidence region around Florence, Italy. Cancer Res 55:2665–2672
Andreyev HJ, Norman AR, Cunningham D, Oates JR, Clarke PA (1998) Kirsten ras mutations in patients with colorectal cancer: the multicenter “RASCAL” study. J Natl Cancer Inst 90:675–684
Stella G, Rojas Llimpe FL, Barone C et al (2009) KRAS and BRAF mutational status and response to cetuximab combination therapy in advanced gastric cancer (GC) patients. In: Proceedings of American society of clinical oncology 2009 gastrointestinal cancers symposium, 15–17 Jan 2009 (abstr 34)
Acknowledgments
This study was supported by grant 0610520 from the Research Institute and Hospital, National Cancer Center, Goyang, Gyeonggi, Republic of Korea.
Conflict of interest statement
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
S. R. Park and M.-C. Kook contributed equally to this work.
Rights and permissions
About this article
Cite this article
Park, S.R., Kook, MC., Choi, I.J. et al. Predictive factors for the efficacy of cetuximab plus chemotherapy as salvage therapy in metastatic gastric cancer patients. Cancer Chemother Pharmacol 65, 579–587 (2010). https://doi.org/10.1007/s00280-009-1067-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-009-1067-9