Abstract
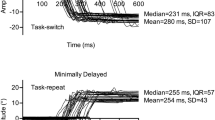
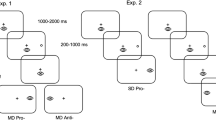
Eye movement circuitry involved in saccade production offers a model for studying cognitive control: visually guided prosaccades are stimulus-directed responses, while goal-driven antisaccades rely upon more complex control processes to inhibit the prepotent tendency to look toward a cue, transform its spatial location, and generate a volitional saccade in the opposite direction. By manipulating the relative probability of these saccade types, we measured participants’ behavioral responses to different levels of implicit trial-type probability and task-switching demands in conditions with relatively long inter-trial fixation and trial-type cue lengths. Results indicated that when prosaccades were less probable in a run, more prosaccade errors were generated; however, for antisaccades, trial-type probability had no effect on the percent of correct responses. For reaction times, specifically in runs with a larger probability of antisaccade trials, latencies increased for both anti- and pro-saccades. Furthermore, task switching resulted in a lower percentage of correct responses on switched trials, but a prior antisaccade trial led to slower reaction times for both trial types (i.e., a task switch cost for prosaccades and switch benefit for antisaccades). These findings indicate that cognitive control demands and residual inhibition from antisaccades alter performance relative to trial-type probability and task switching within a run, with the prosaccade task showing greater susceptibility to the influence of a large probability of cognitively complex antisaccades.



Similar content being viewed by others
References
Abegg M, Manoach DS, Barton JJ (2011) Knowing the future: partial foreknowledge effects on the programming of prosaccades and antisaccades. Vis Res 51:215–221. doi:10.1016/j.visres.2010.11.006
Allport DA, Styles EA, Hsieh S (1994) Shifting intentional set: exploring the dynamic control of tasks. In: Umiltà C, Moscovitch M (eds) Attention and performance 15: conscious and nonconscious information processing. Attention and performance series, 1047-0387 (Print). The MIT Press, Cambridge, MA, pp 421–452
Barton JJ, Greenzang C, Hefter R, Edelman J, Manoach DS (2006) Switching, plasticity, and prediction in a saccadic task-switch paradigm. Exp Brain Res 168:76–87. doi:10.1007/s00221-005-0091-1
Bell AH, Munoz DP (2008) Activity in the superior colliculus reflects dynamic interactions between voluntary and involuntary influences on orienting behaviour. Eur J Neurosci 28:1654–1660. doi:10.1111/j.1460-9568.2008.06393.x
Carpenter RH, Williams ML (1995) Neural computation of log likelihood in control of saccadic eye movements. Nature 377:59–62. doi:10.1038/377059a0
Cherkasova MV, Manoach DS, Intriligator JM, Barton JJ (2002) Antisaccades and task-switching: interactions in controlled processing. Exp Brain Res 144:528–537. doi:10.1007/s00221-002-1075-z
Chiau HY, Tseng P, Su JH, Tzeng OJ, Hung DL, Muggleton NG, Juan CH (2011) Trial type probability modulates the cost of antisaccades. J Neurophysiol 106:515–526. doi:10.1152/jn.00399.2010
Cnaan A, Laird NM, Slasor P (1997) Using the general linear mixed model to analyse unbalanced repeated measures and longitudinal data. Stat Med 16:2349–2380
Cox RW (1996) AFNI: software for analysis and visualization of functional magnetic resonance neuroimages. Comput Biomed Res 29:162–173
Cutsuridis V, Smyrnis N, Evdokimidis I, Perantonis S (2007) A neural model of decision-making by the superior colicullus in an antisaccade task. Neural Netw 20:690–704. doi:10.1016/j.neunet.2007.01.004
Dorris MC, Munoz DP (1998) Saccadic probability influences motor preparation signals and time to saccadic initiation. J Neurosci 18:7015–7026
Evdokimidis I et al (2002) The antisaccade task in a sample of 2,006 young men. I. Normal population characteristics. Exp Brain Res 147:45–52. doi:10.1007/s00221-002-1208-4
Everling S, Fischer B (1998) The antisaccade: a review of basic research and clinical studies. Neuropsychologia 36:885–899
Everling S, Johnston K (2013) Control of the superior colliculus by the lateral prefrontal cortex. Philos Trans R Soc Lond B Biol Sci 368:20130068. doi:10.1098/rstb.2013.0068
Everling S, Munoz DP (2000) Neuronal correlates for preparatory set associated with pro-saccades and anti-saccades in the primate frontal eye field. J Neurosci 20:387–400
Everling S, Dorris MC, Munoz DP (1998) Reflex suppression in the anti-saccade task is dependent on prestimulus neural processes. J Neurophysiol 80:1584–1589
Everling S, Dorris MC, Klein RM, Munoz DP (1999) Role of primate superior colliculus in preparation and execution of anti-saccades and pro-saccades. J Neurosci 19:2740–2754
Fecteau JH, Au C, Armstrong IT, Munoz DP (2004) Sensory biases produce alternation advantage found in sequential saccadic eye movement tasks. Exp Brain Res 159:84–91. doi:10.1007/s00221-004-1935-9
Franke C, Reuter B, Breddin A, Kathmann N (2009) Response switching in schizophrenia patients and healthy subjects: effects of the inter-response interval. Exp Brain Res 196:429–438. doi:10.1007/s00221-009-1871-9
Gueorguieva R, Krystal JH (2004) Move over ANOVA: progress in analyzing repeated-measures data and its reflection in papers published in the Archives of General Psychiatry. Arch Gen Psychiatry 61:310–317. doi:10.1001/archpsyc.61.3.310
Hallett PE (1978) Primary and secondary saccades to goals defined by instructions. Vis Res 18:1279–1296
Hanes DP, Schall JD (1996) Neural control of voluntary movement initiation. Science 274:427–430
Hoffman JE, Subramaniam B (1995) The role of visual attention in saccadic eye movements. Percept Psychophys 57:787–795
Hutton SB (2008) Cognitive control of saccadic eye movements. Brain Cogn 68:327–340. doi:10.1016/j.bandc.2008.08.021
Kan JY, Niel U, Dorris MC (2012) Evidence for a link between the experiential allocation of saccade preparation and visuospatial attention. J Neurophysiol 107:1413–1420. doi:10.1152/jn.00534.2011
Lee AK, Hamalainen MS, Dyckman KA, Barton JJ, Manoach DS (2011) Saccadic preparation in the frontal eye field is modulated by distinct trial history effects as revealed by magnetoencephalography. Cereb Cortex 21:245–253. doi:10.1093/cercor/bhq057
Littell RC, Pendergast J, Natarajan R (2000) Modelling covariance structure in the analysis of repeated measures data. Stat Med 19:1793–1819
Liu CL, Chiau HY, Tseng P, Hung DL, Tzeng OJ, Muggleton NG, Juan CH (2010) Antisaccade cost is modulated by contextual experience of location probability. J Neurophysiol 103:1438–1447. doi:10.1152/jn.00815.2009
Manoach DS, Thakkar KN, Cain MS, Polli FE, Edelman JA, Fischl B, Barton JJ (2007) Neural activity is modulated by trial history: a functional magnetic resonance imaging study of the effects of a previous antisaccade. J Neurosci 27:1791–1798. doi:10.1523/jneurosci.3662-06.2007
Massen C (2004) Parallel programming of exogenous and endogenous components in the antisaccade task. Q J Exp Psychol A 57:475–498. doi:10.1080/02724980343000341
McDowell JE, Dyckman KA, Austin BP, Clementz BA (2008) Neurophysiology and neuroanatomy of reflexive and volitional saccades: evidence from studies of humans. Brain Cogn 68:255–270
Meiran N (1996) Reconfiguration of processing mode prior to task performance. J Exp Psychol Learn Mem Cogn 22:1423–1442. doi:10.1037/0278-7393.22.6.1423
Miller EK, Cohen JD (2001) An integrative theory of prefrontal cortex function. Annu Rev Neurosci 24:167–202. doi:10.1146/annurev.neuro.24.1.167
Monsell S (2003) Task switching. Trends Cogn Sci 7:134–140
Monsell S, Yeung N, Azuma R (2000) Reconfiguration of task-set: is it easier to switch to the weaker task? Psychol Res 63:250–264
Morrell CH, Pearson JD, Brant LJ (1997) Linear transformations of linear mixed-effects models. Am Stat 51:338–343. doi:10.2307/2685902
Munoz DP, Everling S (2004) Look away: the anti-saccade task and the voluntary control of eye movement. Nat Rev Neurosci 5:218–228. doi:10.1038/nrn1345
Olk B, Kingstone A (2003) Why are antisaccades slower than prosaccades? A novel finding using a new paradigm. NeuroReport 14:151–155. doi:10.1097/01.wnr.0000053062.88427.d7
Pierrot-Deseilligny C, Muri RM, Ploner CJ, Gaymard B, Rivaud-Pechoux S (2003) Cortical control of ocular saccades in humans: a model for motricity. Prog Brain Res 142:3–17. doi:10.1016/s0079-6123(03)42003-7
Pierrot-Deseilligny C, Muri RM, Nyffeler T, Milea D (2005) The role of the human dorsolateral prefrontal cortex in ocular motor behavior. Ann N Y Acad Sci 1039:239–251. doi:10.1196/annals.1325.023
Reuter B, Philipp AM, Koch I, Kathmann N (2006) Effects of switching between leftward and rightward pro- and antisaccades. Biol Psychol 72:88–95. doi:10.1016/j.biopsycho.2005.08.005
Rogers RD, Monsell S (1995) Costs of a predictible switch between simple cognitive tasks. J Exp Psychol Gen 124:207–231. doi:10.1037/0096-3445.124.2.207
Smyrnis N et al (2002) The antisaccade task in a sample of 2,006 young males. II. Effects of task parameters. Exp Brain Res 147:53–63. doi:10.1007/s00221-002-1207-5
Stuyven E, Van der Goten K, Vandierendonck A, Claeys K, Crevits L (2000) The effect of cognitive load on saccadic eye movements. Acta Psychol (Amst) 104:69–85
Unsworth N, Schrock JC, Engle RW (2004) Working memory capacity and the antisaccade task: individual differences in voluntary saccade control. J Exp Psychol Learn Mem Cogn 30:1302–1321. doi:10.1037/0278-7393.30.6.1302
Vandierendonck A, Liefooghe B, Verbruggen F (2010) Task switching: interplay of reconfiguration and interference control. Psychol Bull 136:601–626. doi:10.1037/a0019791
Verbeke G, Molenberghs G, Rizopoulos D (2010) Random effects models for longitudinal data. In: van Montfort K, Oud JHL, Satorra A (eds) Longitudinal research with latent variables. Springer, Berlin. doi:10.1007/978-3-642-11760-2
Vermeiren A, Liefooghe B, Vandierendonck A (2010) Switch performance in peripherally and centrally triggered saccades. Exp Brain Res 206:243–248. doi:10.1007/s00221-010-2401-5
Weiler J, Heath M (2012) Task-switching in oculomotor control: unidirectional switch-cost when alternating between pro- and antisaccades. Neurosci Lett 530:150–154. doi:10.1016/j.neulet.2012.10.007
Weiler J, Heath M (2014) Repetitive antisaccade execution does not increase the unidirectional prosaccade switch-cost. Acta Psychol (Amst) 146:67–72. doi:10.1016/j.actpsy.2013.12.005
Weiler J, Mitchell T, Heath M (2014) Response suppression delays the planning of subsequent stimulus-driven saccades. PLoS One 9:e86408. doi:10.1371/journal.pone.0086408
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pierce, J.E., McCardel, J.B. & McDowell, J.E. Trial-type probability and task-switching effects on behavioral response characteristics in a mixed saccade task. Exp Brain Res 233, 959–969 (2015). https://doi.org/10.1007/s00221-014-4170-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-014-4170-z