Abstract
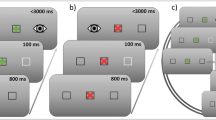
When the eyes fixate at a point in a visual scene, small saccades rapidly shift the image on the retina. The effect of these microsaccades on the latency of subsequent large-scale saccades may be twofold. First, microsaccades are associated with an enhancement of visual perception. Their occurrence during saccade target perception could, thus, decrease saccade latencies. Second, microsaccades are likely to indicate activity in fixation-related oculomotor neurons. These represent competitors to saccade-related cells in the interplay of gaze holding and shifting. Consequently, an increase in saccade latencies would be expected after microsaccades. Here, we present evidence for both aspects of microsaccadic impact on saccade latency. In a delayed response task, participants made saccades to visible or memorized targets. First, microsaccade occurrence up to 50 ms before target disappearance correlated with 18 ms (or 8%) faster saccades to memorized targets. Second, if microsaccades occurred shortly (i.e., <150 ms) before a saccade was required, mean saccadic reaction time in visual and memory trials was increased by about 40 ms (or 16%). Hence, microsaccades can have opposite consequences for saccade latencies, pointing at a differential role of these fixational eye movements in the preparation of saccade motor programs.




Similar content being viewed by others
Notes
Separate ANOVAs were performed because of the differences in the number of missing cells.
Criteria for trial rejection applied here partly differed from those used by Supèr et al. (2004). A post-hoc rejection of trials were fixation left an area of 1×1° (as it was done on-line by Supèr et al.) did not significantly change any of the results reported here.
We want to emphasize that the continuum of movement fields along the rostral–caudal dimension extends to saccade amplitudes well below 1° as observed in the cited studies. Thus, microsaccades fall in the range of amplitudes represented in the very rostral pole of the SC.
References
Bair W, O’Keefe LP (1998) The influence of fixational eye movements on the response of neurons in area MT of the macaque. Vis Neurosci 15:779–786
Brainard DH (1997) The Psychophysics Toolbox. Spat Vis 10:433–436
Bruce CJ, Friedman HR, Kraus MS, Stanton GB (2004) The primate frontal eye field. In: Chalupa LM, Werner JS (eds) The visual neurosciences, vol 2. MIT Press, Cambridge, MA, pp 1428–1448
Cornelissen FW, Peters EM, Palmer J (2002) The Eyelink Toolbox: Eye tracking with MATLAB and the Psychophysics Toolbox. Behav Res Methods Instrum Comput 34:613–617
Ditchburn RW (1980) The function of small saccades. Vision Res 20:271–272
Ditchburn RW, Ginsborg BL (1953) Involuntary eye movements during fixation. J Physiol 119:1–17
Engbert R, Kliegl R (2003) Microsaccades uncover the orientation of covert attention. Vision Res 43:1035–1045
Engbert R (2005) Microsaccades: a microcosm for research on oculomotor control, attention, and visual perception. Prog Brain Res (in press)
Friedman HR, Burman DD, Russo GS, Dias EC, Stanton GB, Shi D, Bruce CJ (1997) Neuronal activity in primate frontal eye field during memory-guided saccades. Abstr Soc Neurosci 24:522
Galfano G, Betta E, Turatto M (2004) Inhibition of return in microsaccades. Exp Brain Res 159:400–404
Gandhi NJ, Keller EL (1999) Comparison of saccades perturbed by stimulation of the rostral superior colliculus, the caudal superior colliculus, and the omnipause neuron region. J Neurophysiol 82:3236–3253
Gerrits HJM, Vendrik AJH (1974) The influence of stimulus movements on perception in parafoveal stabilized vision. Vision Res 14:175–180
Krauskopf J, Cornsweet TN, Riggs LA (1960) Analysis of eye movements during monocular and binocular fixation. J Opt Soc Am 50:572–578
Krauzlis RJ, Basso MA, Wurtz RH (1997) Shared motor error for multiple eye movements. Science 276:1693–1695
Laubrock J, Engbert R, Kliegl R (2005) Microsaccade dynamics during covert attention. Vision Res 45:721–730
Leopold DA, Logothetis NK (1998) Microsaccades differentially modulate neural activity in the striate and extrastriate visual cortex. Exp Brain Res 123:341–345
Lord MP (1951) Measurement of binocular eye movements of subjects in the sitting position. Br J Ophthalmol 35:21–30
Martinez-Conde S, Macknik SL, Hubel DH (2000) Microsaccadic eye movements and firing of single cells in the striate cortex of macaque monkeys. Nat Neurosci 3:251–258
Martinez-Conde S, Macknik SL, Hubel DH (2002) The function of bursts of spikes during visual fixation in the awake primate lateral geniculate nucleus and primary visual cortex. Proc Natl Acad Sci USA 99:13920–13925
Martinez-Conde S, Macknik SL, Hubel DH (2004) The role of fixational eye movements in visual perception. Nat Rev Neurosci 5:229–240
Munoz DP, Fecteau JH (2002) Vying for dominance: Dynamic interactions control visual fixation and saccadic initiation in the superior colliculus. Prog Brain Res 140:3–19
Munoz DP, Guitton D (1991) Control of orienting gaze shifts by the tectoreticulospinal system in the head-free cat II. Sustained discharges during motor preparation and fixation. J Neurophysiol 66:1624–1641
Munoz DP, Istvan PJ (1998) Lateral inhibitory interactions in the intermediate layers of the monkey superior colliculus. J Neurophysiol 79:1193–1209
Munoz DP, Wurtz RH (1993a) Fixation cells in monkey superior colliculus. I. Characteristics of cell discharge. J Neurophysiol 70:559–575
Munoz DP, Wurtz RH (1993b) Fixation cells in monkey superior colliculus. II. Reversible activation and deactivation. J Neurophysiol 70:576–589
Munoz DP, Wurtz RH (1995a) Saccade-related activity in monkey superior colliculus. I. Characteristics of burst and buildup cells. J Neurophysiol 73:2313–2333
Munoz DP, Wurtz RH (1995b) Saccade-related activity in monkey superior colliculus. II. Spread of activity during saccades. J Neurophysiol 73:2334–2348
Munoz DP, Dorris MC, Paré M, Everling S (2000) On your mark get set: Brainstem circuitry underlying saccadic initiation. Can J Physiol Pharmacol 78:934–944
Pelli DG (1997) The VideoToolbox software for visual psychophysics: Transforming numbers into movies. Spat Vis 10:437–442
Ratliff F, Riggs LA (1950) Involuntary motions of the eye during monocular fixation. J Exp Psychol 40:687–701
Robinson DA (1972) Eye movements evoked by collicular stimulation in the alert monkey. Vision Res 12:1795–1808
Rolfs M, Engbert R, Kliegl R (2004) Microsaccade orientation supports attentional enhancement opposite a peripheral cue. Psychol Sci 15:705–707
Rolfs M, Engbert R, Kliegl R (2005) Crossmodal coupling of oculomotor control and spatial attention in vision and audition. Exp Brain Res 166:427–439
Schall JD, Morel A, King DJ, Bullier J (1995) Topography of visual cortex connections with frontal eye field in macaque: Convergence and segregation of processing streams. J Neurosci 15:4464–4487
Scudder CA, Kaneko CRS, Fuchs AF (2002) The brainstem burst generator for saccadic eye movements: A modern synthesis. Exp Brain Res 142:439–462
Snodderly DM, Kagan I, Gur M (2001) Selective activation of visual cortex neurons by fixational eye movements: Implications for neural coding. Vis Neurosci 18:259–277
Sparks DL (2002) The brainstem control of saccadic eye movements. Nat Rev Neurosci 3:952–964
Sparks DL, Holland R, Guthrie BL (1976) Size and distribution of movement fields in the monkey superior colliculus. Brain Res 113:21–34
Supèr H, van der Togt C, Spekreijse H, Lamme VAF (2004) Correspondence of presaccadic activity in the monkey primary visual cortex with saccadic eye movements. Proc Natl Acad Sci USA 101:3230–3235
Wurtz RH, Goldberg ME (1972) Activity of superior colliculus in behaving monkey. III. Cells discharging before eye movements. J Neurophysiol 35:575–586
Zuber BL, Stark L, Cook G (1965) Microsaccades and the velocity–amplitude relationship for saccadic eye movements. Science 150:1459–1460
Acknowledgments
We thank Eike Martin Richter, Ralf Engbert, and two anonymous reviewers for their helpful conversation and/or comments on earlier drafts of the manuscript. This research was supported by Deutsche Forschungsgemeinschaft (Grants KL955/3 and /6).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rolfs, M., Laubrock, J. & Kliegl, R. Shortening and prolongation of saccade latencies following microsaccades. Exp Brain Res 169, 369–376 (2006). https://doi.org/10.1007/s00221-005-0148-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-005-0148-1