Abstract
Rationale
Although numerous studies have documented that nicotine can function as an effective reinforcer of intravenous self-administration behavior in animals, it has not been clearly shown to maintain intravenous self-administration behavior above vehicle placebo levels in humans.
Objectives
To compare the reinforcing effectiveness of nicotine versus saline placebo in human research volunteers responding under fixed-ratio (FR) schedules of intravenous drug self-administration while systematically increasing response requirements.
Methods
Eight male cigarette smokers resided in an inpatient research unit. During 3-h sessions, intravenous injections of nicotine and saline were available concurrently and were contingent on responding (pulling a lever). Nicotine dose (0.75, 1.5, 3.0 mg/injection), time out (TO) value after each injection (1–20 min) and FR response requirement (10–1600) were varied in different subjects over consecutive sessions.
Results
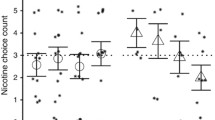
Number of nicotine injections/session significantly decreased as dose/injection increased and the number of self-administered nicotine injections was significantly greater than the number of self-administered saline injections across conditions. When FR value was progressively increased over sessions, response rates for nicotine, but not saline, injections increased, with maximal rates at the highest FR values. Rates of responding and injections/session were markedly and significantly higher for nicotine than for saline at FR values of 200 and above. Subjects rated effects of nicotine as both significantly more positive and more negative than saline placebo, with positive ratings significantly higher than negative ratings.
Conclusions
Nicotine functioned as a prototypic drug of abuse, serving as an effective reinforcer of intravenous drug-taking behavior in human cigarette smokers. Subjects adjusted their responding to response requirements in a way that maintained relatively constant levels of nicotine injections per session.




Similar content being viewed by others
References
Ator NA, Griffiths RR (1983) Nicotine self-administration in baboons. Pharmacol Biochem Behav 19:993–1003
Comer SD, Collins ED (2002) Self-administration of intravenous buprenorphine and the buprenorphine/naloxone combination by recently detoxified heroin abusers. J Pharmacol Exp Ther 303:695–703
Corrigall WA (1999) Nicotine self-administration in animals as a dependence model. Nicotine Tob Res 1:11–20
Corrigall WA, Coen KM (1989) Nicotine maintains robust self-administration in rats on a limited-access schedule. Psychopharmacology 99:473–478
Donny EC, Caggiula AR, Mielke MM, Jacobs KS, Rose C, Sved AF (1998) Acquisition of nicotine self-administration in rats: the effects of dose, feeding schedule, and drug contingency. Psychopharmacology 136:83–90
Donny EC, Caggiula AR, Mielke MM, Booth S, Gharib MA, Hoffman A, Maldovan V, Shupenko C, McCallum SE (1999) Nicotine self-administration in rats on a progressive ratio schedule of reinforcement. Psychopharmacology 147:135–142
Ettenberg A, Geist TD (1991) Animal model for investigating the anxiogenic effects of self-administered cocaine. Psychopharmacology 103:455–461
Fiore MC, Bailey WC, Cohen SJ et al. (2000) Treating tobacco use and dependence, Clinical Practice Guideline. US Department of Health and Human Services, Rockville, Md.
Garrett BE, Griffiths RR (2001) Intravenous nicotine and caffeine: subjective and physiological effects in cocaine abusers. J Pharmacol Exp Ther 296:486–494
Giordano LA, Bickel WK, Shahan TA, Badger GJ (2001) Behavioral economics of human drug self-administration: progressive ratio versus random sequences of response requirements. Behav Pharmacol 12:343–347
Goldberg SR, Spealman RD (1982) Maintenance and suppression of behavior by intravenous nicotine injections in squirrel monkeys. Fed Proc 41:216–220
Goldberg SR, Spealman RD, Goldberg DM (1981) Persistent behavior at high rates maintained by intravenous self-administration of nicotine. Science 214:573–575
Goldberg SR, Spealman RD, Risner ME, Henningfield JE (1983) Control of behavior by intravenous nicotine injections in laboratory animals. Pharmacol Biochem Behav 19:1011–1020
Griffiths RR, Bigelow GE, Henningfield JE (1980) Similarities in animal and human drug-taking behavior. In: Mello NK (ed) Advances in substance abuse. JAI Press, Greenwich, pp 1–90
Heishman SJ, Schuh KJ, Schuster CR, Henningfield JE, Goldberg SR (2000) Reinforcing and subjective effects of morphine in human opioid abusers: effect of dose and alternative reinforcer. Psychopharmacology 148:272–280
Henningfield JE, Goldberg SR (1983) Control of behavior by intravenous nicotine injections in human subjects. Pharmacol Biochem Behav 19:1021–1026
Henningfield JE, Miyasato K, Jasinski DR (1983) Cigarette smokers self-administer intravenous nicotine. Pharmacol Biochem Behav 19:887–890
Henningfield JE, Miyasato K, Jasinski DR (1985) Abuse liability and pharmacodynamic characteristics of intravenous and inhaled nicotine. J Pharmacol Exp Ther 234:1–12
Hughes JR, Pickens RW, Spring W, Keenan RM (1985) Instructions control whether nicotine will serve as a reinforcer. J Pharmacol Exp Ther 235:106–112
Hughes JR, Gust SW, Skoog K, Keenan RM, Fenwick JW (1991) Symptoms of tobacco withdrawal. A replication and extension. Arch Gen Psychiatry 48:52–59
Hughes JR, Rose GL, Callas PW (2000) Nicotine is more reinforcing in smokers with a past history of alcoholism than in smokers without this history. Alcohol Clin Exp Res 24:1633–1638
Hursh SR, Winger G (1995) Normalized demand for drugs and other reinforcers. J Exp Anal Behav 64:373–384
Johnson MW, Bickel WK (2003) The behavioral economics of cigarette smoking: the concurrent presence of a substitute and an independent reinforcer. Behav Pharmacol 14:137–144
Jones HE, Griffiths RR (2003) Oral caffeine maintenance potentiates the reinforcing and stimulant subjective effects of intravenous nicotine in cigarette smokers. Psychopharmacology 165:280–290
Jones HE, Garrett BE, Griffiths RR (1999) Subjective and physiological effects of intravenous nicotine and cocaine in cigarette smoking cocaine abusers. J Pharmacol Exp Ther 288:188–197
Kalman D (2002) The subjective effects of nicotine: methodological issues, a review of experimental studies, and recommendations for future research. Nicotine Tob Res 4:25–70
Lamb RJ, Preston KL, Schindler CW, Meisch RA, Davis F, Katz JL, Henningfield JE, Goldberg SR (1991) The reinforcing and subjective effects of morphine in post-addicts: a dose-response study. J Pharmacol Exp Ther 259:1165–1173
Lynch WJ, Carroll ME (1999) Regulation of intravenously administered nicotine in rats. Exp Clin Psychopharmacol 7:198–207
Panlilio LV, Katz JL, Pickens RW, Schindler CW (2003) Variability of drug self-administration in rats. Psychopharmacology 167:9–19
Perkins KA, Grobe JE, Weiss D, Fonte C, Caggiula (1996) Nicotine preference in smokers as a function of smoking abstinence. Pharmacol Biochem Behav 55:257–263
Perkins KA, Grobe JE, Caggiula A, Wilson AS, Stiller RL (1997) Acute reinforcing effects of low-dose nicotine nasal spray in humans. Pharmacol Biochem Behav 56:235–241
Risner ME, Goldberg SR (1983) A comparison of nicotine and cocaine self-administration in the dog: fixed-ratio and progressive-ratio schedules of intravenous drug infusion. J Pharmacol Exp Ther 224:319–326
Rose JE, Behm FM, Westman EC, Bates JE (2003) Mecamylamine acutely increases human nicotine self-administration. Pharmacol Biochem Behav 76:307–313
Sannerud CA, Prada J, Goldberg DM, Goldberg SR (1994) The effects of sertraline on nicotine self-administration and food-maintained responding in squirrel monkeys. Eur J Pharmacol 271:461–469
Schuster CR, Fischman MW, Johanson CE (1981) Internal stimulus control and subjective effects of drugs. NIDA Res Monogr 37:116–129
Shoaib M, Schindler CW, Goldberg SR (1997) Nicotine self-administration in rats: strain and nicotine pre-exposure effects on acquisition. Psychopharmacology 129:35–43
Shoaib M, Swanner LS, Yasar S, Goldberg SR (1999) Chronic caffeine exposure potentiates nicotine self-administration in rats. Psychopharmacology 142:327–333
Singer JD (1998) Using SAS PROC MIXED to fit multilevel models, hierarchical models, and individual growth models. J Ed Behav Stat 24:323–355
Slifer BL, Balster RL (1985) Intravenous self-administration of nicotine with and without schedule-induction. Pharmacol Biochem Behav 22:61–69
Soria R, Stapleton JM, Gilson SF, Sampson-Cone A, Henningfield JE, London ED (1996) Subjective and cardiovascular effects of intravenous nicotine in smokers and non-smokers. Psychopharmacology 128:221–226
Spealman RD, Goldberg SR (1982) Maintenance of schedule-controlled behavior by intravenous injections of nicotine in squirrel monkeys. J Pharmacol Exp Ther 223:402–408
Stolerman IP, Naylor C, Elmer GI, Goldberg SR (1999) Discrimination and self-administration of nicotine by inbred strains of mice. Psychopharmacology 141:297–306
US Department of Health and Human Services (1988) The health consequences of smoking: Nicotine addiction (DHHS Publication No. CDC 88-8406). US Government Printing Office, Washington D.C.
Wakasa Y, Takada K, Yanagita T (1995) Reinforcing effect as a function of infusion speed in intravenous self-administration of nicotine in rhesus monkeys. Nihon Shinkei Seishin Yakurigaku Zasshi 15:53–59
Winger G, Hursh SR, Casey KL, Woods JH (2002) Relative reinforcing strength of three n-methyl-d-aspartate antagonists with different onsets of action. J Pharmacol Exp Ther 301:690–697
Acknowledgements
This study was supported by NIDA, NIH and DHHS. We would like to thank Drs. David Epstein and Zuzana Justinova for their help in analyzing the data and Mr. Richard Taylor for his help in retrieving subject histories.
Author information
Authors and Affiliations
Corresponding author
Additional information
Abstracts of these experiments previously appeared in Pharmacologist 25:219, 1983, and Neurosci Lett 14(Suppl):140, 1983.
Rights and permissions
About this article
Cite this article
Harvey, D.M., Yasar, S., Heishman, S.J. et al. Nicotine serves as an effective reinforcer of intravenous drug-taking behavior in human cigarette smokers. Psychopharmacology 175, 134–142 (2004). https://doi.org/10.1007/s00213-004-1818-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-004-1818-6