Abstract
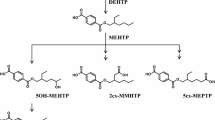
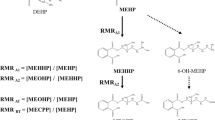
The metabolism of di(2-ethylhexyl)phthalate (DEHP) in humans was studied after three doses of 0.35 mg (4.7 μg/kg), 2.15 mg (28.7 μg/kg) and 48.5 mg (650 μg/kg) of D4-ring-labelled DEHP were administered orally to a male volunteer. Two new metabolites, mono(2-ethyl-5-carboxypentyl)phthalate (5cx-MEPP) and mono[2-(carboxymethyl)hexyl]phthalate (2cx-MMHP) were monitored for 44 h in urine and for 8 h in serum for the high-dose case, in addition to the three metabolites previously analysed: mono(2-ethyl-5-hydroxyhexyl)phthalate (5OH-MEHP), mono(2-ethyl-5-oxohexyl)phthalate (5oxo-MEHP) and mono(2-ethylhexyl)phthalate (MEHP). For the medium- and low-dose cases, 24 h urine samples were analysed. Up to 12 h after the dose, 5OH-MEHP was the major urinary metabolite, after 12 h it was 5cx-MEPP, and after 24 h it was 2cx-MMHP. The elimination half-lives of 5cx-MEHP and 2cx-MMHP were between 15 and 24 h. After 24 h 67.0% (range: 65.8–70.5%) of the DEHP dose was excreted in urine, comprising 5OH-MEHP (23.3%), 5cx-MEPP (18.5%), 5oxo-MEHP (15.0%), MEHP (5.9%) and 2cx-MMHP (4.2%). An additional 3.8% of the DEHP dose was excreted on the second day, comprising 2cx-MMHP (1.6%), 5cx-MEPP (1.2%), 5OH-MEHP (0.6%) and 5oxo-MEHP (0.4%). In total about 75% of the administered DEHP dose was excreted in urine after two days. Therefore, in contrast to previous studies, most of the orally administered DEHP is systemically absorbed and excreted in urine. No dose dependency in metabolism and excretion was observed. The secondary metabolites of DEHP are superior biomonitoring markers compared to any other parameters, such as MEHP in urine or blood. 5OH-MEHP and 5oxo-MEHP in urine reflect short-term and 5cx-MEHP and 2cx-MMHP long-term exposure. All secondary metabolites are unsusceptible to contamination. Furthermore, there are strong hints that the secondary oxidised DEHP metabolites—not DEHP or MEHP—are the ultimate developmental toxicants.







Similar content being viewed by others
References
Akingbemi BT, Youker RT, Sottas CM, Ge R, Katz E, Klinefelter GR, Zirkin BR, Hardy MP (2001) Modulation of rat Leydig cell steroidogenic function by di(2-ethylhexyl)phthalate. Biol Reprod 65(4):1252–1259
Albro PW, Corbett JT, Schroeder JL, Jordan ST, Matthews HB (1982) Pharmacokinetics, interactions with macromolecules and species differences in metabolism of DEHP. Environ Health Perspect 45:19–25
ATSDR Agency for Toxic Substances and Disease Registry (1993) Toxicological Profile for di(2-ethylhexyl)phthalate. Update. Final report, ATSDR/TP-92/05, NTIS Accession No. PB93-182400. ATSDR, Public Health Service, U.S. Department of Health and Human Services, Atlanta, GA, pp 171
Barr DB, Silva MJ, Kato K, Reidy JA, Malek NA, Hurtz D, Sadowski M, Needham LL, Calafat AM (2003) Assessing human exposure to phthalates using monoesters and their oxidized metabolites as biomarkers. Environ Health Perspect 111(9):1148–1151
Becker K, Seiwert M, Angerer J, Heger W, Koch HM, Nagorka R, Rosslamp E, Schluter C, Seifert B, Ullrich D (2004) DEHP metabolites in urine of children and DEHP in house dust. Int J Environ Health 207(5):409–417
CSTEE—Scientific Committee for Toxicity, Ecotoxicity and the Environment (2004) Opinion on the results of a second risk assessment of bis(2-ethylhexyl)phthalate[DEPH]. Human health part. Adopted by the CSTEE during the 41th plenary meeting of 8 January 2004. European Commission, Brussels (see http://europa.eu.int/comm/health/ph_risk/committees/sct/documents/out214_en.pdf)
CSTEE—Scientific committee on toxicity, ecotoxicity and the environment (1998) Opinion on phthalate migration from soft PVC toys and child-care articles—data made available since the 16th of June 1998, opinion expressed at the 6th CSTEE plenary meeting, Brussels, 26/27 November 1998. European Commission, Brussels
DFG, Deutsche Forschungsgemeinschaft (2002) Di(2-ethylhexyl)phthalat (DEHP). In: H Greim (ed) Gesundheitsschädliche Arbeitsstoffe—Toxikologisch-arbeitsmedizinische Begründungen von MAK-Werten. 35. Erganzüngslieferung, Wiley, New York
Dirven HA, van den Broek PH, Jongeneelen FJ (1993) Determination of four metabolites of the plasticizer di(2-ethylhexyl)phthalate in human urine samples. Int Arch Occup Environ Health 64(8):555–560
EPA—US Environmental Protection Agency (1999) Integrated risk information system on di(2-ethylhexyl)phthalate. National Center for Environmental Assessment, Office of Research and Development, Washington DC
Fisher JS (2004) Environmental anti-androgens and male reproductive health: focus on phthalates and testicular dysgenesis syndrome. Reproduction 127(3):305–315
Gilsing H-D, Angerer J, Prescher D (2002) Monophthalates with oxidized C5-carbon in the ester chain: a simple synthetic access to two major metabolites of bis-(2-ethylhexyl)-phthalate. Monatsh Chem 133:1147–1155
Gilsing HD, Angerer J, Prescher D (2003) (2-Ethyl-5-oxo-hexyl)methylthiomethyl phthalate as by-product of the Swern oxidation: Improved synthesis of ring-deuterated major metabolites of bis(2-ethylhexyl) phthalate. Monatsh Chem 134(9):1207–1213
Gilsing HD, Angerer J, Prescher D (2005) Convenient and high-yielding preparation procedures for mono(5-carboxy-2-ethylpentyl) phthalate and its ring-deuterated isomer—the “third” major metabolite of bis(2-ethylhexyl) phthalate. Monatsh Chem (in press)
Kato K, Silva MJ, Brock JW, Reidy JA, Malek NA, Hodge CC, Nakazawa H, Needham LL, Barr DB (2003) Quantitative detection of nine phthalate metabolites in human serum using reversed-phase high-performance liquid chromatography-electrospray ionization-tandem mass spectrometry. J Anal Toxicol 27(5):284–289
Kato K, Silva MJ, Reidy JA, Hurtz D III, Malek NA, Needham LL, Nakazawa H, Barr DB, Calafat AM (2004) Mono(2-ethyl-5-hydroxyhexyl) phthalate and mono-(2-ethyl-5-oxohexyl) phthalate as biomarkers for human exposure assessment to di-(2-ethylhexyl) phthalate. Environ Health Perspect 112(3):327–330
Kavlock R, Boeckelheide K, Chapin R, Cunningham M, Faustman E, Foster P et al. (2002) NTP Center for the Evaluation of Risks to Human Reproduction: Phthalates expert panel report on the reproductive and developmental toxicity of di(2-ethylhexyl)phthalate. Reprod Toxicol 16:529–653
Kessler W, Numtip W, Grote K, Csanady GA, Chahoud I, Filser JG (2004) Blood burden of di(2-ethylhexyl) phthalate and its primary metabolite mono(2-ethylhexyl) phthalate in pregnant and nonpregnant rats and marmosets. Toxicol Appl Pharmacol 195(2):142–153
Koch HM, Gonzalez-Reche LM, Angerer J (2003a) On-line cleanup by multidimensional LC-ESI-MS/MS for high throughput quantification of primary and secondary phthalate metabolites in human urine. J Chromatogr B 784:169–182
Koch HM, Rossbach B, Dexler H, Angerer J (2003b) Internal exposure of the general population to DEHP and other phthalates—determination of secondary and primary phthalate monoester metabolites in urine. Environ Res 93(2):177–185
Koch HM, Drexler H, Angerer J (2003c) An estimation of the daily intake of di(2-ethylhexyl)phthalate (DEHP) and other phthalates in the general population. Int J Hyg Environ Health 206(2):77–83
Koch HM, Drexler H, Angerer J (2004a) Internal exposure of nursery-school children and their parents and teachers to di(2-ethylhexyl)phthalate (DEHP). Int J Hyg Environ Health 207(1):15–22
Koch HM, HM Bolt, Angerer J (2004c) Di(2-ethylhexyl)phthalate (DEHP) metabolites in human urine and serum after a single oral dose of deuterium labelled DEHP. Arch Toxicol 78:123–130
Koch HM, Drexler H, Angerer J (2004b) Response to the letter of R. M. David (Int J Hyg Environ Health 207:75–76). Int J Hyg Environ Health 207(1):77–78
Lamb JC IV, Chapin RE, Teague J, Lawton AD, Reel JR (1987) Reproductive effects of four phthalic acid esters in the mouse. Toxicol Appl Pharmacol 88(2):255–269
Ljungvall K, Tienpont B, David F, Magnusson U, Torneke K (2004) Kinetics of orally administered di(2-ethylhexyl) phthalate and its metabolite, mono(2-ethylhexyl) phthalate, in male pigs. Arch Toxicol 78(7):384–389
Lovekamp-Swan T, Davis BJ (2003) Mechanisms of phthalate ester toxicity in the female reproductive system. Environ Health Perspect 111(2):139–145
Moore RW, Rudy TA, Lin TM, Ko K, Peterson RE (2001) Abnormalities of sexual development in male rats with in utero and lactational exposure to the antiandrogenic plasticizer di(2-ethylhexyl) phthalate. Environ Health Perspect 109(3):229–237
Parks LG, Ostby JS, Lambright CR, Abbott BD, Klinefelter GR, Barlow NJ, Gray LE Jr (2000) The plasticizer diethylhexyl phthalate induces malformations by decreasing fetal testosterone synthesis during sexual differentiation in the male rat. Toxicol Sci 58(2):339–349
Peck CC, Albro PW (1982) Toxic potential of the plasticizer di(2-ethylhexyl) phthalate in the context of its disposition and metabolism in primates and man. Environ Health Perspect 45:11–17
Preuss R, Koch HM, Angerer J (2005) Biological monitoring of the major metabolites of di-(2-ethylhexyl)phthalate (DEHP) in human urine using column-switching liquid chromatography—tandem mass spectrometry. J Chromatogr B 816:269–280
Regnier J, Bowden C, Lhuguenot J (2004) Effects on rat embryonic development in vitro of di-(2-ethylhexyl) phthalate (DEHP) and its metabolites. The Toxicologist CD—an Official Journal of the Society of Toxicology 78(1):187
Schmid P, Schlatter C (1985) Excretion and metabolism of di(2-ethylhexyl)phthalate in man. Xenobiotica 15(3):251–256
Sharpe RM, Irvine DS (2004) How strong is the evidence of a link between environmental chemicals and adverse effects on human reproductive health? BMJ 328:447–451
Silva MJ, Reidy JA, Herbert AR, Preau JL, Needham LL, Calafat AM (2004) Detection of phthalate metabolites in human amniotic fluid. Bull Environ Contam Toxicol 72(6):1226–1231
Wolfe GW, Layton KA (2003) Multigeneration reproduction toxicity study in rats (unaudited draft): diethylhexylphthalate:multigenerational reproductive assessment by continuous breeding when administered to Spragur-Dawley rats in the diet (TRC study No 7244-200). Therimmune Research Corporation, Gaithersburg, MD
Acknowledgements
We especially thank the DFG (German Research Foundation) for their financial support of the project (No.: AN 107/16-1-3).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koch, H.M., Bolt, H.M., Preuss, R. et al. New metabolites of di(2-ethylhexyl)phthalate (DEHP) in human urine and serum after single oral doses of deuterium-labelled DEHP. Arch Toxicol 79, 367–376 (2005). https://doi.org/10.1007/s00204-004-0642-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-004-0642-4