Abstract
Purpose
To compare relative and absolute dose–volume parameters (DV) of the rectum and their clinical correlation with acute and late radiation proctitis (RP) after radiotherapy (RT) for prostate cancer (PCa).
Patients and methods
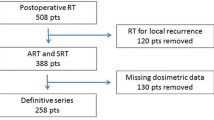
366 patients received RT for PCa. In total, 49.2% received definitive RT, 20.2% received postoperative RT and 30.6% received salvage RT for biochemical recurrence. In 77.9% of patients, RT was delivered to the prostate or prostate bed, and additional whole pelvic RT was performed in 22.1%. 33.9% received 3D-RT, and 66.1% received IMRT. The median follow-up was 59.5 months (18.0–84.0 months). The relative (in %) and absolute (in ccm) rectal doses from 20–75 Gy including the receiver operating characteristics curves (rAUC) from 30–65 Gy (in % and ccm) and several other clinical parameters were analyzed in univariate and multivariate analyses. We performed the statistical analyses separately for the entire cohort (n = 366), patients with (n = 81) and without (n = 285) pelvic RT, comparing RP vs. RP ≥ grade I.
Results
With the exception of the V50Gyccm (p = 0.02) in the univariate analyses for acute RP in the entire patient cohort, no absolute DV parameter (in ccm) was statistically significant associated with either acute or late RP. In the multivariate analyses, 3D-RT (p < 0.008) and rAUCV30–50 Gy% (p = 0.006) were significant parameters for acute RP for the entire cohort, and the V50Gy% (p = 0.01) was the significant parameter for patients with pelvic RT. The rAUCV40–50 Gy% (p = 0.004) was significant for RT to the prostate/prostate bed. Regarding the statistical analysis for late RP, the rAUCV30–65 Gy% (p = 0.001) was significant for the entire cohort, and rAUCV30–50 Gy% (p = 0.001) was significant for RT of the prostate/prostate bed. No parameter was significant in patients with pelvic RT.
Conclusion
Absolute DV parameters in ccm are not required for RT in PCa patients.
Zusammenfassung
Ziel
Vergleich von absoluter und relativer Dosisvolumenbelastung des Rektums und Korrelation mit akuter und später radiogener Proktitis (RP) nach Radiotherapie (RT) beim Prostatakarzinom (PCa)
Patienten und Methodik
366 PCa-Patienten erhielten eine RT: 49,2 % eine definitive, 20,2 % eine postoperative und 30,9 % eine Salvage-RT. Bei 78,3 % wurde Prostata oder Prostatabett bestrahlt und bei 21,7 % zusätzlich der Beckenlymphabfluss. Die Dosen betrugen 60 Gy bei postoperativer, 66 Gy bei Salvage- und 74–78 Gy bei definitiver RT. Eine 3D-RT erhielten 33,9 %, eine IMRT 66,1 %. Das mediane Follow-up betrug 59.5 Monate (18.0–84.0 Monate). Die relativen (%) und absoluten (ccm) rektalen Dosisvolumina (DV) von 20–75 Gy, sowie die Fläche unter der Kurve (rAUC) von 30–65 Gy (in % und ccm) und weitere klinische Parameter wurden univariat und multivariat untersucht. Die statistische Auswertung hinsichtlich keine RP vs. RP ≥ Grad I wurde für das gesamte Kollektiv (n = 366), für Patienten mit (n = 81) und ohne (n = 285) pelvine RT durchgeführt.
Ergebnisse
Mit Ausnahme der V50Gyccm (p = 0,02) in der univariaten Analyse für die akute RP im Gesamtkollektiv war kein absoluter DV-Parameter in ccm signifikant mit akuter oder später RP assoziiert. In der multivariaten Analyse waren die 3‑D-RT (p < 0,008) und die rAUCVGy30–50% (p = 0,006) im Gesamtkollektiv und die V50Gy% (p = 0,01) bei pelviner RT signifikant mit einer akuten RP assoziiert. Die rAUCVGy40–50% (p = 0,004) war bei der Bestrahlung beschränkt auf Prostata/Prostatabett multivariat signifikant. Hinsichtlich der späten RP war die rAUCVGy30–65% (p = 0,001) für das Gesamtkollektiv und die rAUCVGy30–50% (p = 0.001) für die Bestrahlung von Prostata/Prostatabett signifikant. Kein Parameter war multivariat signifikant für die späte RP bei additiver Beckenbestrahlung.
Schlussfolgerung
Absolute DV-Parameter in ccm zusätzlich zu relativen DV „constraints“ des Rektums werden für die RT beim PCa nicht benötigt.


Similar content being viewed by others
References
Wortel RC, Incrocci L, Pos FJ, van der Heide UA, Lebesque JV, Aluwini S et al (2016) Late side effects after image guided intensity modulated radiation therapy compared to 3D-Conformal radiation therapy for prostate cancer: results from 2 prospective cohorts. Int J Radiat Oncol Biol Phys 95:680–689
Zapatero A, Guerrero A, Maldonado X, Alvarez A, Gonzalez San Segundo C, Cabeza Rodriguez MA (2015) High-dose radiotherapy with short-term or long-term androgen deprivation in localised prostate cancer (DART01/05 GICOR): a randomised, controlled, phase 3 trial. Lancet Oncol 16:320–327
Waldstein C, Dörr W, Pötter R, Widder J, Goldner G (2017) Postoperative radiotherapy for prostate cancer: morbidity of local-only or local-plus-pelvic radiotherapy. Strahlenther Onkol 194(1):23–30. https://doi.org/10.1007/s00066-017-1215-9
Wiegel T, Bartkowiak D, Bottke D, Bronner C, Steiner U, Siegmann A et al (2014) Adjuvant radiotherapy versus wait-and-see after radical prostatectomy: 10-year follow-up of the ARO 96-02/UO AP 09/95 trial. Eur Urol 66:243–250
Budäus L, Schiffmann J, Graefen M, Huland H, Tennstedt P, Siegmann A et al (2017) Defining biochemical recurrence after radical prostatectomy and timing of early salvage radiotherapy: informing the debate. Strahlenther Onkol 193:692–699
Lohm G, Neumann K, Budach V, Wiegel T, Hoecht T, Gollrad J (2017) Salvage radiotherapy in prostate cancer patients with biochemical relapse after radical prostatectomy: prolongation of prostate-specific antigen doubling time in patients with subsequent biochemical progression. Strahlenther Onkol 194(4):325–332. https://doi.org/10.1007/s00066-017-1247-1
Gulliford SL, Foo K, Morgan RC, Aird EG, Bidmead AM, Critchley H et al (2010) Dose-volume constraints to reduce rectal side effects from prostate radiotherapy: evidence from MRC RT01 Trial ISRCTN 47772397. Int J Radiat Oncol Biol Phys 76:747–754
Marks LB, Yorke ED, Jackson A, Ten Haken RK, Constine LS, Eisbruch A et al (2010) Use of normal tissue complication probability models in the clinic. Int J Radiat Oncol Biol Phys 76:10–19
Michalski JM, Gay H, Jackson A, Tucker SL, Deasy JO (2010) Radiation dose-volume effects in radiation-induced rectal injury. Int J Radiat Oncol Biol Phys 76(3 Suppl):123–129
Casares-Magaz O, Muren LP, Moiseenko V, Petersen SE, Pettersson NJ, Høyer M et al (2017) Spatial rectal dose/volume metrics predict patient-reported gastro-intestinal symptoms after radiotherapy for prostate cancer. Acta Oncol 56:1507–1513
Kupelian PA, Reddy CA, Carlson TP, Wiloughby TR (2002) Dose/volume relationship of late rectal bleeding after external beam radiotherapy for localized prostate cancer: absolute or relative rectal volume? Cancer J 8:62–66
D’Amico AV, Whittington R, Malkowicz SB, Schultz D, Blank K, Broderick GA et al (1998) Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA 280:969–974
National Cancer Institute (2010) Common terminology criteria for adverse events v4.0. https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010-06-14_QuickReference_8.5x11.pdf. Accessed 22 Dec 2017
LENT SOMA scales for all anatomic sites. (1995) Int J Radiat Oncol Biol Phys 31:1049–1092.
Fiorino C, Sanguineti G, Cozzarini C, Foppiano F, Piazzolla A, Menegotti L et al (2003) Rectal dose-volume constraints in high-dose radiotherapy of localized prostate cancer. Int J Radiat Oncol Biol Phys 57:953–962
Fiorino C, Fellin G, Rancati T, Vavassori V, Bianchi VC, Borca VC, Girelli G et al (2008) Clinical and dosimetric predictors of late rectal syndrome after 3D-CRT for localized prostate cancer: preliminary results of a multicenter prospective study. Int J Radiat Oncol Biol Phys 70:1130–1137
Fiorino C, Cozzarini C, Vavassori V, Sanguineti G, Bianchi C, Cattaneo GM et al (2002) Relationships between DVHs and late rectal bleeding after radiotherapy for prostate cancer: analysis of a large group of patients pooled from three institutions. Radiother Oncol 64:1–12
Someya M, Hori M, Tatoeka K, Nakata K, Takagi M, Saito M et al (2015) Results and DVH analysis of late rectal bleeding in patients treated with 3D-CRT or IMRT for localized prostate cancer. J Radiat Res 56:122–127
Mirjolet C, Walker PM, Gauthier M, Dalban C, Naudy S, Mazoyer F et al (2016) Absolute volume of the rectum and AUC from rectal DVH between 25 and 50 Gy predict acute gastrointestinal toxicity with IG-IMRT in prostate cancer. Radiat Oncol 11:145
Nuyttens JJ, Milito S, Rust PF, Turrisi AT 3rd (2002) Dose-volume relationship for acute side effects during high dose conformal radiotherapy for prostate cancer. Radiother Oncol 64:209–214
Huang EH, Pollak A, Levy L, Starkschall G, Dong L, Rosen I et al (2002) Late rectal toxicity: dose-volume effects of conformal radiotherapy for prostate cancer. Int J Radiat Oncol Biol Phys 54:1314–1321
McDonald AM, Baker CB, Popple RA, Shekar K, Yang ES, Jacob R et al (2014) Different rectal toxicity tolerance with and without simultaneous conventionally-fractionated pelvic lymph node treatment in patients receiving hypofractionated prostate radiotherapy. Radiat Oncol. https://doi.org/10.1186/1748-717X-9-129
Guckenberger M, Andratschke N, Alheit N, Holy R, Moustakis C, Nestle U et al (2014) Definition of stereotactic body radiotherapy: principles and practice for the treatment of stage I non-small cell lung cancer. Strahlenther Onkol 190:26–33
Kocher M, Wittig A, Piroth MD, Treuer H, Seegenschmiedt H, Ruge M et al (2014) Stereotactic radiosurgery for treatment of brain metastases. A report of the DEGRO Working Group on Stereotactic Radiotherapy. Strahlenther Onkol 190:521–531
Shu HK, Lee TT, Vigneauly E, Xia P, Pickett B, Phillips TL et al (2001) Toxicity following high-dose three-dimensional conformal and intensity-modulated radiation therapy for clinically localized prostate cancer. Urology 57:102–107
Chan LW, Xia P, Gottschalk AR, Akazawa M, Scala M, Picket B et al (2008) Proposed rectal dose constraints for patients undergoing definitive whole pelvic radiotherapy for clinically localized prostate cancer. Int J Radiat Oncol Biol Phys 72:69–77
Zelefsky MJ, Levin EJ, Hunt M, Yamada Y, Shippy AM, Jackson A et al (2008) Incidence of late rectal and urinary toxicities after three-dimensional conformal radiotherapy and intensity-modulated radiotherapy for localized prostate cancer. Int J Radiat Oncol Biol Phys 70:1124–1129
Wortel RC, Incrocci L, Pos FJ, Lebesque JV, Witte MG, van der Heide UA et al (2015) Acute toxicity after image-guided intensity modulated radiation therapy compared to 3D conformal radiation therapy in prostate cancer patients. Int J Radiat Oncol Biol Phys 91:737–744
Tucker SL, Dong L, Cheung R, Johnson J, Mohan R, Huang EH et al (2004) Comparison of rectal dose-wall histogram versus dose-volume histogram for modeling the incidence of late rectal bleeding after radiotherapy. Int J Radiat Oncol Biol Phys 60:1589–1601
Guckenberger M, Pohl F, Baier K, Meyer J, Koebl O, Flentje M et al (2006) Influence of rectum delineation (rectal volume vs. rectal wall) on IMRT treatment planning of the prostate. Strahlenther Onkol 182:721–726
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
R. Paleny, M. Bremer, D. Walacides, S. Mainwaring, K. Weber and C. Henkenberens declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Paleny, R., Bremer, M., Walacides, D. et al. Comparison of relative and absolute rectal dose–volume parameters and clinical correlation with acute and late radiation proctitis in prostate cancer patients. Strahlenther Onkol 195, 103–112 (2019). https://doi.org/10.1007/s00066-018-1365-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-018-1365-4