Abstract
The concept of renoprotection has evolved significantly, driven by improved understanding of the pathophysiology of chronic kidney disease (CKD) and the advent of novel treatment options. Glomerular hyperfiltration, hypertension and proteinuria represent key mediators of CKD progression. It is increasingly recognized that proteinuria may actually be pathological and etiological in CKD progression and not just symptomatic. It initiates a sequence of events involving activation of proinflammatory and profibrotic signaling pathways in proximal tubular epithelial cells with transmission of the disease to the tubulointerstitium and progression to end-stage kidney disease (ESKD). Although the etiology and epidemiology of pediatric CKD differs to that in adults, studies in the various animal models of kidney disease, from obstructive uropathy to glomerulonephritis, have revealed that many common proinflammatory and profibrotic pathways are induced in progressive proteinuric CKD, irrespective of the primary disease. This pathomechanistic overlap therefore translates into the potential for common treatment targets for a wide spectrum of kidney diseases. In this review we therefore discuss the experimental and clinical evidence for an array of prospective future drug treatments of CKD progression. While conceptually promising, clear definitive evidence beyond preclinical data does not exist for many of these treatments, and others are limited by serious adverse effects. More studies are needed before general recommendations can be given.
Similar content being viewed by others
Abbreviations
- ACE:
-
Angiotensin converting enzyme
- ANP:
-
Atrial natriuretic peptide
- ARB:
-
Angiotensin receptor I blocker
- BMI:
-
Body mass index
- CAKUT:
-
Congenital anomalies of the kidney and urinary tract
- CCL 2:
-
Chemokine (C-C motif) ligand 2 (MCP 1)
- CCL 5/RANTES:
-
Chemokine (C-C motif) ligand 5
- CCR:
-
β chemokine receptor
- CKD:
-
Chronic kidney disease
- CKiD:
-
Chronic Kidney Disease in Children cohort
- CTGF:
-
Connective tissue growth factor
- DN:
-
Diabetic nephropathy
- ECM:
-
Extracellular matrix
- ESKD:
-
End-stage kidney disease
- ET:
-
Endothelin
- FSGS:
-
Focal segmental sclerosis
- GBM:
-
Glomerular basement membrane
- GFR:
-
Glomerular filtration rate
- HMG CoA:
-
3-Hydroxy-3-Methylglutaryl CoA
- IF:
-
Interstitial fibrosis
- KDOQI:
-
Kidney Disease Outcomes Quality Initiative
- MCP 1:
-
Monocyte chemotactic protein 1 (CCL 2)
- MMP:
-
Matrix metalloproteinase
- MSC:
-
Mesenchymal stromal cell
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- NEP:
-
Neutral endopeptidase
- NO:
-
Nitric oxide
- NOS:
-
Nitric oxide synthase
- PAN:
-
Puromycin aminonucleoside
- PPAR γ:
-
Peroxisome-proliferator-activated-receptor γ
- PTEC:
-
Peritubular epithelial cell
- RAAS:
-
Renin angiotensin aldosterone system
- RANTES:
-
Regulated upon activation normal T-cell expressed, and secreted (CCL 5)
- ROS:
-
Reactive oxygen species
- TGF β:
-
Transforming growth factor β
- TNF α:
-
Tumor necrosis factor α
- VDR:
-
Vitamin D receptor
- VEGF:
-
Vascular endothelial growth factor
References
Taal MW, Brenner BM (2000) Renoprotective benefits of RAS inhibition: from ACEI to angiotensin II antagonists. Kidney Int 57:1803–1817
Khan S, Amedia CA Jr (2008) Economic burden of chronic kidney disease. J Eval Clin Pract 14:422–434
Nichols GA, Vupputuri S, Lau H (2011) Medical care costs associated with progression of diabetic nephropathy. Diabetes Care 34:2374–2378
Brenner BM, Hostetter TH, Humes HD (1978) Molecular basis of proteinuria of glomerular origin. N Engl J Med 298:826–833
Brenner BM, Meyer TW, Hostetter TH (1982) Dietary protein intake and the progressive nature of kidney disease: the role of hemodynamically mediated glomerular injury in the pathogenesis of progressive glomerular sclerosis in aging, renal ablation, and intrinsic renal disease. N Engl J Med 307:652–659
Hostetter TH, Olson JL, Rennke HG, Venkatachalam MA, Brenner BM (1981) Hyperfiltration in remnant nephrons: a potentially adverse response to renal ablation. Am J Physiol 241:F85–F93
Klag MJ, Whelton PK, Randall BL, Neaton JD, Brancati FL, Ford CE, Shulman NB, Stamler J (1996) Blood pressure and end-stage renal disease in men. N Engl J Med 334:13–18
Ihle BU, Becker GJ, Whitworth JA, Charlwood RA, Kincaid-Smith PS (1989) The effect of protein restriction on the progression of renal insufficiency. N Engl J Med 321:1773–1777
Locatelli F, Alberti D, Graziani G, Buccianti G, Redaelli B, Giangrande A (1991) Prospective, randomised, multicentre trial of effect of protein restriction on progression of chronic renal insufficiency. Northern Italian Cooperative Study Group. Lancet 337:1299–1304
Klahr S, Levey AS, Beck GJ, Caggiula AW, Hunsicker L, Kusek JW, Striker G (1994) The effects of dietary protein restriction and blood-pressure control on the progression of chronic renal disease. Modification of diet in Renal Disease Study Group. N Engl J Med 330:877–884
Ruggenenti P, Cravedi P, Remuzzi G (2012) Mechanisms and treatment of CKD. J Am Soc Nephrol 23:1917–1928
Abbate M, Zoja C, Remuzzi G (2006) How does proteinuria cause progressive renal damage? J Am Soc Nephrol 17:2974–2984
Baines RJ, Brunskill NJ (2011) Tubular toxicity of proteinuria. Nat Rev Nephrol 7:177–180
Liu Y (2011) Cellular and molecular mechanisms of renal fibrosis. Nat Rev Nephrol 7:684–696
Gorriz JL, Martinez-Castelao A (2011) Proteinuria: detection and role in native renal disease progression. Transplant Rev (Orlando) 26:3–13
Hodgkins KS, Schnaper HW (2011) Tubulointerstitial injury and the progression of chronic kidney disease. Pediatr Nephrol 27:901–909
Garg P, Rabelink T (2011) Glomerular proteinuria: a complex interplay between unique players. Adv Chronic Kidney Dis 18:233–242
[No authors listed] (1997) Randomised placebo-controlled trial of effect of ramipril on decline in glomerular filtration rate and risk of terminal renal failure in proteinuric, non-diabetic nephropathy. The GISEN Group (Gruppo Italiano di Studi Epidemiologici in Nefrologia). Lancet 349:1857–1863
Brenner BM, Cooper ME, de Zeeuw D, Keane WF, Mitch WE, Parving HH, Remuzzi G, Snapinn SM, Zhang Z, Shahinfar S (2001) Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med 345:861–869
Van der Velde M, Matsushita K, Coresh J, Astor BC, Woodward M, Levey A, de Jong P, Gansevoort RT, Levey AS, de Jong PE, El-Nahas M, Eckardt KU, Kasiske BL, Ninomiya T, Chalmers J, Macmahon S, Tonelli M, Hemmelgarn B, Sacks F, Curhan G, Collins AJ, Li S, Chen SC, Hawaii Cohort KP, Lee BJ, Ishani A, Neaton J, Svendsen K, Mann JF, Yusuf S, Teo KK, Gao P, Nelson RG, Knowler WC, Bilo HJ, Joosten H, Kleefstra N, Groenier KH, Auguste P, Veldhuis K, Wang Y, Camarata L, Thomas B, Manley T (2011) Lower estimated glomerular filtration rate and higher albuminuria are associated with all-cause and cardiovascular mortality. A collaborative meta-analysis of high-risk population cohorts. Kidney Int 79:1341–1352
Van der Velde M, Halbesma N, de Charro FT, Bakker SJ, de Zeeuw D, de Jong PE, Gansevoort RT (2009) Screening for albuminuria identifies individuals at increased renal risk. J Am Soc Nephrol 20:852–862
Halbesma N, Kuiken DS, Brantsma AH, Bakker SJ, Wetzels JF, De Zeeuw D, De Jong PE, Gansevoort RT (2006) Macroalbuminuria is a better risk marker than low estimated GFR to identify individuals at risk for accelerated GFR loss in population screening. J Am Soc Nephrol 17:2582–2590
Wong H, Mylrea K, Feber J, Drukker A, Filler G (2006) Prevalence of complications in children with chronic kidney disease according to KDOQI. Kidney Int 70:585–590
Wong CS, Pierce CB, Cole SR, Warady BA, Mak RH, Benador NM, Kaskel F, Furth SL, Schwartz GJ (2009) Association of proteinuria with race, cause of chronic kidney disease, and glomerular filtration rate in the chronic kidney disease in children study. Clin J Am Soc Nephrol 4:812–819
Ardissino G, Testa S, Dacco V, Vigano S, Taioli E, Claris-Appiani A, Procaccio M, Avolio L, Ciofani A, Dello Strologo L, Montini G (2004) Proteinuria as a predictor of disease progression in children with hypodysplastic nephropathy. Data from the Ital Kid Project. Pediatr Nephrol 19:172–177
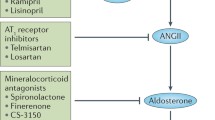
Berl T (2009) Review: renal protection by inhibition of the renin-angiotensin-aldosterone system. J Renin Angiotensin Aldosterone Syst 10:1–8
Gross O, Beirowski B, Koepke ML, Kuck J, Reiner M, Addicks K, Smyth N, Schulze-Lohoff E, Weber M (2003) Preemptive ramipril therapy delays renal failure and reduces renal fibrosis in COL4A3-knockout mice with Alport syndrome. Kidney Int 63:438–446
Gross O, Schulze-Lohoff E, Koepke ML, Beirowski B, Addicks K, Bloch W, Smyth N, Weber M (2004) Antifibrotic, nephroprotective potential of ACE inhibitor vs AT1 antagonist in a murine model of renal fibrosis. Nephrol Dial Transplant 19:1716–1723
Wuhl E, Trivelli A, Picca S, Litwin M, Peco-Antic A, Zurowska A, Testa S, Jankauskiene A, Emre S, Caldas-Afonso A, Anarat A, Niaudet P, Mir S, Bakkaloglu A, Enke B, Montini G, Wingen AM, Sallay P, Jeck N, Berg U, Caliskan S, Wygoda S, Hohbach-Hohenfellner K, Dusek J, Urasinski T, Arbeiter K, Neuhaus T, Gellermann J, Drozdz D, Fischbach M, Moller K, Wigger M, Peruzzi L, Mehls O, Schaefer F (2009) Strict blood-pressure control and progression of renal failure in children. N Engl J Med 361:1639–1650
Webb NJ, Lam C, Loeys T, Shahinfar S, Strehlau J, Wells TG, Santoro E, Manas D, Gleim GW (2010) Randomized, double-blind, controlled study of losartan in children with proteinuria. Clin J Am Soc Nephrol 5:417–424
Webb NJ, Shahinfar S, Wells TG, Massaad R, Gleim GW, Santoro EP, Sisk CM, Lam C (2012) Losartan and enalapril are comparable in reducing proteinuria in children. Kidney Int 82:819–826
Gross O, Licht C, Anders HJ, Hoppe B, Beck B, Tonshoff B, Hocker B, Wygoda S, Ehrich JH, Pape L, Konrad M, Rascher W, Dotsch J, Muller-Wiefel DE, Hoyer P, Knebelmann B, Pirson Y, Grunfeld JP, Niaudet P, Cochat P, Heidet L, Lebbah S, Torra R, Friede T, Lange K, Muller GA, Weber M (2012) Early angiotensin-converting enzyme inhibition in Alport syndrome delays renal failure and improves life expectancy. Kidney Int 81:494–501
Temme J, Kramer A, Jager KJ, Lange K, Peters F, Muller GA, Kramar R, Heaf JG, Finne P, Palsson R, Reisaeter AV, Hoitsma AJ, Metcalfe W, Postorino M, Zurriaga O, Santos JP, Ravani P, Jarraya F, Verrina E, Dekker FW, Gross O (2012) Outcomes of male patients with Alport syndrome undergoing renal replacement therapy. Clin J Am Soc Nephrol 7:1969–1976
Kim HJ, Ryu JH, Han SW, Park IK, Paik SS, Park MH, Paik DJ, Chung HS, Kim SW, Lee JU (2004) Combined therapy of cilazapril and losartan has no additive effects in ameliorating adriamycin-induced glomerulopathy. Nephron Physiol 97:p58–p65
Catapano F, Chiodini P, De Nicola L, Minutolo R, Zamboli P, Gallo C, Conte G (2008) Antiproteinuric response to dual blockade of the renin-angiotensin system in primary glomerulonephritis: meta-analysis and metaregression. Am J Kidney Dis 52:475–485
Bakris GL (2010) Dual RAAS blockade is desirable in kidney disease: con. Kidney Int 78:546–549
Ruggenenti P, Perticucci E, Cravedi P, Gambara V, Costantini M, Sharma SK, Perna A, Remuzzi G (2008) Role of remission clinics in the longitudinal treatment of CKD. J Am Soc Nephrol 19:1213–1224
Conn JW (1955) Presidential address. I. Painting background. II. Primary aldosteronism, a new clinical syndrome. J Lab Clin Med 45:3–17
Conn JW, Knopf RF, Nesbit RM (1964) Clinical characteristics of primary aldosteronism from an analysis 145 cases. Am J Surg 107:159–172
Greene EL, Kren S, Hostetter TH (1996) Role of aldosterone in the remnant kidney model in the rat. J Clin Invest 98:1063–1068
Nishiyama A, Yao L, Fan Y, Kyaw M, Kataoka N, Hashimoto K, Nagai Y, Nakamura E, Yoshizumi M, Shokoji T, Kimura S, Kiyomoto H, Tsujioka K, Kohno M, Tamaki T, Kajiya F, Abe Y (2005) Involvement of aldosterone and mineralocorticoid receptors in rat mesangial cell proliferation and deformability. Hypertension 45:710–716
Nagase M, Fujita T (2008) Aldosterone and glomerular podocyte injury. Clin Exp Nephrol 12:233–242
Ishizawa K, Izawa Y, Ito H, Miki C, Miyata K, Fujita Y, Kanematsu Y, Tsuchiya K, Tamaki T, Nishiyama A, Yoshizumi M (2005) Aldosterone stimulates vascular smooth muscle cell proliferation via big mitogen-activated protein kinase 1 activation. Hypertension 46:1046–1052
Endemann DH, Touyz RM, Iglarz M, Savoia C, Schiffrin EL (2004) Eplerenone prevents salt-induced vascular remodeling and cardiac fibrosis in stroke-prone spontaneously hypertensive rats. Hypertension 43:1252–1257
Miyata K, Rahman M, Shokoji T, Nagai Y, Zhang GX, Sun GP, Kimura S, Yukimura T, Kiyomoto H, Kohno M, Abe Y, Nishiyama A (2005) Aldosterone stimulates reactive oxygen species production through activation of NADPH oxidase in rat mesangial cells. J Am Soc Nephrol 16:2906–2912
Shibata S, Nagase M, Yoshida S, Kawachi H, Fujita T (2007) Podocyte as the target for aldosterone: roles of oxidative stress and Sgk1. Hypertension 49:355–364
Nagai Y, Miyata K, Sun GP, Rahman M, Kimura S, Miyatake A, Kiyomoto H, Kohno M, Abe Y, Yoshizumi M, Nishiyama A (2005) Aldosterone stimulates collagen gene expression and synthesis via activation of ERK1/2 in rat renal fibroblasts. Hypertension 46:1039–1045
Chun TY, Chander PN, Kim JW, Pratt JH, Stier CT Jr (2008) Aldosterone, but not angiotensin II, increases profibrotic factors in kidney of adrenalectomized stroke-prone spontaneously hypertensive rats. Am J Physiol Endocrinol Metab 295:E305–E312
Sun GP, Kohno M, Guo P, Nagai Y, Miyata K, Fan YY, Kimura S, Kiyomoto H, Ohmori K, Li DT, Abe Y, Nishiyama A (2006) Involvements of Rho-kinase and TGF-beta pathways in aldosterone-induced renal injury. J Am Soc Nephrol 17:2193–2201
Nagase M, Shibata S, Yoshida S, Nagase T, Gotoda T, Fujita T (2006) Podocyte injury underlies the glomerulopathy of Dahl salt-hypertensive rats and is reversed by aldosterone blocker. Hypertension 47:1084–1093
Aldigier JC, Kanjanbuch T, Ma LJ, Brown NJ, Fogo AB (2005) Regression of existing glomerulosclerosis by inhibition of aldosterone. J Am Soc Nephrol 16:3306–3314
Nakhoul F, Khankin E, Yaccob A, Kawachi H, Karram T, Awaad H, Nakhoul N, Hoffman A, Abassi Z (2008) Eplerenone potentiates the antiproteinuric effects of enalapril in experimental nephrotic syndrome. Am J Physiol Renal Physiol 294:F628–F637
Taira M, Toba H, Murakami M, Iga I, Serizawa R, Murata S, Kobara M, Nakata T (2008) Spironolactone exhibits direct renoprotective effects and inhibits renal renin-angiotensin-aldosterone system in diabetic rats. Eur J Pharmacol 589:264–271
Fujisawa G, Okada K, Muto S, Fujita N, Itabashi N, Kusano E, Ishibashi S (2004) Spironolactone prevents early renal injury in streptozotocin-induced diabetic rats. Kidney Int 66:1493–1502
Yuan J, Jia R, Bao Y (2007) Beneficial effects of spironolactone on glomerular injury in streptozotocin-induced diabetic rats. J Renin Angiotensin Aldosterone Syst 8:118–126
Ubaid-Girioli S, Ferreira-Melo SE, Souza LA, Nogueira EA, Yugar-Toledo JC, Coca A, Moreno H Jr (2007) Aldosterone escape with diuretic or angiotensin-converting enzyme inhibitor/angiotensin II receptor blocker combination therapy in patients with mild to moderate hypertension. J Clin Hypertens (Greenwich) 9:770–774
Sato A, Hayashi K, Naruse M, Saruta T (2003) Effectiveness of aldosterone blockade in patients with diabetic nephropathy. Hypertension 41:64–68
Sato A, Hayashi K, Saruta T (2005) Antiproteinuric effects of mineralocorticoid receptor blockade in patients with chronic renal disease. Am J Hypertens 18:44–49
Bianchi S, Bigazzi R, Campese VM (2006) Long-term effects of spironolactone on proteinuria and kidney function in patients with chronic kidney disease. Kidney Int 70:2116–2123
Bomback AS, Kshirsagar AV, Amamoo MA, Klemmer PJ (2008) Change in proteinuria after adding aldosterone blockers to ACE inhibitors or angiotensin receptor blockers in CKD: a systematic review. Am J Kidney Dis 51:199–211
Navaneethan SD, Nigwekar SU, Sehgal AR, Strippoli GF (2009) Aldosterone antagonists for preventing the progression of chronic kidney disease: a systematic review and meta-analysis. Clin J Am Soc Nephrol 4:542–551
Kaito H, Nozu K, Iijima K, Nakanishi K, Yoshiya K, Kanda K, Przybyslaw Krol R, Yoshikawa N, Matsuo M (2006) The effect of aldosterone blockade in patients with Alport syndrome. Pediatr Nephrol 21:1824–1829
Buczko W, Hermanowicz JM (2008) Pharmacokinetics and pharmacodynamics of aliskiren, an oral direct renin inhibitor. Pharmacol Rep 60:623–631
Choi DE, Jeong JY, Lim BJ, Chang YK, Na KR, Shin YT, Lee KW (2011) Aliskiren ameliorates renal inflammation and fibrosis induced by unilateral ureteral obstruction in mice. J Urol 186:694–701
Gross O, Girgert R, Rubel D, Temme J, Theissen S, Muller GA (2011) Renal protective effects of aliskiren beyond its antihypertensive property in a mouse model of progressive fibrosis. Am J Hypertens 24:355–361
Moriyama T, Tsuruta Y, Kojima C, Itabashi M, Sugiura H, Takei T, Ogawa T, Uchida K, Tsuchiya K, Nitta K (2011) Beneficial effect of aliskiren combined with olmesartan in reducing urinary protein excretion in patients with chronic kidney disease. Int Urol Nephrol 44:841–845
Nakamura T, Sato E, Amaha M, Kawagoe Y, Maeda S, Yamagishi SI (2011) Addition of aliskiren to olmesartan ameliorates tubular injury in chronic kidney disease patients partly by reducing proteinuria. J Renin Angiotensin Aldosterone Syst 40:798–803
Tang SC, Lin M, Tam S, Au WS, Ma MK, Yap DY, Ho YW, Lai KN (2010) Aliskiren combined with losartan in immunoglobulin A nephropathy: an open-labeled pilot study. Nephrol Dial Transplant 27:613–618
Kelland EE, McAuley LM, Filler G (2011) Are we ready to use aliskiren in children? Pediatr Nephrol 26:473–477
Flynn JT (2011) Not ready for prime time: aliskiren for treatment of hypertension or proteinuria in children. Pediatr Nephrol 26:491–492
Parving HH, Brenner BM, McMurray JJ, de Zeeuw D, Haffner SM, Solomon SD, Chaturvedi N, Persson F, Nicolaides M, Richards A, Xiang Z, Armbrecht J, Pfeffer MA; ALTITUDE Investigators (2012) Baseline characteristics in the Aliskiren trial in type 2 diabetes using cardio-renal endpoints (ALTITUDE). J Renin Angiotensin Aldosterone Syst 12:387–393
McMurray JJ, Abraham WT, Dickstein K, Kober L, Massie BM, Krum H (2012) Aliskiren, ALTITUDE, and the implications for ATMOSPHERE. Eur J Heart Fail 14:341–343
Harel Z, Gilbert C, Wald R, Bell C, Perl J, Juurlink D, Beyene J, Shah PS (2012) The effect of combination treatment with aliskiren and blockers of the renin-angiotensin system on hyperkalaemia and acute kidney injury: systematic review and meta-analysis. BMJ 344:e42
Weber MA (2001) Vasopeptidase inhibitors. Lancet 358:1525–1532
Sagnella GA (2002) Vasopeptidase inhibitors. J Renin Angiotensin Aldosterone Syst 3:90–95
Pawluczyk IZ, Patel SR, Harris KP (2006) Pharmacological enhancement of the kallikrein-kinin system promotes anti-fibrotic responses in human mesangial cells. Cell Physiol Biochem 18:327–336
Benigni A, Zoja C, Zatelli C, Corna D, Longaretti L, Rottoli D, Maggioni P, Todeschini M, Noris M, Remuzzi G (2004) Vasopeptidase inhibitor restores the balance of vasoactive hormones in progressive nephropathy. Kidney Int 66:1959–1965
Gross O, Koepke ML, Beirowski B, Schulze-Lohoff E, Segerer S, Weber M (2005) Nephroprotection by antifibrotic and anti-inflammatory effects of the vasopeptidase inhibitor AVE7688. Kidney Int 68:456–463
Dhaun N, Goddard J, Webb DJ (2006) The endothelin system and its antagonism in chronic kidney disease. J Am Soc Nephrol 17:943–955
Barton M (2010) Therapeutic potential of endothelin receptor antagonists for chronic proteinuric renal disease in humans. Biochim Biophys Acta 1802:1203–1213
Hocher B, Thone-Reineke C, Rohmeiss P, Schmager F, Slowinski T, Burst V, Siegmund F, Quertermous T, Bauer C, Neumayer HH, Schleuning WD, Theuring F (1997) Endothelin-1 transgenic mice develop glomerulosclerosis, interstitial fibrosis, and renal cysts but not hypertension. J Clin Invest 99:1380–1389
Ortmann J, Amann K, Brandes RP, Kretzler M, Munter K, Parekh N, Traupe T, Lange M, Lattmann T, Barton M (2004) Role of podocytes for reversal of glomerulosclerosis and proteinuria in the aging kidney after endothelin inhibition. Hypertension 44:974–981
Gagliardini E, Corna D, Zoja C, Sangalli F, Carrara F, Rossi M, Conti S, Rottoli D, Longaretti L, Remuzzi A, Remuzzi G, Benigni A (2009) Unlike each drug alone, lisinopril if combined with avosentan promotes regression of renal lesions in experimental diabetes. Am J Physiol Renal Physiol 297:F1448–F1456
Dhaun N, Macintyre IM, Melville V, Lilitkarntakul P, Johnston NR, Goddard J, Webb DJ (2009) Blood pressure-independent reduction in proteinuria and arterial stiffness after acute endothelin-a receptor antagonism in chronic kidney disease. Hypertension 54:113–119
Lavelle A, Sugrue R, Lawler G, Mulligan N, Kelleher B, Murphy DM, Gaine SP (2009) Sitaxentan-induced hepatic failure in two patients with pulmonary arterial hypertension. Eur Respir J 34:770–771
Wenzel RR, Littke T, Kuranoff S, Jurgens C, Bruck H, Ritz E, Philipp T, Mitchell A (2009) Avosentan reduces albumin excretion in diabetics with macroalbuminuria. J Am Soc Nephrol 20:655–664
Mann JF, Green D, Jamerson K, Ruilope LM, Kuranoff SJ, Littke T, Viberti G (2010) Avosentan for overt diabetic nephropathy. J Am Soc Nephrol 21:527–535
Ritz E, Wenzel R (2010) Endothelin receptor antagonists in proteinuric renal disease: every rose has its thorn. J Am Soc Nephrol 21:392–394
Kohan DE, Pritchett Y, Molitch M, Wen S, Garimella T, Audhya P, Andress DL (2011) Addition of atrasentan to renin-angiotensin system blockade reduces albuminuria in diabetic nephropathy. J Am Soc Nephrol 22:763–772
Weiss J, Haefeli WE (2011) Interaction potential of the endothelin-A receptor antagonist atrasentan with drug transporters and drug-metabolising enzymes assessed in vitro. Cancer Chemother Pharmacol 68:1093–1098
Fogo AB (2011) PPARgamma and chronic kidney disease. Pediatr Nephrol 26:347–351
Yang HC, Ma LJ, Ma J, Fogo AB (2006) Peroxisome proliferator-activated receptor-gamma agonist is protective in podocyte injury-associated sclerosis. Kidney Int 69:1756–1764
Tolman KG (2011) The safety of thiazolidinediones. Expert Opin Drug Saf 10:419–428
Benigni A, Zoja C, Tomasoni S, Campana M, Corna D, Zanchi C, Gagliardini E, Garofano E, Rottoli D, Ito T, Remuzzi G (2006) Transcriptional regulation of nephrin gene by peroxisome proliferator-activated receptor-gamma agonist: molecular mechanism of the antiproteinuric effect of pioglitazone. J Am Soc Nephrol 17:1624–1632
Kanjanabuch T, Ma LJ, Chen J, Pozzi A, Guan Y, Mundel P, Fogo AB (2007) PPAR-gamma agonist protects podocytes from injury. Kidney Int 71:1232–1239
Miyazaki Y, Cersosimo E, Triplitt C, DeFronzo RA (2007) Rosiglitazone decreases albuminuria in type 2 diabetic patients. Kidney Int 72:1367–1373
Katavetin P, Eiam-Ong S, Suwanwalaikorn S (2006) Pioglitazone reduces urinary protein and urinary transforming growth factor-beta excretion in patients with type 2 diabetes and overt nephropathy. J Med Assoc Thai 89:170–177
Kincaid-Smith P, Fairley KF, Farish S, Best JD, Proietto J (2008) Reduction of proteinuria by rosiglitazone in non-diabetic renal disease. Nephrology (Carlton) 13:58–62
Zanchi A, Pechere-Bertschi A, Burnier M, Bonny O (2011) Effects of pioglitazone on renal calcium excretion. J Clin Endocrinol Metab 96:E1482–E1485
Kusunoki H, Taniyama Y, Azuma J, Iekushi K, Sanada F, Otsu R, Iwabayashi M, Okayama K, Rakugi H, Morishita R (2012) Telmisartan exerts renoprotective actions via peroxisome proliferator-activated receptor-gamma/hepatocyte growth factor pathway independent of angiotensin II type 1 receptor blockade. Hypertension 59:308–316
Fujita H, Sakamoto T, Komatsu K, Fujishima H, Morii T, Narita T, Takahashi T, Yamada Y (2011) Reduction of circulating superoxide dismutase activity in type 2 diabetic patients with microalbuminuria and its modulation by telmisartan therapy. Hypertens Res 34:1302–1308
Nakamura T, Sato E, Fujiwara N, Kawagoe Y, Yamada S, Ueda Y, Koide H (2012) Changes in urinary albumin excretion, inflammatory and oxidative stress markers in ADPKD patients with hypertension. Am J Med Sci 343:46–51
Zhang Q, Xiao X, Li M, Li W, Yu M, Zhang H, Sun X, Mao L, Xiang H (2012) Telmisartan improves kidney function through inhibiting oxidative phosphorylation pathway in diabetic rats. J Mol Endocrinol 49:35–46
Destro M, Cagnoni F, Dognini GP, Galimberti V, Taietti C, Cavalleri C, Galli E (2011) Telmisartan: just an antihypertensive agent? A literature review. Expert Opin Pharmacother 12:2719–2735
Toba H, Tojo C, Wang J, Noda K, Kobara M, Nakata T (2012) Telmisartan inhibits vascular dysfunction and inflammation via activation of peroxisome proliferator-activated receptor-gamma in subtotal nephrectomized rat. Eur J Pharmacol 685:91–98
Takagi H, Yamamoto H, Iwata K, Goto SN, Umemoto T (2012) Effects of telmisartan on proteinuria or albuminuria: a meta-analysis of randomized trials. Int J Cardiol 159:69–72
Harenberg J (1998) Review of pharmacodynamics, pharmacokinetics, and therapeutic properties of sulodexide. Med Res Rev 18:1–20
Van den Hoven MJ, Rops AL, Vlodavsky I, Levidiotis V, Berden JH, van der Vlag J (2007) Heparanase in glomerular diseases. Kidney Int 72:543–548
Holt RC, Webb NJ, Ralph S, Davies J, Short CD, Brenchley PE (2005) Heparanase activity is dysregulated in children with steroid-sensitive nephrotic syndrome. Kidney Int 67:122–129
Miner JH (2008) Glomerular filtration: the charge debate charges ahead. Kidney Int 74:259–261
Ciszewicz M, Polubinska A, Antoniewicz A, Suminska-Jasinska K, Breborowicz A (2009) Sulodexide suppresses inflammation in human endothelial cells and prevents glucose cytotoxicity. Transl Res 153:118–123
Bang K, Chin HJ, Chae DW, Joo KW, Kim YS, Kim S, Ju KD, Kim H, Ahn C, Oh KH (2010) Anti-proteinuric effect of sulodexide in immunoglobulin a nephropathy. Yonsei Med J 52:588–594
Gaddi AV, Cicero AF, Gambaro G (2010) Nephroprotective action of glycosaminoglycans: why the pharmacological properties of sulodexide might be reconsidered. Int J Nephrol Renovasc Dis 3:99–105
Lewis EJ, Lewis JB, Greene T, Hunsicker LG, Berl T, Pohl MA, de Zeeuw D, Heerspink HL, Rohde RD, Atkins RC, Reutens AT, Packham DK, Raz I (2011) Sulodexide for kidney protection in type 2 diabetes patients with microalbuminuria: a randomized controlled trial. Am J Kidney Dis 58:729–736
House AA, Weir MA (2011) Sulodexide for diabetic nephropathy: another one bites the dust. Am J Kidney Dis 58:692–694
Packham DK, Wolfe R, Reutens AT, Berl T, Heerspink HL, Rohde R, Ivory S, Lewis J, Raz I, Wiegmann TB, Chan JC, de Zeeuw D, Lewis EJ, Atkins RC (2012) Sulodexide fails to demonstrate renoprotection in overt type 2 diabetic nephropathy. J Am Soc Nephrol 23:123–130
Rossini M, Naito T, Yang H, Freeman M, Donnert E, Ma LJ, Dunn SR, Sharma K, Fogo AB (2010) Sulodexide ameliorates early but not late kidney disease in models of radiation nephropathy and diabetic nephropathy. Nephrol Dial Transplant 25:1803–1810
Lauver DA, Booth EA, White AJ, Poradosu E, Lucchesi BR (2005) Sulodexide attenuates myocardial ischemia/reperfusion injury and the deposition of C-reactive protein in areas of infarction without affecting hemostasis. J Pharmacol Exp Ther 312:794–800
Smith RJ, Alexander J, Barlow PN, Botto M, Cassavant TL, Cook HT, de Cordoba SR, Hageman GS, Jokiranta TS, Kimberling WJ, Lambris JD, Lanning LD, Levidiotis V, Licht C, Lutz HU, Meri S, Pickering MC, Quigg RJ, Rops AL, Salant DJ, Sethi S, Thurman JM, Tully HF, Tully SP, van der Vlag J, Walker PD, Wurzner R, Zipfel PF (2007) New approaches to the treatment of dense deposit disease. J Am Soc Nephrol 18:2447–2456
Chung AC, Lan HY (2011) Chemokines in renal injury. J Am Soc Nephrol 22:802–809
Sayyed SG, Ryu M, Kulkarni OP, Schmid H, Lichtnekert J, Gruner S, Green L, Mattei P, Hartmann G, Anders HJ (2011) An orally active chemokine receptor CCR2 antagonist prevents glomerulosclerosis and renal failure in type 2 diabetes. Kidney Int 80:68–78
Awad AS, Kinsey GR, Khutsishvili K, Gao T, Bolton WK, Okusa MD (2011) Monocyte/macrophage chemokine receptor CCR2 mediates diabetic renal injury. Am J Physiol Renal Physiol 301:F1358–F1366
Urushihara M, Ohashi N, Miyata K, Satou R, Acres OW, Kobori H (2011) Addition of angiotensin II type 1 receptor blocker to CCR2 antagonist markedly attenuates crescentic glomerulonephritis. Hypertension 57:586–593
Chen YM, Lin SL, Chiang WC, Wu KD, Tsai TJ (2006) Pentoxifylline ameliorates proteinuria through suppression of renal monocyte chemoattractant protein-1 in patients with proteinuric primary glomerular diseases. Kidney Int 69:1410–1415
Navarro-Gonzalez JF, Muros M, Mora-Fernandez C, Herrera H, Meneses B, Garcia J (2011) Pentoxifylline for renoprotection in diabetic nephropathy: the PREDIAN study. Rationale and basal results. J Diabetes Complications 25:314–319
Ng YY, Chen YM, Tsai TJ, Lan XR, Yang WC, Lan HY (2009) Pentoxifylline inhibits transforming growth factor-beta signaling and renal fibrosis in experimental crescentic glomerulonephritis in rats. Am J Nephrol 29:43–53
Zhou QG, Zheng FL, Hou FF (2009) Inhibition of tubulointerstitial fibrosis by pentoxifylline is associated with improvement of vascular endothelial growth factor expression. Acta Pharmacol Sin 30:98–106
McCormick BB, Sydor A, Akbari A, Fergusson D, Doucette S, Knoll G (2008) The effect of pentoxifylline on proteinuria in diabetic kidney disease: a meta-analysis. Am J Kidney Dis 52:454–463
Lin SL, Chen YM, Chiang WC, Wu KD, Tsai TJ (2008) Effect of pentoxifylline in addition to losartan on proteinuria and GFR in CKD: a 12-month randomized trial. Am J Kidney Dis 52:464–474
Perkins RM, Aboudara MC, Uy AL, Olson SW, Cushner HM, Yuan CM (2009) Effect of pentoxifylline on GFR decline in CKD: a pilot, double-blind, randomized, placebo-controlled trial. Am J Kidney Dis 53:606–616
Harris E, Schulzke SM, Patole SK (2010) Pentoxifylline in preterm neonates: a systematic review. Paediatr Drugs 12:301–311
Shibata S, Nagase M, Fujita T (2006) Fluvastatin ameliorates podocyte injury in proteinuric rats via modulation of excessive Rho signaling. J Am Soc Nephrol 17:754–764
Kang CS, Chang NC, Chang ST, Lin CC, Lee TM (2009) Effect of pravastatin on nephroprotection in deoxycorticosterone acetate-salt hypertensive rats. J Hypertens 27:2232–2243
Zhou MS, Schuman IH, Jaimes EA, Raij L (2008) Renoprotection by statins is linked to a decrease in renal oxidative stress, TGF-beta, and fibronectin with concomitant increase in nitric oxide bioavailability. Am J Physiol Renal Physiol 295:F53–F59
Fiore MC, Jimenez PM, Cremonezzi D, Juncos LI, Garcia NH (2011) Statins reverse renal inflammation and endothelial dysfunction induced by chronic high salt intake. Am J Physiol Renal Physiol 301:F263–F270
Feldstein CA (2010) Statins in hypertension: are they a new class of antihypertensive agents? Am J Ther 17:255–262
Borghi C, Veronesi M, Prandin MG, Dormi A, Ambrosioni E (2001) Statins and blood pressure regulation. Curr Hypertens Rep 3:281–288
Wang Y, Chang H, Zou J, Jin X, Qi Z (2011) The effect of atorvastatin on mRNA levels of inflammatory genes expression in human peripheral blood lymphocytes by DNA microarray. Biomed Pharmacother 65:118–122
Zoja C, Corna D, Gagliardini E, Conti S, Arnaboldi L, Benigni A, Remuzzi G (2010) Adding a statin to a combination of ACE inhibitor and ARB normalizes proteinuria in experimental diabetes, which translates into full renoprotection. Am J Physiol Renal Physiol 299:F1203–F1211
Goicoechea M, de Vinuesa SG, Lahera V, Cachofeiro V, Gomez-Campdera F, Vega A, Abad S, Luno J (2006) Effects of atorvastatin on inflammatory and fibrinolytic parameters in patients with chronic kidney disease. J Am Soc Nephrol 17:S231–S235
Strippoli GF, Navaneethan SD, Johnson DW, Perkovic V, Pellegrini F, Nicolucci A, Craig JC (2008) Effects of statins in patients with chronic kidney disease: meta-analysis and meta-regression of randomised controlled trials. BMJ 336:645–651
Campese VM, Ku E, Bigazzi R, Bianchi S (2011) Do HMG-CoA reductase inhibitors improve kidney function? The saga continues. J Nephrol 24:550–553
Kalaitzidis RG, Elisaf MS (2011) The role of statins in chronic kidney disease. Am J Nephrol 34:195–202
Romayne Kurukulasuriya L, Athappan G, Saab G, Whaley Connell A, Sowers JR (2007) HMG CoA reductase inhibitors and renoprotection: the weight of the evidence. Ther Adv Cardiovasc Dis 1:49–59
Matsui I, Hamano T, Tomida K, Inoue K, Takabatake Y, Nagasawa Y, Kawada N, Ito T, Kawachi H, Rakugi H, Imai E, Isaka Y (2009) Active vitamin D and its analogue, 22-oxacalcitriol, ameliorate puromycin aminonucleoside-induced nephrosis in rats. Nephrol Dial Transplant 24:2354–2361
Li YC, Kong J, Wei M, Chen ZF, Liu SQ, Cao LP (2002) 1,25-Dihydroxyvitamin D(3) is a negative endocrine regulator of the renin-angiotensin system. J Clin Invest 110:229–238
Zhang Z, Sun L, Wang Y, Ning G, Minto AW, Kong J, Quigg RJ, Li YC (2008) Renoprotective role of the vitamin D receptor in diabetic nephropathy. Kidney Int 73:163–171
Okamura M, Takano Y, Saito Y, Yao J, Kitamura M (2009) Induction of nephrin gene expression by selective cooperation of the retinoic acid receptor and the vitamin D receptor. Nephrol Dial Transplant 24:3006–3012
Tan X, Wen X, Liu Y (2008) Paricalcitol inhibits renal inflammation by promoting vitamin D receptor-mediated sequestration of NF-kappaB signaling. J Am Soc Nephrol 19:1741–1752
Szeto CC, Chow KM, Kwan BC, Chung KY, Leung CB, Li PK (2008) Oral calcitriol for the treatment of persistent proteinuria in immunoglobulin A nephropathy: an uncontrolled trial. Am J Kidney Dis 51:724–731
Fishbane S, Chittineni H, Packman M, Dutka P, Ali N, Durie N (2009) Oral paricalcitol in the treatment of patients with CKD and proteinuria: a randomized trial. Am J Kidney Dis 54:647–652
Kim MJ, Frankel AH, Donaldson M, Darch SJ, Pusey CD, Hill PD, Mayr M, Tam FW (2011) Oral cholecalciferol decreases albuminuria and urinary TGF-beta1 in patients with type 2 diabetic nephropathy on established renin-angiotensin-aldosterone system inhibition. Kidney Int 80:851–860
de Zeeuw D, Agarwal R, Amdahl M, Audhya P, Coyne D, Garimella T, Parving HH, Pritchett Y, Remuzzi G, Ritz E, Andress D (2011) Selective vitamin D receptor activation with paricalcitol for reduction of albuminuria in patients with type 2 diabetes (VITAL study): a randomised controlled trial. Lancet 376:1543–1551
Agarwal R, Hynson JE, Hecht TJ, Light RP, Sinha AD (2011) Short-term vitamin D receptor activation increases serum creatinine due to increased production with no effect on the glomerular filtration rate. Kidney Int 80:1073–1079
Kumar R (2011) New clinical trials with vitamin D and analogs in renal disease. Kidney Int 80:793–796
Shroff R, Wan M, Rees L (2012) Can vitamin D slow down the progression of chronic kidney disease? Pediatr Nephrol 27:167–173
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Noone, D., Licht, C. Chronic kidney disease: a new look at pathogenetic mechanisms and treatment options. Pediatr Nephrol 29, 779–792 (2014). https://doi.org/10.1007/s00467-013-2436-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-013-2436-5